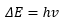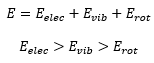The basis of rotational and vibrational spectroscopy is the discrete energy values that are formed in the solving of the wavefunction.
A topic visited regularly in physical chemistry is the fact that molecules have discrete energy levels, given by the solutions of the quantum mechanical Schrodinger equation. The difference in energy levels can be measured by the absorption of certain frequencies of light.
Vibrational spectroscopy is based on using radiation to excite the movement of atoms. The forces required to move atoms are smaller than those required to move electrons. This is because the movement of electrons is based on the movement of electrons away from protons which is pulling on coulomb forces whereas kinetic movement is just based on strength and flexibility of the bonds. This is carried out by using microwave radiation. The molecular vibration levels are closely related to quantum mechanics and anharmoicity.
Energy in a molecule
The energy in a molecule is made up of a mixture of the molecules electronic energy, its vibrational energy and its rotational energy:
Energy in a molecule
The energy in a molecule is made up of a mixture of the molecules electronic energy, its vibrational energy and its rotational energy:
To this translational energies can also
be added although these normally regard the movement of the entire molecule
over a large difference forming an effective continuum. This makes the study of
these movements less important. This shows that there is a relationship between
all of these different energies that make up the total energy.
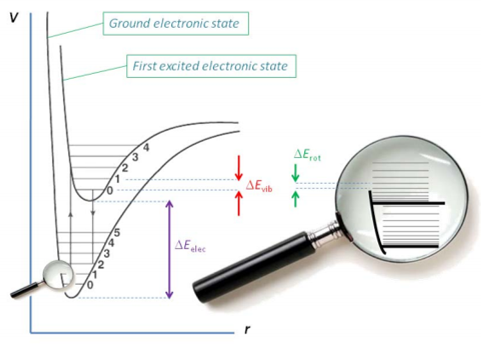
The diagram below shows how the
electronic energy levels make the largest difference in energy, think different
bonding occurring, and how the vibrational energy levels make up the next
biggest energy. These are then subdivided by rotational energy levels. These
are very small in energy and the energy gap increases in size exponentially
across the vibrational energy level. The rotational frequencies are observed in
the microwave region and the far infrared red.
The knowledge of how radiation works to induce an excited state is quite simple. A molecule in its ground state can absorb energy and enter an excited state which can then be removed through relaxation or fluorescence.
The understanding of how frequencies of electromagnetic radiation effect different molecules is an important one. The change in dipole formed on the exposure of a molecule to electromagnetic radiation is what allows electrical circuits to be formed. The other understanding comes from the fact that a series of parameters need to be fulfilled for the transition to be allowed and not forbidden.
The understanding of how frequencies of electromagnetic radiation effect different molecules is an important one. The change in dipole formed on the exposure of a molecule to electromagnetic radiation is what allows electrical circuits to be formed. The other understanding comes from the fact that a series of parameters need to be fulfilled for the transition to be allowed and not forbidden.