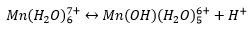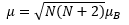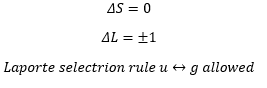A revision page of the important and easily forgotten aspects of inorganic chemistry. This will be added to over time.
Transition metal complexes· As one moves along the d-block:
· Adding to the nuclear charge. · Poor shielding of d-electrons. · Increase in the effective nuclear charge. · Contraction of the d-orbitals into the core. |
What does this effect?
Ionisation enthalpies: As the atoms get smaller the ionisation enthalpies get larger. Ionic radii: As the atoms get smaller the ionic radii gets smaller, slightly smaller radii are seen than predicted due to lack of circular symmetry due to partially filled orbitals. Hydration enthalpies or the ions: As the ions get smaller the hydration enthalpy gets higher. Variations are caused by lack of spherical symmetry. |
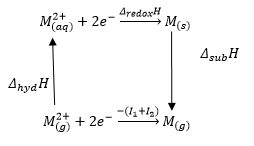
Electrode potentials
The electrode potential is the removal of electrons from an atom in the aqueous state.
The electrode potential is the removal of electrons from an atom in the aqueous state.
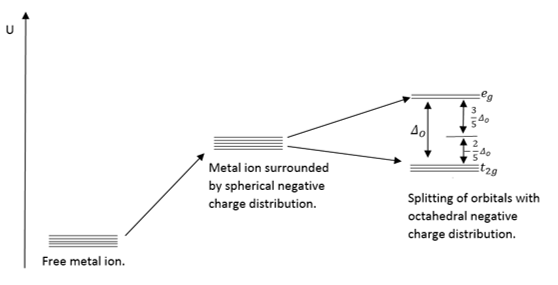
Ligand field splitting
Molecular orbital describtion of ligand field theory
|
σ donor
A σ donor is one which forms a σ bond on overlapping of orbitals. This is simply found in ligands such as chloride but also in alkene ligands were a σ bond is formed through the bonding of a π orbital in a symmetrical fashion. |
π donor
A π donation is normally small due to their being weak orbital overlap. This is where lone pairs can bond in a π overlap to un-occupied orbitals on the metal centre. |
π acceptor
A π acceptor ligand is one which accepts electron density from the metal into the ligand's π* orbitals. This forms a π bond but does reduce the electron density of the metal centre as well as the bond order of the ligand. |
Effects of ligand field splitting
|
Hydration energy
Hydration energies are related to the filling of the d orbitals. The stabilisation of the orbitals relates to the shape of the structure. As water is a weak field ligand it is more likely to be found in a low field spin state favouring octahedral symmetry. |
Electrode potentials
It can be seen that with addition of ligands there is a change in the redox potential. This can mean that for a given energy cycle there can be different enthalpy values of the metal depending on what ligands are binded to it. |
Lattice site preferences
It can be seen that the ligand field stabalisation energy favours certain geometries. These still hold true in solid lattice structures. |
Magnetism effects
The effect of the stabalisation effects on the magnatism is obvious. The different number of lone electrons that can be formed is directly related to the magnetism of the complex represented by the spin only formula.
Spin-only formula
The spin only formula is based on the total number of lone electrons N.
The effect of the stabalisation effects on the magnatism is obvious. The different number of lone electrons that can be formed is directly related to the magnetism of the complex represented by the spin only formula.
Spin-only formula
The spin only formula is based on the total number of lone electrons N.
Deviations from the spin-only formula
Deviations of the spin only formula can be cause by coupling with the electrons and different orbitals (spin orbit coupling).
High spin/ low spin crossover
On heating the boltzmann relationship means that more orbitals are in the high spin state which can therefore change the number of lone electrons causing a magnetism value to be measured.
Deviations of the spin only formula can be cause by coupling with the electrons and different orbitals (spin orbit coupling).
High spin/ low spin crossover
On heating the boltzmann relationship means that more orbitals are in the high spin state which can therefore change the number of lone electrons causing a magnetism value to be measured.
UV-Vis spectra
The selection rules that apply are:
The selection rules that apply are: