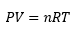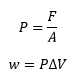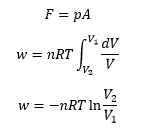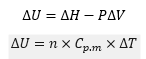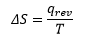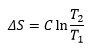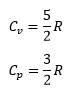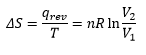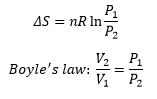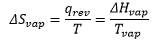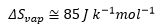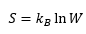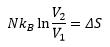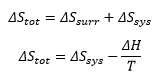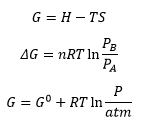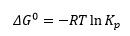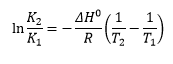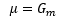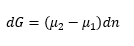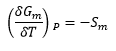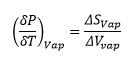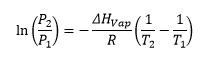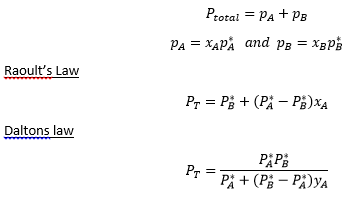The important equations and explanations you'll meet in thermodynamics are discussed here.
Thermodynamics Year 1
Where P is pressure in Pa, V is volume in m3, n is moles, R is the gas constant 8.31 J K-1 mol-1, T is temperature in K.
Work is maximum when expanding against a pressure, work is 0 expanding into a vacuum. P is the outside pressure in Pa.
Work done during volume changes:
Work done during volume changes:
Internal energy and enthalpy:
Year 2
Entropy:
Entropy:
Entropy change on heating assuming that the heat capacity stays constant is:
It is important to remember that:
Entropy change with volume can be given by:
There is also a change of entropy with pressure which is closely related to the above equation through Boyle’s law:
At a state transition it is important to remember that the heat capacity will remain constant causing the following:
Trouton’s law also states that:
Statistical thermodynamics considers the difference found in different molecules and the different ways they can be arranged.
Where W is the number of ways that the molecules can be rearranged.
The number of microstates that can be maintained by molecules can be shown by the equation:
The number of microstates that can be maintained by molecules can be shown by the equation:
When it needs to be found whether a process is spontaneous certain calculations need to be carried out. Such as finding the total entropy change of the system and surroundings.
This allows the Gibbs energy of a system to be calculated for the process. If the Gibbs free energy decreases in value then a process is spontaneous if the Gibbs energy is negative in value then the process is not spontaneous.
This then leaves the Standard Gibbs energy which can be calculated in the above equation and below:
This allows the standard enthalpy to be calculated:
Complex thermodynamics
The chemical potential is identical to the Gibbs free energy of the movement.
The chemical potential is identical to the Gibbs free energy of the movement.
This means that the chemical potential to move one molecule from one system to another is:
The change in Gibbs energy with a change in temperature is equal to the entropy. The entropy is represented as being negative as this is the graph shape formed, it is negative for an increase in T.
The Clapeyron equation relates the gradient of the phase transition with the state functions.
Clasusius Clapeyron equation is used for relating the pressure with the temperature and giving a change in enthalpy.
Binary systems
The pressure of a binary system can be given by the total pressure of the two components.
The pressure of a binary system can be given by the total pressure of the two components.