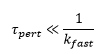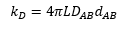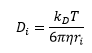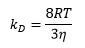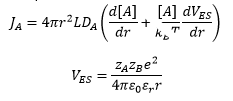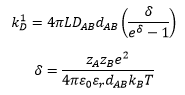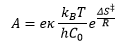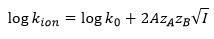Perturbation methods need to be carried out much quicker than the fastest rate of reaction being measured. This allows the concentration of the reactants to then be measured over the course of the reaction afterwards.
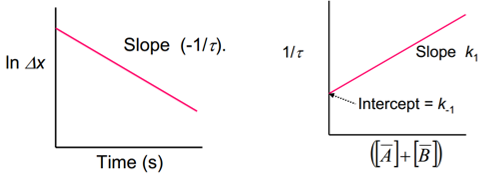
The analysis of the perturbation methods are discussed in better detail here but the final result can be plotted as a sum of the reactant concentrations against 1/ the perturbation time.
Diffusion controlled reactions
The rate of reaction in the diffusion controlled reaction is related to Avogadro's number, the diffusion constant of the two molecules and the separation distance of the two molecules. This can then be related to the viscosity and the energy.
Ionic potential has an effect on the rate of reaction also, this is where the relative charge of the ions and the separation of the ions is taken into account.
The rate constant of an ion controlled reaction can be given by:
The Arrhenius rate equation can be plotted and the activation enthalpy and the pre-exponential factor.
The effect of the ionic strength on a compound can be given as: