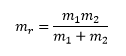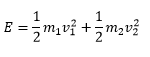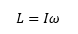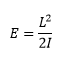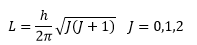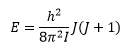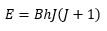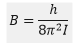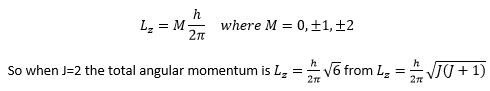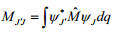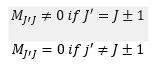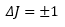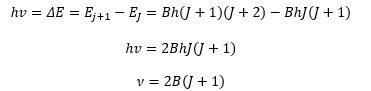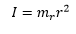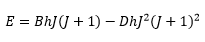Rotational spectroscopy uses the discrete energy levels of rotation to measure the inertia of the bond and therefore the bond length.
Rotational spectroscopy
Rotation spectroscopy measures how the rotation of a molecule about its centre mass can give an idea of inertia and therefore bond length. The excitation of a molecule can cause the molecule to start rotating around a point, the energy of this movement is quantised around the point. It is important to note that these transitions can only occur between molecules that do not have an inversion of symmetry.
The value for is the reduced mass of the molecule:
Rotation spectroscopy measures how the rotation of a molecule about its centre mass can give an idea of inertia and therefore bond length. The excitation of a molecule can cause the molecule to start rotating around a point, the energy of this movement is quantised around the point. It is important to note that these transitions can only occur between molecules that do not have an inversion of symmetry.
The value for is the reduced mass of the molecule:
The masses of the molecules have a very real effect on how the molecule rotates and pivots around the molecule. This can give the energy as two masses and then as a subject of inertia:
These two atoms have to move at different speeds, proportional to their distances from the centre of mass otherwise the bond would begin to bend inwards. This is shown by:
And relating this to inertia:
The angular momentum can now be related very closely to kinetic energy, this leaves the angular momentum to be given as:
The rotational energy can then be written as terms of inertia and angular momentum:
This only leaves the quantum mechanical result to be achieved by using Schrodinger’s equation. Which isn’t necessary as we can use the shortcut:
This is where J is the rotational quantum number, the angular momentum that is a vector directed along an axis of rotation with magnitude L. This shows how the value of J is an integer in every molecule, the difference is caused by the change in value of the inertia I.
The energies of this can therefore be given by:
The energies of this can therefore be given by:
Or:
As B and h are constants given by:
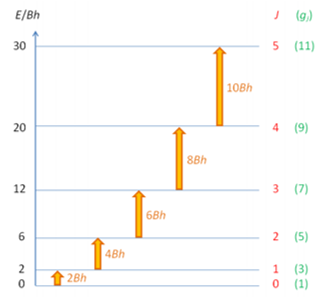
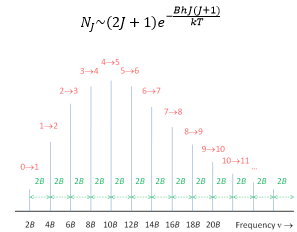
B is the rotational constant of the molecule with units of Hz. The first few rotational energy levels are shown below:
Because the spacing between rotational levels depends on B, which is inversely proportional to the moment of inertia the levels become more and more tightly packed as inertia increases. This is true for both linear and non-linear molecules.
The degeneracy of the angular momentum is also important it is quantised and related to quantum mechanical principles also:
The degeneracy of the angular momentum is also important it is quantised and related to quantum mechanical principles also:
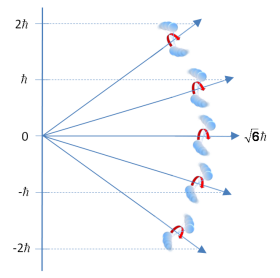
The degeneracy causes 2J+1 energy values to form. As the value of L is the angular momentum vector when there is no external force acting on the molecule L is unchanged. So there is no preferred direction when there is no magnetic field present so all values of M have the same energy. When a magnetic field is induced along an axis any extra energy produced needs to be displaced. The vectors can vary by multiples of h/2π. This is shown in the diagram below:
The degeneracy is important as with an increase in degeneracy there is a greater distribution of molecules in various energy states. Having found a formula for the rotational vibration energy levels and knowing that a transition occurs if there is a transition dipole moment, an experiment can be designed to measure the rotational energy levels.
The equation above represents how for a dipole moment is directly related to the azimuthal quantum number.
There is a great importance on what transitions need to occur from these vibrations. Now we know that they occur and what degeneracy they have it is important that when the molecule is irradiated the light responds in a certain way. For the rotational spectrum to show there needs to be a permanent dipole moment present. So the molecular quantum number has to move for a transition to be present this is represented by the following:
There is a great importance on what transitions need to occur from these vibrations. Now we know that they occur and what degeneracy they have it is important that when the molecule is irradiated the light responds in a certain way. For the rotational spectrum to show there needs to be a permanent dipole moment present. So the molecular quantum number has to move for a transition to be present this is represented by the following:
This leaves the selection rule:
Knowing an increase in J means that there is an increase energy on absorption and a reduction in J means that there has been an emission
This means that the absorption frequencies in a vibration can be given by:
This means that the absorption frequencies in a vibration can be given by:
The spectrum therefore shows absorption lines at 2B, 4B etc. because there is only a small splitting energy between these the low J values are mostly filled at room temperature. This is governed by the Boltzmann factor.
The spacing in the diagram can give the value of B. This is a great example of how rotational spectroscopy can then be used to find a large number of key properties of the molecule being analysed.
Now that we have B and the inertia value the bond length can be calculated. This is useful for analysis methods and for further calculation:
Now that we have B and the inertia value the bond length can be calculated. This is useful for analysis methods and for further calculation:
In a polyatomic molecule it can be much harder to find bond lengths of each of the molecules. This can be helped by using isotopic substitution and assuming the bond lengths are unchanged on isotopic substitution which is not quite true.
The observed frequencies work well for low J values but not for high J values. When centrifugal stretching of the molecule is taken into account a correction term needs to be added. This shows how the centrifugal constant has quite a large effect on the matter.
The observed frequencies work well for low J values but not for high J values. When centrifugal stretching of the molecule is taken into account a correction term needs to be added. This shows how the centrifugal constant has quite a large effect on the matter.
This is where D is the centrifugal distortion constant, this is inversely proportional to the moment of inertia.
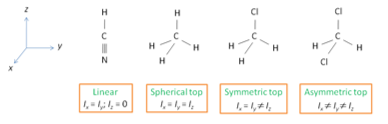
Non-linear polyatomic molecules.
For these molecules there is a stress on which “top” each molecule is.
Non-linear polyatomic molecules.
For these molecules there is a stress on which “top” each molecule is.
Spherical top molecules have similar rotational levels to linear molecules but they do not have a dipole moment. This means there cannot be a rotation spectra. Symmetric and asymmetric top molecules do give vibration spectra. Knowing the moment of inertia of these molecules allows the prediction of bond lengths and bond angles. On the whole, rotational spectroscopy provides a useful method of determining molecular structure.