Key points in electrochemistry: There are a number of rules that need to be remembered.
Ion selective electrodes
|
The electrochemical cell must have:
· Two electrodes · Electrolytes · Inclusion of a device to measure current or voltage. · A saltbridge if the two solutions cannot mix. |
These have an analytical advantage:
· They have a fast response time as they are continuously measuring of the sample. · Simple to use. · Cheap, small, robust and portable. · High sensitivity and selectivity for the ion. · No interference from colour changes. · Passive measurement (some exceptions). |
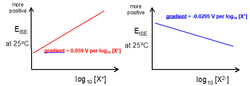
- Double charge:- ½ gradient.
- Positive gradient:- Positive charge.
- Negative gradient:- Negative charge.
Electrodes
It is common for electrodes to have interfering ions and distinct characteristics that need to be learnt.
It is common for electrodes to have interfering ions and distinct characteristics that need to be learnt.
Amperometric sensors
Definition: Electrochemical devices that allow the measurement of the concentration of a selected redox active species througmeasure of flow of current due to oxidation or reduction of species.
Applications There are many applications such as dissolved oxygen, glucose and neurotransmitters.
Analytical advantages: Same as the ISE but the analyte concentration is detected at the electrode surface so can allow many physiological uses.
Electron flow: Oxidation has a positive current.
Reduction has a negative current.
Key uses and features
· Flow of current directly related to analyte.
· Redox chemistry causes current flow.
· Concentration of analyte is lowered by the measurement. It has to be stirred.
· Analyte can be an ion or have no charge, only redox ability is needed although it needs to be able to be oxidised or reduced quickly.
In principle a molecule that reaches the electrode should react although:
· Physical exclusion can stop analytes reaching the sensor electrode.
· Selectivity can be obtained by enzyme use.
· Electrostatic repulsion of other analytes from the sensor electrode can occur.
Doubling the area of the sensor doubles the current.
Definition: Electrochemical devices that allow the measurement of the concentration of a selected redox active species througmeasure of flow of current due to oxidation or reduction of species.
Applications There are many applications such as dissolved oxygen, glucose and neurotransmitters.
Analytical advantages: Same as the ISE but the analyte concentration is detected at the electrode surface so can allow many physiological uses.
Electron flow: Oxidation has a positive current.
Reduction has a negative current.
Key uses and features
· Flow of current directly related to analyte.
· Redox chemistry causes current flow.
· Concentration of analyte is lowered by the measurement. It has to be stirred.
· Analyte can be an ion or have no charge, only redox ability is needed although it needs to be able to be oxidised or reduced quickly.
In principle a molecule that reaches the electrode should react although:
· Physical exclusion can stop analytes reaching the sensor electrode.
· Selectivity can be obtained by enzyme use.
· Electrostatic repulsion of other analytes from the sensor electrode can occur.
Doubling the area of the sensor doubles the current.
|
Voltammetry
To make best use of this ability the following need to be considered: · The reduction potentials and electron stoichiometry of redox reactions. · The reversibility of the redox reactions undergone by a molecule. · Diffusion coefficients of species in stable oxidation states. |
