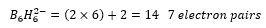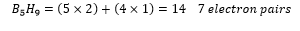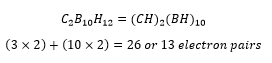Cluster chemistry
Boranes usually form clusters in the right chemical environments. Due to the ability for the 2c-3e bond a number of electrons can be available for cluster bonding to occur. This does mean that the same amount of boron atoms can have a different number of hydrogen atoms due to the presence of either a 2c 3e bond with another boron or two terminal hydrogens. Thiere is a simple classification of boranes according to the number of electron pairs. This is known as Wade’s rule.
Wade’s rule
Each H-B unit contributes 2 electrons to cluster bonding H-B. Each H-B contributes 2 electrons to the cluster bonding. The negative charge is then added to this to give the number of electrons pairs. The number of electrons pairs is directly related to the number of vertices, by n vertices is n+1 pairs of electrons.
Closo structures
A closo borane is one which has n+1 electron pairs available for cluster bonding. This is seen in:
Boranes usually form clusters in the right chemical environments. Due to the ability for the 2c-3e bond a number of electrons can be available for cluster bonding to occur. This does mean that the same amount of boron atoms can have a different number of hydrogen atoms due to the presence of either a 2c 3e bond with another boron or two terminal hydrogens. Thiere is a simple classification of boranes according to the number of electron pairs. This is known as Wade’s rule.
Wade’s rule
Each H-B unit contributes 2 electrons to cluster bonding H-B. Each H-B contributes 2 electrons to the cluster bonding. The negative charge is then added to this to give the number of electrons pairs. The number of electrons pairs is directly related to the number of vertices, by n vertices is n+1 pairs of electrons.
Closo structures
A closo borane is one which has n+1 electron pairs available for cluster bonding. This is seen in:
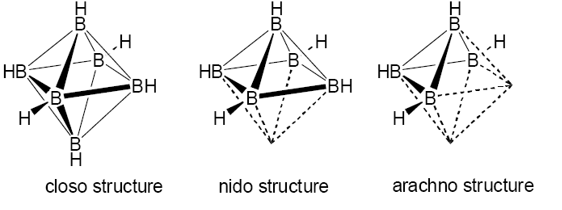
This cluster therefore has 6 vertices. This forms a closo structure. This is one with boron atoms on each vertex.
Nido structure
The nido structure is a more open structure. This can be seen by the following:
Nido structure
The nido structure is a more open structure. This can be seen by the following:
Arachno Structure.
The arachno structure is one which there are n+3 electron pairs.
The arachno structure is one which there are n+3 electron pairs.
Carboranes
These are boron groups with carbon present, there are a number of different methods that can be used when counting the total number of electron pairs. It is generally considered best to assume that the carbon hydrogen group donates 3 electrons to the group.
These are boron groups with carbon present, there are a number of different methods that can be used when counting the total number of electron pairs. It is generally considered best to assume that the carbon hydrogen group donates 3 electrons to the group.
These then follow excactly the same rules as the other structures. Almost all electron deficient structures are able to work using Wade’s rules.