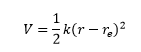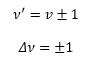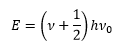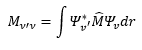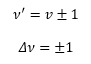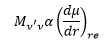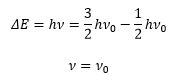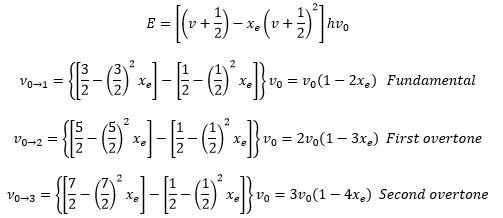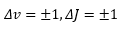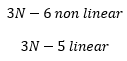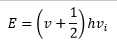Vibrational spectroscopy looks at the differences in energy between the vibrational modes of a molecule. These are larger than the rotational energy states. This spectroscopy can provide a direct measure of bond strength.
The vibration energy levels can be explained using diatomic molecules. To start with they are treated as a simple harmonic oscillator. Classically this means they have a restoring force based on their separation distance r.
Using this we find the classical frequency of vibration is:
This is again in relation to the reduced mass.
With the quantum mechanical treatment of these compounds the energy is found to move in discrete amounts. This gives the energy of the vibration to be discrete:
With the quantum mechanical treatment of these compounds the energy is found to move in discrete amounts. This gives the energy of the vibration to be discrete:
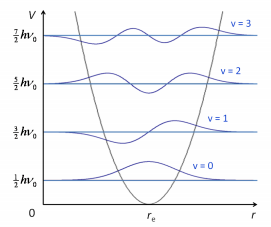
This is where the assumption of simple harmonic motion can be made producing a diagram with equally spaced energy levels.
This is where v is the vibrational quantum number often given by the symbol n. These have the possible values of 0, 1, 2,…. So there are equally spaced levels starting from ½ hv0. With increase vibrational quantum number the probability of finding a molecule in its classical position, approaches the classical limits. The transition the dipole moment is still given by.
Evaluation of the integral reveals for the transition moment to be non-zero we have the selection rule.
This follows that an increase in energy
is a positive change in value and a decrease in energy is a negative change in
value.
It is also found that for the transition
to be present there needs to be a change in dipole moment of the molecule.
This means that there must be a change in the dipole moment during the vibration.
The difference in energy given by a transition is given by:
The difference in energy given by a transition is given by:
This means the absorbed frequency of light equals the classical frequency of molecule vibration. The vibrational frequencies are typically in the range of a few hundred to a few thousand wave numbers. This means absorption occurs in the infrared.
Like rotational spectroscopy as the size of the bond increases the less the restoring force is. This means that there is a problem with the simple harmonic oscillator.
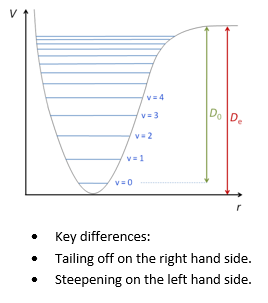
The problem with these as stated is the fact that using the simple harmonic oscillator approach doesn’t take into account that if the molecules are pulled far enough apart they dissociate and if they are pushed too close together they begin to repel. This means that the energy diagram looks more like this: with a block of energy levels at the top:
Like rotational spectroscopy as the size of the bond increases the less the restoring force is. This means that there is a problem with the simple harmonic oscillator.
The problem with these as stated is the fact that using the simple harmonic oscillator approach doesn’t take into account that if the molecules are pulled far enough apart they dissociate and if they are pushed too close together they begin to repel. This means that the energy diagram looks more like this: with a block of energy levels at the top:
This shows how the energy levels are non-degenerate, this is the point of the anharmonic quantum oscillator. This is shown below:
Hot bands are when a vibration occurred that is already in an excited state such as 1 à 3 and overtone are from 0 à2.
The rotations can then be quantised by showing how the fundamental, overtone and hot bands are related. The transitions that start from poorly populated excited states such as hot bands are much weaker than the fundamental bands. The following equation can be used to describe both the fundamental and the overtone bands:
The rotations can then be quantised by showing how the fundamental, overtone and hot bands are related. The transitions that start from poorly populated excited states such as hot bands are much weaker than the fundamental bands. The following equation can be used to describe both the fundamental and the overtone bands:
This gives the fundamental band itself not having a frequency that is precisely v0 this means that it is important to find overtone bands to calculate the anharmonicity constant xe for a small v the result differs very little from the harmonic result.
This leaves the vibration-rotation spectra of diatomic molecules to be found. This can be seen if we look at fine structure. This is where a complete set of rotational and vibrational levels can be seen. Knowing the selection rules are:
This leaves the vibration-rotation spectra of diatomic molecules to be found. This can be seen if we look at fine structure. This is where a complete set of rotational and vibrational levels can be seen. Knowing the selection rules are:
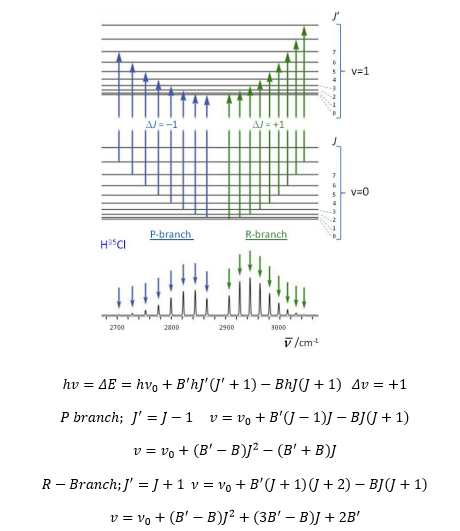
So for each movement of ν there are a large number of J movements. It is known that the lines draw closer at higher frequencies. This is due to the rotational levels becoming closer to one another due to increased inertia.
The transition frequencies are thus given with the overall ν value which then varies down depending on the J value. The diagram below shows how the rotational properties of the molecule can be used with a change in the vibrational frequency of the molecule to give the different branches that form. It is worth noting that these different bands are very close together, this explains why the absorption on an IR spectrum is not discrete but quite a broad line. Even broader when the vibrations have increased rotational freedom.
The transition frequencies are thus given with the overall ν value which then varies down depending on the J value. The diagram below shows how the rotational properties of the molecule can be used with a change in the vibrational frequency of the molecule to give the different branches that form. It is worth noting that these different bands are very close together, this explains why the absorption on an IR spectrum is not discrete but quite a broad line. Even broader when the vibrations have increased rotational freedom.
Vibrational spectra of polyatomic molecules
When it comes to the analysis of polyatomic molecules there are more than one type of vibration that may take place. There are three translational degrees of freedom as all of the molecules are present in three dimensions.
Polyatomic molecules which possess N number of atoms have a certain number of vibrations that are associated with them. These are related depending on whether the molecules are linear or non-linear. The Vibrational nodes that are present are as follows:
When it comes to the analysis of polyatomic molecules there are more than one type of vibration that may take place. There are three translational degrees of freedom as all of the molecules are present in three dimensions.
Polyatomic molecules which possess N number of atoms have a certain number of vibrations that are associated with them. These are related depending on whether the molecules are linear or non-linear. The Vibrational nodes that are present are as follows:
Each vibration behaves as a simple harmonic oscillation and the corresponding energy levels are given by:
Consequently the selection rules are exactly the same as the diatomic case:
If the dipole moment changes in the course of a given vibration as the molecule passes through its equilibrium configuration, that vibration will be active in the infra-red spectrum.
Mutual exclusion principle
If a molecule has a centre of inversion then the mutual exclusion principle applies. This is where transitions seen in the IR spectrum will not be seen in the Raman spectrum, or Raman forbidden. The maths from this is well set in group theory and does not need to be understood but can be easily thought of as the idea that the dipole in the molecule can be represented as vectors so when a inversion symmetry is present then all of the dipole will cancel each other out so to allow either the formation of a dipole so IR active or no formation of a dipole becoming Raman active.
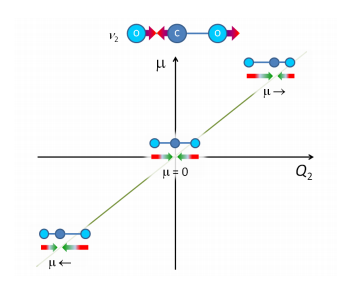
This means for carbon dioxide where there is no permanent dipole moment a dipole has to be produced during the stretch. For molecules with a point of inversion the spectrum can either be completely IR active or completely IR inactive. A good example of this is the asymmetric stretch where the different extremes are all of a different dipole moment so this is IR active throughout.
Mutual exclusion principle
If a molecule has a centre of inversion then the mutual exclusion principle applies. This is where transitions seen in the IR spectrum will not be seen in the Raman spectrum, or Raman forbidden. The maths from this is well set in group theory and does not need to be understood but can be easily thought of as the idea that the dipole in the molecule can be represented as vectors so when a inversion symmetry is present then all of the dipole will cancel each other out so to allow either the formation of a dipole so IR active or no formation of a dipole becoming Raman active.
This means for carbon dioxide where there is no permanent dipole moment a dipole has to be produced during the stretch. For molecules with a point of inversion the spectrum can either be completely IR active or completely IR inactive. A good example of this is the asymmetric stretch where the different extremes are all of a different dipole moment so this is IR active throughout.
When larger molecules are considered the situation becomes rather complicated. This is why there is great importance on studying certain groups. The stretching frequency varies slightly due to changes in the reduced mass of the molecule although the stretches remain similar enough to be quantified.
Working through an example
Knowing how the vibrations are either Raman or IR active and knowing:
The centre of mass of the molecule must not change during the transition.
Predictions of the spectrum can be made.
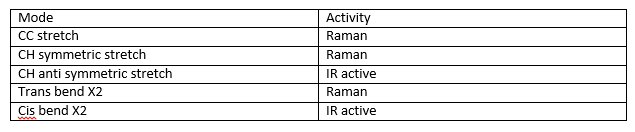
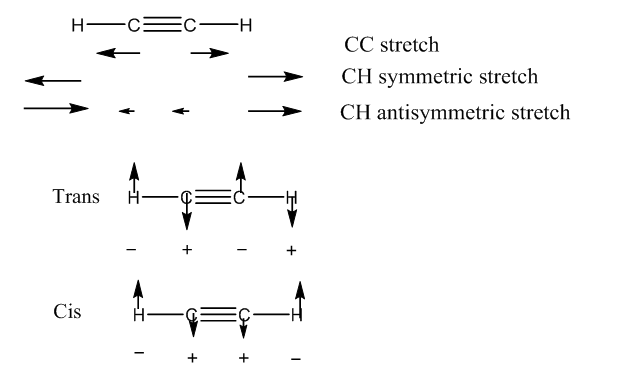
For molecules that involve bending and stretching there are a series of modes that can occur. Using the linear molecule ethyne as an example. H—C==C—H. This has a total of 7 modes:
Working through an example
Knowing how the vibrations are either Raman or IR active and knowing:
The centre of mass of the molecule must not change during the transition.
Predictions of the spectrum can be made.
For molecules that involve bending and stretching there are a series of modes that can occur. Using the linear molecule ethyne as an example. H—C==C—H. This has a total of 7 modes: