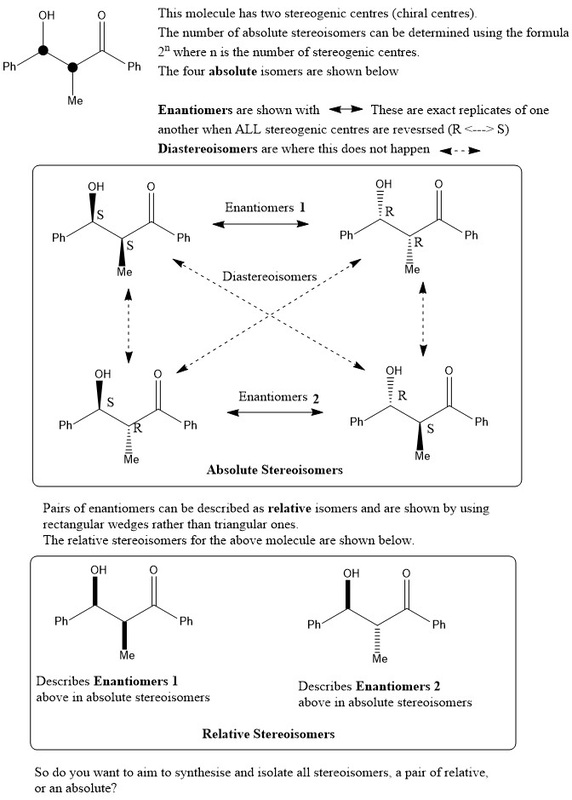
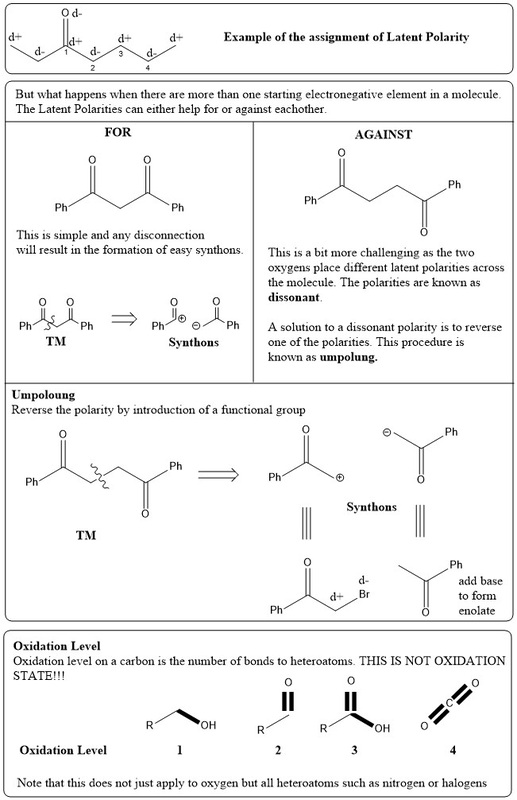
Before we begin you need to know some key terms:
Retrosynthetic analysis
Strategy
Controls; Chemoselectivity, Regioselectivity, Stereoselectivity
Target Molecule (TM)
Disconnection
Synthons
Synthetic Equivalents
Functional Group Interconversion (FGI)
If you do not know what any of these terms actually mean, CLICK HERE to look them up now!
Controls:
Chemoselectivity
Which functional groups react when there are multiple functional groups in a molecule
Example: The selective reduction of a ketone in the presence of an ester using sodium borohydride
Regioselectivity
Where will it react when there are different positions
Example: The regioselective bromination of an aromatic amide
Stereoselectivity
The spatial arrangement of the product molecule
Example: Aldol reaction between benzaldehyde and its enolate
Retrosynthetic analysis
Strategy
Controls; Chemoselectivity, Regioselectivity, Stereoselectivity
Target Molecule (TM)
Disconnection
Synthons
Synthetic Equivalents
Functional Group Interconversion (FGI)
If you do not know what any of these terms actually mean, CLICK HERE to look them up now!
Controls:
Chemoselectivity
Which functional groups react when there are multiple functional groups in a molecule
Example: The selective reduction of a ketone in the presence of an ester using sodium borohydride
Regioselectivity
Where will it react when there are different positions
Example: The regioselective bromination of an aromatic amide
Stereoselectivity
The spatial arrangement of the product molecule
Example: Aldol reaction between benzaldehyde and its enolate