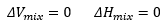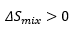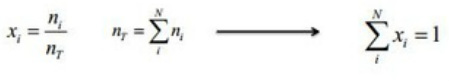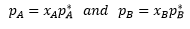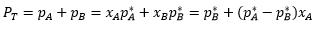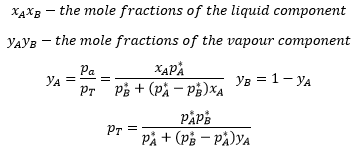Part Two: Thermodynamics of liquid mixtures
- Two component liquid mixture in equilibrium with its vapour
- Ideal mixtures (Raoult's Law)
- Pressure / Vapour pressure Composition Diagrams
- Temperature composition diagrams
- Distillation processes, in both laboratory and industry
- Questions
Binary mixtures
Two component mixtures (liquid + gas / vapour)
We want to consider a two component liquid mixture in equilibrium with its vapour.
We want to relate the composition (and pressure) of the vapour phase to the composition of the liquid phase.
This will depend on the vapour pressure of the individual liquid components (A & B) and the temperature
Ideal mixing presents the simplest case and is described by Raoult's Law.
Two component - Liquid solution
Ideal solution: A solution in which the change in enthalpy of solution is zero, this assumes that the two particles do not interact. As two particles that interacted strongly would cause a decrease in enthalpy and two particles that repelled each other would increase enthalpy.
Assumption: All molecular interactions between components are identical!
We want to consider a two component liquid mixture in equilibrium with its vapour.
We want to relate the composition (and pressure) of the vapour phase to the composition of the liquid phase.
This will depend on the vapour pressure of the individual liquid components (A & B) and the temperature
Ideal mixing presents the simplest case and is described by Raoult's Law.
Two component - Liquid solution
Ideal solution: A solution in which the change in enthalpy of solution is zero, this assumes that the two particles do not interact. As two particles that interacted strongly would cause a decrease in enthalpy and two particles that repelled each other would increase enthalpy.
Assumption: All molecular interactions between components are identical!
Upon mixing, the entropy must increase, due to the presence of two species being present.ch
Mole Fraction
The mole fraction, xi, of a mixture is the amount (i.e. number of moles) of a constituent, ni, divided by the total amount of all constituents in a mixture, nT.
The mole fraction, xi, of a mixture is the amount (i.e. number of moles) of a constituent, ni, divided by the total amount of all constituents in a mixture, nT.
Question
If we have 3.0 moles of A, 2.5 moles of B and 1.0 moles of C, what are the mole fractions of A , B and C?
If we have 3.0 moles of A, 2.5 moles of B and 1.0 moles of C, what are the mole fractions of A , B and C?
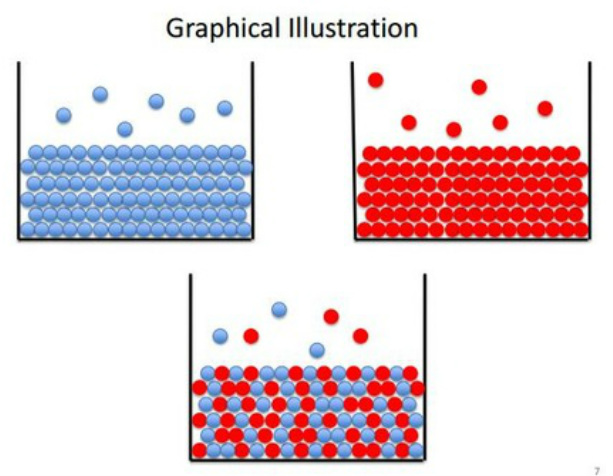
Graphical Illustration
Raoults' Law:
The vapour pressure of an ideal solution is dependent on the vapour pressure of each chemical component and the mole fraction of the components present in the solution
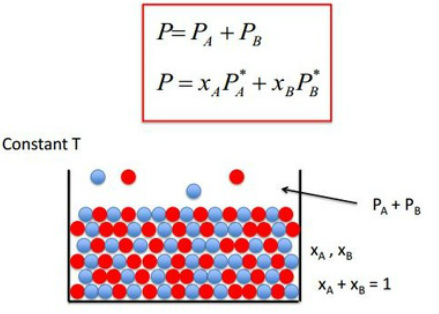
Partial pressure (Dalton's Law): In a mixture of gases, the partial pressure is the pressure of the gas would have if it occupied the volume alone.
For the mixtures of ideal liquids A and B
The vapour pressure of an ideal solution is dependent on the vapour pressure of each chemical component and the mole fraction of the components present in the solution
Partial pressure (Dalton's Law): In a mixture of gases, the partial pressure is the pressure of the gas would have if it occupied the volume alone.
For the mixtures of ideal liquids A and B
Where PA : Partial pressure of A in vapour
PA* = vapour pressure of pure liquid A
xA = mole fraction of A in liquid mixture
PA* = vapour pressure of pure liquid A
xA = mole fraction of A in liquid mixture
The two solutions mixed together forms a vapour pressure that is roughly equivalent to both of the compounds present. At higher partial pressures there is a larger concentration of that component in the gas phase. This allows concentrations of one of the solutions to be inferred from others.
Question
A solution is prepared by dissolving 1.5 moles of naphthalene (C10H8) in 1.0 kg of benzene. The vapour pressure of pure benzene is 94.6 Torr at 25'C. What is the partial pressure of benzene in the solution?
A solution is prepared by dissolving 1.5 moles of naphthalene (C10H8) in 1.0 kg of benzene. The vapour pressure of pure benzene is 94.6 Torr at 25'C. What is the partial pressure of benzene in the solution?
Raoult's Law
Examples of Ideal Liquid Mixtures
n - heptane dissolved in n - nonane
benzene dissolved in toluene
n - heptane dissolved in n - nonane
benzene dissolved in toluene
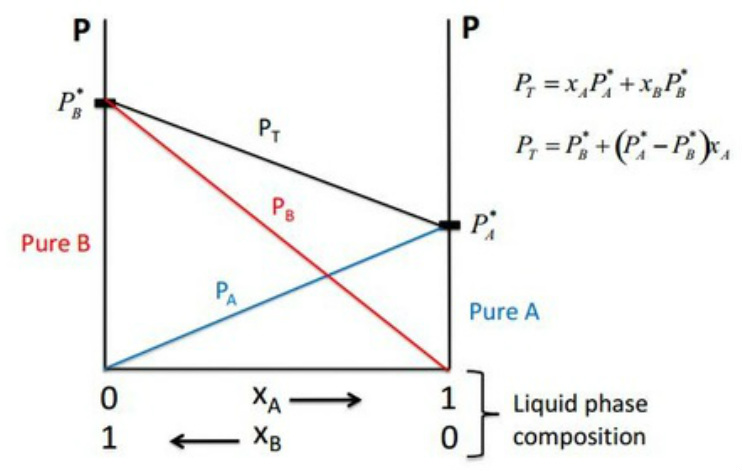
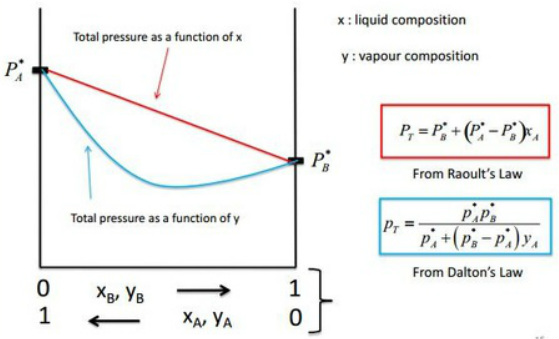
Raoult's Law: Pressure - composition diagram
- PT is the sum of each components partial pressure so should never drop below a component line.
- PX* is the maximum vapour pressure that a ideal gas can produce so there should be no pressure above this line from a single component.
- Both lines start at 0 and are straight to their partial pressure.
- PT goes from one partial pressure to the other in ideal mixtures.
Now let's consider both phases!
We want to be able to construct a diagram that fully describes both the liquid and vapour compositions over all possible ratios.
Compositions of the liquid and vapour that are in equilibrium are not generally the same - hence we need two different variables to describe the system.
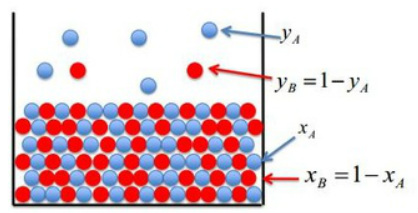
Consideration of liquid and gas phases together
xA and xB : mole fractions of the liquid component
yA and yB : mole fractions of the vapour components
The calculation of yA through the use of the pressure of each allows the concentration of yB to be found. The total pressure can also be calculated with just one gas phase concentration and its relative partial pressure.
Compositions of the liquid and vapour that are in equilibrium are not generally the same - hence we need two different variables to describe the system.
Consideration of liquid and gas phases together
xA and xB : mole fractions of the liquid component
yA and yB : mole fractions of the vapour components
The calculation of yA through the use of the pressure of each allows the concentration of yB to be found. The total pressure can also be calculated with just one gas phase concentration and its relative partial pressure.
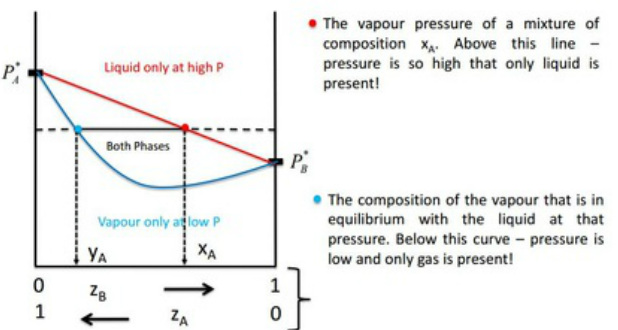
Vapour - Pressure Composition (VPC) Diagrams
Interpretation of a Vapour Pressure Composition (VPC) Diagram
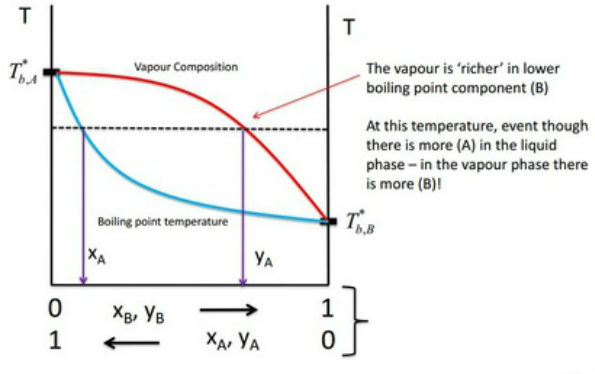
Temperature - Composition (Constant Pressure)
Distillation of mixtures
Repeated vapourisation and condensation through decreasing the temperature gradient leading to separation. This is easy to make sense of if looked at systematically. The process starts at point A. The liquid mixture is heated to point B. At point B the liquid has a vapour component mixture of point C. So, on cooling the liquid components are that now of point D. At point D the vapour component mixture is now that of point E and the cycle continues. This shows how from a solution of nearly Pure A the solution can be made to pure B.
Repeated vapourisation and condensation through decreasing the temperature gradient leading to separation. This is easy to make sense of if looked at systematically. The process starts at point A. The liquid mixture is heated to point B. At point B the liquid has a vapour component mixture of point C. So, on cooling the liquid components are that now of point D. At point D the vapour component mixture is now that of point E and the cycle continues. This shows how from a solution of nearly Pure A the solution can be made to pure B.
In Simple Distillation, the vapour is withdrawn and condensed. This technique is used to separate a volatile liquid from a non-volatile solute.
In Fractional Distillation, the boiling and condensation cycles are repeated successively. This technique is used to separate volatile liquids.
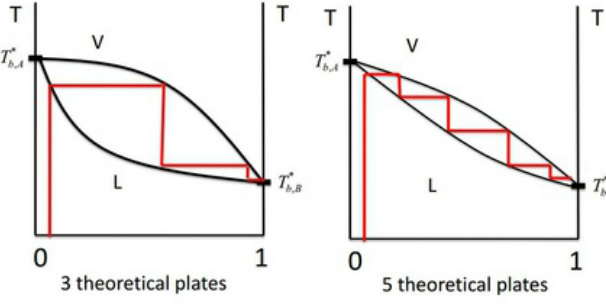
The efficiency of a fractional column is expressed in terms of the number of theoretical plates. That is, the number of vapourisation and condensation phases required to achieve a condensate of a given composition from a given distillate.
Theoretical Plates
In Fractional Distillation, the boiling and condensation cycles are repeated successively. This technique is used to separate volatile liquids.
The efficiency of a fractional column is expressed in terms of the number of theoretical plates. That is, the number of vapourisation and condensation phases required to achieve a condensate of a given composition from a given distillate.
Theoretical Plates
Mixtures whose components have more similar partial pressures will require more theoretical plates for separation.
Question
The vapour pressure of benzene is 53.3 kPa at 60.6 'C, but fell to 51.5 kPa when 19.0 g of an involatile organic compound was dissolved in 500 g of benzene. Calculate the molar mass of the compound.
The vapour pressure of benzene is 53.3 kPa at 60.6 'C, but fell to 51.5 kPa when 19.0 g of an involatile organic compound was dissolved in 500 g of benzene. Calculate the molar mass of the compound.
Summary of Important Concepts: Thermodynamics of liquid mixtures
- Raoult's Law relates the vapour pressure of a substance and its mole fraction in a solution.
P = xAPA* + XBPB*
- Pressure composition diagrams are used to describe how the total vapour pressure of a binary mixture changes with the ratio of the individual constituents.
- Vapour pressure composition diagrams are used to describe both the liquid and vapour phases.
- Temperature composition diagrams can be used to explain the distillation (i.e. the separation process) of liquid mixtures.
|
Topic One: Single Component Mixtures
Topic 2: Thermodynamics of Liquid Mixtures Topic 3: Thermodynamics of Non Ideal Mixtures Topic 4: Two and Three Component Mixtures Topic 5: Colligative Properties |