The stereochemistry and stabilisation of different conformations of cyclohexane is an important part of organic chemistry reactions.
Stereochemistry of cyclohexane
Conformation is how a molecule fills the space that it is in. It can do this in a number of ways and each way can have a different energy. The conformation of compounds is highly important when trying to understand how chirality and regiochemistry might be affected over the course of a reaction.
Introduction
How a molecule sits in space is important when acknowledging how a reaction might occur. Conformational analysis is based on the idea that if there is free rotation of a bond although an infinite number of conformations can be produced. Some will be more favourable than others allowing the possibility for a majority state to form.
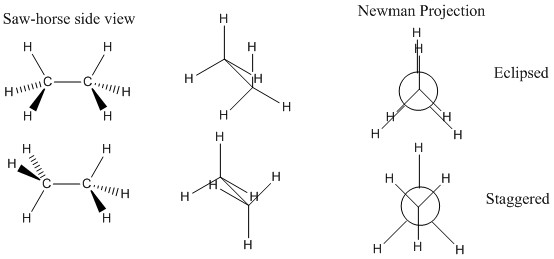
These can be represented in two different ways: The sawhorse and the Newman projections. These allow the different bonds to be seen as a function of their angle to the plane and their angle from each other.
Conformation is how a molecule fills the space that it is in. It can do this in a number of ways and each way can have a different energy. The conformation of compounds is highly important when trying to understand how chirality and regiochemistry might be affected over the course of a reaction.
Introduction
How a molecule sits in space is important when acknowledging how a reaction might occur. Conformational analysis is based on the idea that if there is free rotation of a bond although an infinite number of conformations can be produced. Some will be more favourable than others allowing the possibility for a majority state to form.
These can be represented in two different ways: The sawhorse and the Newman projections. These allow the different bonds to be seen as a function of their angle to the plane and their angle from each other.
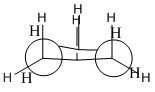
These projections show ethane in two different conformations. Both of these conformations are very important as one represents the eclipsed and the other the staggered conformation. The staggered formation is the one that is most thermodynamically favourable. The eclipsed is the least favourable and has the highest energy.
The reason for the high energy of the staggered formation is the fact that the atoms which naturally repel each other are closer together in space.
Orbital theory
The process of stabalisation seen in the Staggered formation is the interaction between the bonding σ of one carbon-hydrogen bond and the antibonding σ* of the second carbon-hydrogen bond. These interactions occur between carbons and is known as hyper conjugation.
When it comes to the butane the two carbon atoms repel each other even more causing an even less favourable staggered state.
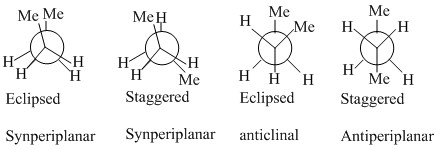
The study of butane is very important as there is the possibility of multiple staggered states. These have different names given to them. The difference in energies between these show that there is still a difference in interactions between the two methyl groups and the methyl and the hydrogen groups.
The reason for the high energy of the staggered formation is the fact that the atoms which naturally repel each other are closer together in space.
Orbital theory
The process of stabalisation seen in the Staggered formation is the interaction between the bonding σ of one carbon-hydrogen bond and the antibonding σ* of the second carbon-hydrogen bond. These interactions occur between carbons and is known as hyper conjugation.
When it comes to the butane the two carbon atoms repel each other even more causing an even less favourable staggered state.
The study of butane is very important as there is the possibility of multiple staggered states. These have different names given to them. The difference in energies between these show that there is still a difference in interactions between the two methyl groups and the methyl and the hydrogen groups.
Conformation of cyclohexane
There are a number of different conformations of cyclohexane. The different shapes that can be formed are important as the energy varies with each one but the integrity needs to be maintained across the molecule and at each bond.
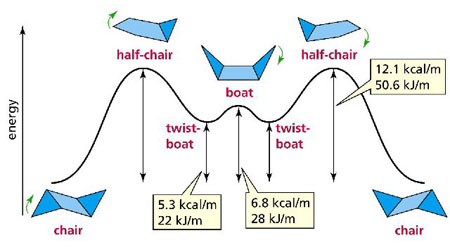
There are 4 important structures that are present in a hexane molecule these are the chair, half-chair, boat, twist-boat.
Now the structures themselves aren’t very complicated, it is just important to learn why the structures are at the energy levels they are found at.
There are a number of different conformations of cyclohexane. The different shapes that can be formed are important as the energy varies with each one but the integrity needs to be maintained across the molecule and at each bond.
There are 4 important structures that are present in a hexane molecule these are the chair, half-chair, boat, twist-boat.
Now the structures themselves aren’t very complicated, it is just important to learn why the structures are at the energy levels they are found at.
Chair conformation
The stereochemistry of the chair formation shows how there are no eclipsing C-H bonds. All of the bonds are fully staggered, giving the lowest energy possible. This is why cyclohexane is strain-free. This is the most common cyclohexane conformation to see due to it being lowest in energy.
The stereochemistry of the chair formation shows how there are no eclipsing C-H bonds. All of the bonds are fully staggered, giving the lowest energy possible. This is why cyclohexane is strain-free. This is the most common cyclohexane conformation to see due to it being lowest in energy.
Boat formation
In the boat formation the C-H bonds are not eclipsed and there are bad repulsion interactions between the flagstaff C-H bonds. Although the boat conformation is free from angle strain the repulsive forces between the flag staff C-H bonds can be avoided if there is a slight twist in the structure.
In the boat formation the C-H bonds are not eclipsed and there are bad repulsion interactions between the flagstaff C-H bonds. Although the boat conformation is free from angle strain the repulsive forces between the flag staff C-H bonds can be avoided if there is a slight twist in the structure.
Half-Chair formation
The half-chair formation is a high energy intemediate that is formed in any ring-flip conformation changes. It is important when drawing this to show the "back" three hydrogen atoms, doing the moving, to be in a middle state. The formation finishes in either the boat or chair formation so check that it will form these as an end product.
The half-chair formation is a high energy intemediate that is formed in any ring-flip conformation changes. It is important when drawing this to show the "back" three hydrogen atoms, doing the moving, to be in a middle state. The formation finishes in either the boat or chair formation so check that it will form these as an end product.
Energy of a system
When considering the energy of a system the following strains need to be considered. These are the:
Torsional strain: This is the strain caused by molecular bonding orbitals and their relationship to one another. It is known that the bonding and stabilisation of antibonding orbitals can help bring the energy down whereas two bonding orbitals being close together can cause a large amount of repulsion. This is the bases of eclipsed and staggered bonding.
Steric strain: This occurs when two groups are brought close to one another. The clashing that his causes raises the energy meaning they try and move apart from one another. This causes an increase in energy in the system.
Angle strain: Angle strain is an important last point to remember as large angle strains can limit the plausibility of a number of different structures.
Torsional strain
Torsional strain is the strain which is caused by the presence of different atoms in the molecule and where the bonds lie. The torsional strain is one of the most important types of strain that there is in a molecule and can add the largest amount of energy when the molecule is not shaped correctly. The biggest example of this is cyclohexane in a planar phase. It can be easily seen here that there is so much eclipsing of bonds that the torsional strain is so high a completely flat cyclohexane molecule is never produced at any stage in cyclohexanes conformation.
When considering the energy of a system the following strains need to be considered. These are the:
Torsional strain: This is the strain caused by molecular bonding orbitals and their relationship to one another. It is known that the bonding and stabilisation of antibonding orbitals can help bring the energy down whereas two bonding orbitals being close together can cause a large amount of repulsion. This is the bases of eclipsed and staggered bonding.
Steric strain: This occurs when two groups are brought close to one another. The clashing that his causes raises the energy meaning they try and move apart from one another. This causes an increase in energy in the system.
Angle strain: Angle strain is an important last point to remember as large angle strains can limit the plausibility of a number of different structures.
Torsional strain
Torsional strain is the strain which is caused by the presence of different atoms in the molecule and where the bonds lie. The torsional strain is one of the most important types of strain that there is in a molecule and can add the largest amount of energy when the molecule is not shaped correctly. The biggest example of this is cyclohexane in a planar phase. It can be easily seen here that there is so much eclipsing of bonds that the torsional strain is so high a completely flat cyclohexane molecule is never produced at any stage in cyclohexanes conformation.
Chem draw hated this one.
This shows why the boat conformation is favoured over all others, there is no torsional strain from the hydrogen bonds meaning that the energy is much lower than any other cyclohexane group.
Steric strain
Another reason why the chair formation is the most favoured and why other molecules tend to favour some sort of non-linear shape is that this helps to minimize steric strain. The boat structure is much less favoured due to the presence of two flagstaff hydrogen atoms causing steric strain on the molecule.
This can be effected by the any heteronuclear atoms present in the ring. This can bring any subsitutents closer to one another and therefore cause an increase in steric strain. If the bonds are slightly elongated then this can work the other way producing less steric strain. This is explained in more detail below.
Angle strain
For small rings the angle strain and torsional strain is high making the production of small rings usually unfavourable. The production of small rings causes a large amount of angle strain as molecules can be forced to bend to 90° or even 60° (for 4 and 3 membered rings) where the favoured angle is 107°. For large rings the idea of angle strain basically disappears as the multiple degrees of freedom allow a large amount of twisting to occur to minimise any angle strain. The best way to calculate angle strain is to compare the heats of combustion of cycloalkane molecules to their straight chain counterparts.
Constituents and their effects
So far we have only considered hydrogen being bonded to the cyclohexane ring. This will obviously not always be the case and it is likely that there will be a large variation in the possible groups that will be seen attached to the ring. It is important to keep in mind how other constituents act and how they effect the energy.
When constituents are added to the cyclohexane ring the conformations available change rapidly, there is also a change to where each group can be found.
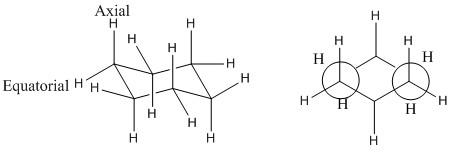
It is known that hydrogen atoms take either an axial or an equatorial position. In the case of cyclohexane there is as likely a hydrogen to be present in either the axial position as it is to be found in the equatorial position. This means that the cyclohexane ring is free to transfer between each conformation rapidly.
This shows why the boat conformation is favoured over all others, there is no torsional strain from the hydrogen bonds meaning that the energy is much lower than any other cyclohexane group.
Steric strain
Another reason why the chair formation is the most favoured and why other molecules tend to favour some sort of non-linear shape is that this helps to minimize steric strain. The boat structure is much less favoured due to the presence of two flagstaff hydrogen atoms causing steric strain on the molecule.
This can be effected by the any heteronuclear atoms present in the ring. This can bring any subsitutents closer to one another and therefore cause an increase in steric strain. If the bonds are slightly elongated then this can work the other way producing less steric strain. This is explained in more detail below.
Angle strain
For small rings the angle strain and torsional strain is high making the production of small rings usually unfavourable. The production of small rings causes a large amount of angle strain as molecules can be forced to bend to 90° or even 60° (for 4 and 3 membered rings) where the favoured angle is 107°. For large rings the idea of angle strain basically disappears as the multiple degrees of freedom allow a large amount of twisting to occur to minimise any angle strain. The best way to calculate angle strain is to compare the heats of combustion of cycloalkane molecules to their straight chain counterparts.
Constituents and their effects
So far we have only considered hydrogen being bonded to the cyclohexane ring. This will obviously not always be the case and it is likely that there will be a large variation in the possible groups that will be seen attached to the ring. It is important to keep in mind how other constituents act and how they effect the energy.
When constituents are added to the cyclohexane ring the conformations available change rapidly, there is also a change to where each group can be found.
It is known that hydrogen atoms take either an axial or an equatorial position. In the case of cyclohexane there is as likely a hydrogen to be present in either the axial position as it is to be found in the equatorial position. This means that the cyclohexane ring is free to transfer between each conformation rapidly.
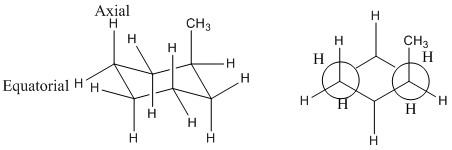
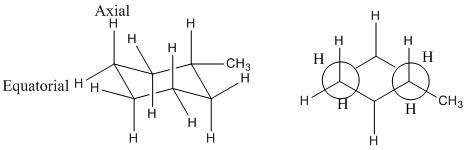
This changes when consituents are added to each ring. An example of this is where a methyl group is added to a cyclohexane ring. It can be seen that on addition of the methyl group the cyclohexane ring is no longer symmetrical over all points meaning that the conformation and where the methyl group is placed makes a big difference.
The methyl group when placed in the axial position has increase steric interactions between the other axial hydrogen atoms. It can also be seen that there is an anticlinal interaction with the another carbon this can be better seen by only looking at one side of the ring.
The methyl group when placed in the axial position has increase steric interactions between the other axial hydrogen atoms. It can also be seen that there is an anticlinal interaction with the another carbon this can be better seen by only looking at one side of the ring.
When the methyl group is placed in the equatorial position then there are no interactions with other hydrogens as they are now staggered and the synclinal interactions are not present.
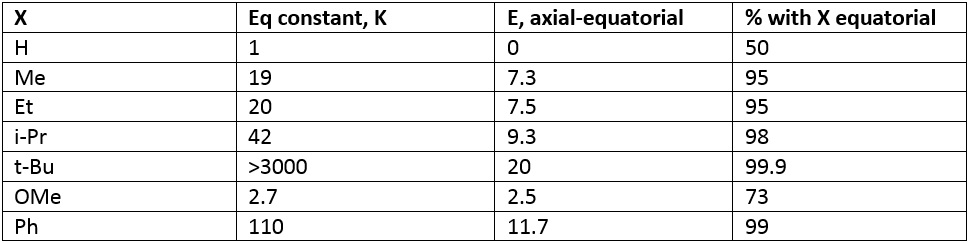
Different substituents have different effects on the ring and how they affect the energy.
Iso-propane has a 98% equatorial percentage chance. This value is very different from tert-butane because there is the presence of one hydrogen. This hydrogen can rotate into place so there is no direct steric contact. This keeps the energy slightly lower although it is still heavily bias to one side. When looking at tert-butane it is easy to see how the size of the molecule has a large effect. The tert-butane molecule can act as a locking mechanism keeping the cyclohexane in the designated chair formation. It is almost impossible for ring flip to occur in this formation.
The above table also shows the effect of a smaller atom being present. The oxygen group is smaller than a methyl group so has less of an effect and there is also an attraction between the oxygen and the hydrogen molecules. This slightly raises the angle strain as well as the torsional strain present in the system.
When it comes to deciding the shape of the molecule the different groups need to be looked at and the effects of each estimated. A rough count of how many diaxial interactions and gauche interactions are present can give a rough idea to the shape of the molecule.
Heteroatoms in or on rings
The presence of heteroatoms on the ring can cause a number of conformation property changes. The most important of these being the relationship between the bond angles and the bond lengths caused by the presence of different angles. With an increase of bond length comes an increase in bond angle although the substituents will be held further apart. This is an important point and is one that needs to be calculated.
The above table also shows the effect of a smaller atom being present. The oxygen group is smaller than a methyl group so has less of an effect and there is also an attraction between the oxygen and the hydrogen molecules. This slightly raises the angle strain as well as the torsional strain present in the system.
When it comes to deciding the shape of the molecule the different groups need to be looked at and the effects of each estimated. A rough count of how many diaxial interactions and gauche interactions are present can give a rough idea to the shape of the molecule.
Heteroatoms in or on rings
The presence of heteroatoms on the ring can cause a number of conformation property changes. The most important of these being the relationship between the bond angles and the bond lengths caused by the presence of different angles. With an increase of bond length comes an increase in bond angle although the substituents will be held further apart. This is an important point and is one that needs to be calculated.
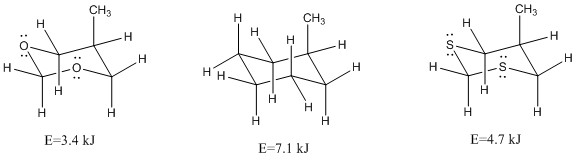
At the longer bond lengths the angles become much smaller and are much more subject to change. This distortion causes a change in the torsion angle this usually increases to 65°. Heteroatoms in the ring normally have lone pairs of electrons present, these are less sterically demanding than the C-H bond. This can mean that the axial position may be favoured for the some atoms as the only interactions are with the lone pairs of electrons in the axial face also.
The shorter bond lengths that are caused by the presence of oxygen in the ring can cause a much larger steric repulsion between the bonds present.
The shorter bond lengths that are caused by the presence of oxygen in the ring can cause a much larger steric repulsion between the bonds present.
This shows how the smaller orbitals of oxygen cause less interaction then the larger orbitals of sulphur.
Configuration changes
Although we are now use to assuming that the conformation is all important and that the configuration of a molecule cannot change. This is not true for heteroatoms. It is possible if the heteroatom is electronegative enough the ring can break forming a C=O bond and a C+ cation. This is then free to rotate and place the constituents in the favoured group. This leads nicely onto the anomeric effect.
Anomeric effect
The anomeric effect leads to question all of the principles that have been discussed so far in configuration and conformation chemistry.
If a highly electronegative atom is found in the ring the favoured positions of consituents can change. In this case they mostly become axial positioned rather than equatorial positioned. This is explained below.
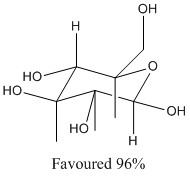
Glucose (shown below) has a number of consituents, as expected the alcohol groups are beterr place at equatorial positions to minimize steric interactions and therefore decrease the total energy in the molecule. This corresponds to the theories discussed above.
Configuration changes
Although we are now use to assuming that the conformation is all important and that the configuration of a molecule cannot change. This is not true for heteroatoms. It is possible if the heteroatom is electronegative enough the ring can break forming a C=O bond and a C+ cation. This is then free to rotate and place the constituents in the favoured group. This leads nicely onto the anomeric effect.
Anomeric effect
The anomeric effect leads to question all of the principles that have been discussed so far in configuration and conformation chemistry.
If a highly electronegative atom is found in the ring the favoured positions of consituents can change. In this case they mostly become axial positioned rather than equatorial positioned. This is explained below.
Glucose (shown below) has a number of consituents, as expected the alcohol groups are beterr place at equatorial positions to minimize steric interactions and therefore decrease the total energy in the molecule. This corresponds to the theories discussed above.
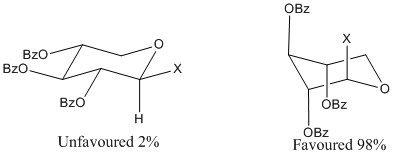
This though is a rarity, it is usually found that the electronegative constituents are found in the axial position when there is an electronegative atom in the anomeric position. This is due to stereoelectronic effects. These are interactions between the consituents and the lone pair orbitals of the electronegative groups. This is shown using xylose ester below:
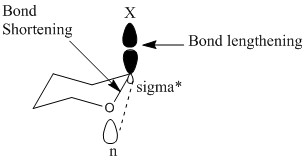
This is due to the antibonding orbitals of the carbon- halogen bond, here represented by C-X. The interactions between these orbitals allow further stabilisation making it much more favourable for the molecule to be found in this state than any other.
These relations also increase the bond length making it much more favourable to be found in this arrangement.
These relations also increase the bond length making it much more favourable to be found in this arrangement.
Basis done, may add more, most should already be here, 13/02/14. 01:13 TM.