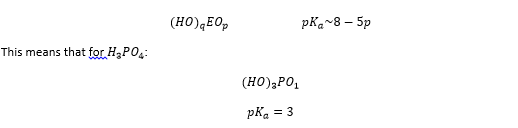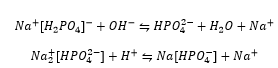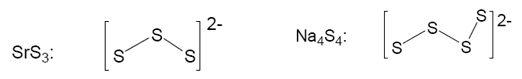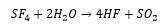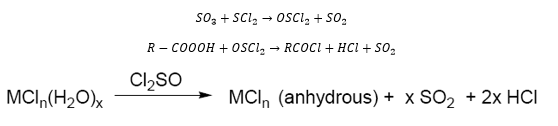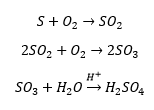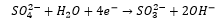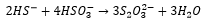Group 16 contains a large mixture of properties for the elements that are present, Oxygen and sulphur are the two elements that are mostly looked at.
Acidic properties of the group 16 compounds
Bronsted acids and bases are based on the idea of donating or accepting a proton. The basis of many acids is a hydroxyl group bonded to another element. The basis of many acids is the capability of this oxygen to be deprotonated to give a proton (in the form of a hydroxonium ion).
Pauling developed a rule for oxyacids to create a system for estimating the pKa value for an acid with an estimated structure. Taking H3PO4 as an example:
Paulings rule:
Acidic properties of the group 16 compounds
Bronsted acids and bases are based on the idea of donating or accepting a proton. The basis of many acids is a hydroxyl group bonded to another element. The basis of many acids is the capability of this oxygen to be deprotonated to give a proton (in the form of a hydroxonium ion).
Pauling developed a rule for oxyacids to create a system for estimating the pKa value for an acid with an estimated structure. Taking H3PO4 as an example:
Paulings rule:
Phosphoric acid is polyprotic more than one value for pKa can be used to describe this species. This can also be described by Pauling’s rule showing that every proton lost causes another oxygen to form therefore raising the pKa.
Buffers
A buffer is an aqueous solution containing a weak acid and its conjugate base or a weak base conjugate acid. The pH will change very little if a small amount of strong acid or base is added. This can be done very easily with oxyacids as this can be designed to be able to take both the protons and the base and in the event that the acid is polyprotic a large amount of base can be added and pH will only rise to a certain point until the acid is fully deprotonated.
An example with phosphoric acid:
The phosphoric acid can be designed as a buffer by adding a mixture of the acid and its conjugate base equivalent in this case that can be designed by the following:
Buffers
A buffer is an aqueous solution containing a weak acid and its conjugate base or a weak base conjugate acid. The pH will change very little if a small amount of strong acid or base is added. This can be done very easily with oxyacids as this can be designed to be able to take both the protons and the base and in the event that the acid is polyprotic a large amount of base can be added and pH will only rise to a certain point until the acid is fully deprotonated.
An example with phosphoric acid:
The phosphoric acid can be designed as a buffer by adding a mixture of the acid and its conjugate base equivalent in this case that can be designed by the following:
This shows the removal of both a proton and hydroxide by the same solution.
Oxygen
Oxygen only has two allotropes unlike the other group 16 elements that tend to have a larger number of possible allotropes.
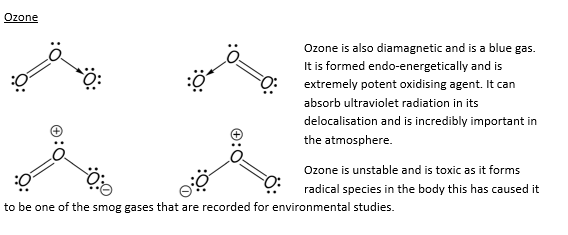
The two allotropes of oxygen are di-oxygen and ozone (tri-oxygen).
Oxygen
Oxygen only has two allotropes unlike the other group 16 elements that tend to have a larger number of possible allotropes.
The two allotropes of oxygen are di-oxygen and ozone (tri-oxygen).
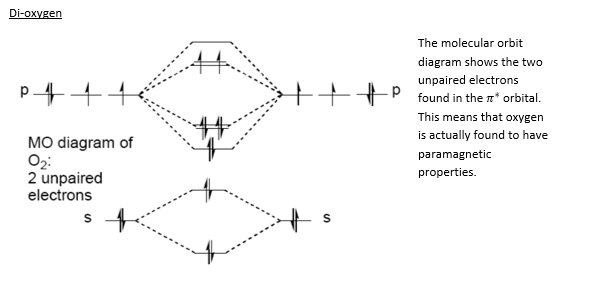
Oxygen is rarely found with an coordination number greater than 3 this is mostly due to its high electronegativity. It forms double bonds with itself due to the smaller orbials that surround the atom. These double bonds make the di-oxygen very strong with a bond enthalpy of 494 kJmol-1 although many of the reactions involving oxygen are combustion in nature due to the high bond enthalpy that oxygen has single bonded to many different atoms.
Oxygen does not form chains of molecules due to the high repulsion present from the two double bonds this means that the reaction involving another oxygen molecule rarely occurs and when it does, in the case of ozone, the bond is unstable.
Sulfur
One of the most “intresting” elements present in chemistry. Sulfer produces a large number of allotropes and is very similar in overall properties to carbon.
Sulfur is most stable in the cyclic S8 which consists of crown like 8 membered rings on heating this strucuture rearranges forming more and more dense molecules. Although on cooling the molecule reverts back to the cyclic structure. This shows a great importance in sulfur chemistry that the sulfur atoms prefers single bonds over double bonds. Rings are high as S20 have been found and isolated although for many of these higher level crystals they can become unstable and slowly revert back to the S8 ring.
Sulfur is found as a vapour as S2 this has a triplet ground state, making it paramagnetic. Disulfur has a blue-violet colour and is stable only at high temperature. S3 is cherry red in colour and is only stable at a high temperature.
Sulfur is intresting as it forms complexes with up to 6 coordination numbers. Sulphur ignites in flourine forming SF6 and reacts with chlorine and bromine giving S2X2 the latter readily dissociates. Liquid iodine does not react with sulfer which means it can actually be used as a low temperature solvent.
Metal bonding of sulfur
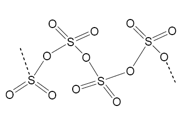
Sulfur can bond to numerous ores because of a tendency to make polysulfides, these polysulfide structures are long linear compounds that have an overall negative charge. These structures can then bond ionically with counter ions. The diagram below shows the linear structures that are produced by these compounds:
Oxygen does not form chains of molecules due to the high repulsion present from the two double bonds this means that the reaction involving another oxygen molecule rarely occurs and when it does, in the case of ozone, the bond is unstable.
Sulfur
One of the most “intresting” elements present in chemistry. Sulfer produces a large number of allotropes and is very similar in overall properties to carbon.
Sulfur is most stable in the cyclic S8 which consists of crown like 8 membered rings on heating this strucuture rearranges forming more and more dense molecules. Although on cooling the molecule reverts back to the cyclic structure. This shows a great importance in sulfur chemistry that the sulfur atoms prefers single bonds over double bonds. Rings are high as S20 have been found and isolated although for many of these higher level crystals they can become unstable and slowly revert back to the S8 ring.
Sulfur is found as a vapour as S2 this has a triplet ground state, making it paramagnetic. Disulfur has a blue-violet colour and is stable only at high temperature. S3 is cherry red in colour and is only stable at a high temperature.
Sulfur is intresting as it forms complexes with up to 6 coordination numbers. Sulphur ignites in flourine forming SF6 and reacts with chlorine and bromine giving S2X2 the latter readily dissociates. Liquid iodine does not react with sulfer which means it can actually be used as a low temperature solvent.
Metal bonding of sulfur
Sulfur can bond to numerous ores because of a tendency to make polysulfides, these polysulfide structures are long linear compounds that have an overall negative charge. These structures can then bond ionically with counter ions. The diagram below shows the linear structures that are produced by these compounds:
There are a number of economically important sulfur ores although many of them follow the same pattern come of the versions that don’t follow this pattern are shown below:
Hydrogen sulfide is a weak acid, the reason behind its slight acidity is due to the weaker bond caused by the larger difference in relative orbital energies, sulfur is also a bigger atom and can stabalise the negative charge better.
Sulfur halides
SF4 can rapidly hydrolyse to form HF and SO2 this makes this compound a selective fluorinating agent being able to convert ketones into alkylhalides.
Sulfur halides
SF4 can rapidly hydrolyse to form HF and SO2 this makes this compound a selective fluorinating agent being able to convert ketones into alkylhalides.
There can also be the addition of an increased number of fluoride groups to form S2F10 this disproportionates into SF4 and SF6. SF6 is a very stable chemically inert gas caused by a large electron density. These are used as insulating gases for high-volt generators and are often used as a blanketing gas due to its high density. It also has small use in medical applications.
Sulfur oxyhalides
Sulfur oxyhalides are also very important as the reaction between sulfuroxide and sulfurdichloride to form thionyl chloirde. Which can then be used as a chlorination and dehydration agent. This can take part in the following:
Sulfur oxyhalides
Sulfur oxyhalides are also very important as the reaction between sulfuroxide and sulfurdichloride to form thionyl chloirde. Which can then be used as a chlorination and dehydration agent. This can take part in the following:
Oxides of sulfur
These are made on an industrial scale by roasting sulfide ores in air. This releases a large amount of sulfur dioxide gas which causes acid rain etc. There has been a large amount of research into its effects and how they can be hindered. These are made on an industrial scal be roasting sulfide ores in air. This releases a large amount of sulfur dioxide gas which causes acid rain etc. There has been a large amount of research into its effects and how they can be hindered.
A solution of SO2 in water forms a species that is described as H2SO3 although this is untrue as the molecule “H2SO3” doesn't actually forms. A mixture of SO2(H2O) forms which produces H3O+ ions.
These are made on an industrial scale by roasting sulfide ores in air. This releases a large amount of sulfur dioxide gas which causes acid rain etc. There has been a large amount of research into its effects and how they can be hindered. These are made on an industrial scal be roasting sulfide ores in air. This releases a large amount of sulfur dioxide gas which causes acid rain etc. There has been a large amount of research into its effects and how they can be hindered.
A solution of SO2 in water forms a species that is described as H2SO3 although this is untrue as the molecule “H2SO3” doesn't actually forms. A mixture of SO2(H2O) forms which produces H3O+ ions.
SO3 sulfur trioxide can be made on a large scale by SO2 oxidation. This is the basis for H2SO4 manufacture. This is carried out over a catalyst which is usually Pt gauze. This forms the polymeric chain shown below:
This is a polymeric reaction and below 16.8°C even forms ice like crystals. The helical chain structure is reminisent of most polyphosphates. Oxyacids of sulfur are highy potent even being able to protonate nitric acid. This is important in the formation of the nitrogen oxide cation which is used in the nitration of aromatics. At its melting point H2S2O7 can degrade forming disulfate. These are sulfur oxy anions which are pyramidal in structure and can be used as a reducing agent especially in basic solution.
The sulfate structure is pyramidal.
Thiosulfate can be produced from:
Thiosulfate can be produced from:
This is a mild reducing agent and can be used in idometric titrations.
Sulfur oxy anions:
Sulfur oxy anions:
Sulphides also form the zinc blend structure and are usually semiconductors that are light sensitive. The same can be produced using the Cadium mercury telluride structures which are low band gap materials and are usually in infrared dectectors and heat seeking devices.
Selenium and tellurium are both chemicals which are similar to sulfates. They can produce semicondctor films and the E-H bond decreases strongly down the group.
The main point that has to be made is that sulphuric acid is a very potent dehydration agent making it extremely dangerous for use in laboratorys this is due to the fact that H2SO4 will form hydrates very easily drawing water away from what ever it is in contact with.
Selenium and tellurium are both chemicals which are similar to sulfates. They can produce semicondctor films and the E-H bond decreases strongly down the group.
The main point that has to be made is that sulphuric acid is a very potent dehydration agent making it extremely dangerous for use in laboratorys this is due to the fact that H2SO4 will form hydrates very easily drawing water away from what ever it is in contact with.