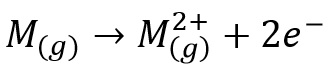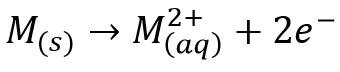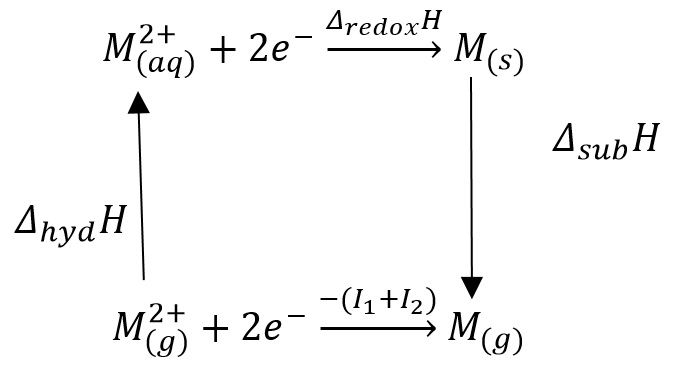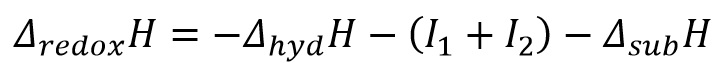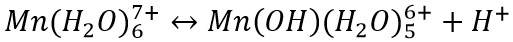Transition metal chemistry
The d-block elements have a variety of properties, and there are significant trends along the periodic table.
These compounds are important as they have a very good ability to both gain and lose electrons allowing them to act as reducing agents or oxidising agents. These compounds can also be magnetic depending on how many different electrons are present and in what state the crystal field theory explains the orbitals surrounding the metal.
The metals that are present have a large variety of properties and have a large importance in how they can fill a number of different oxidation states.
The main importance with understanding the presence of the orbitals involved in the bonding of the transition metals is the fact that shielding and effective nuclear charge make the largest noticeable differences to how a reaction occurs and how it proceeds.
The key points are:
· As one moves along the d-block:
· Adding to the nuclear charge.
· Poor shielding of d-electrons.
· Increase in the effective nuclear charge.
· Contraction of the d-orbitals into the core.
All of these key points can be represented by the group of properties that the transition metals have, this is: The variation in ionisation enthalpy, ionic radii and hydration enthalpies.
Ionisation enthalpies
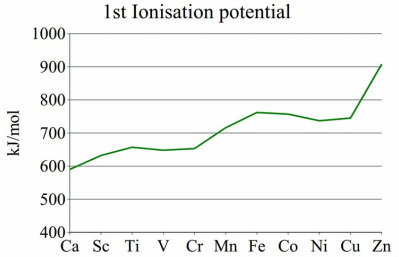
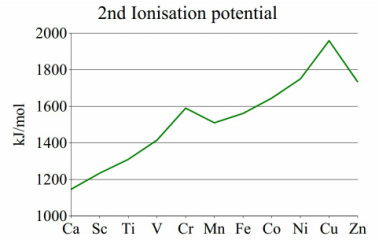
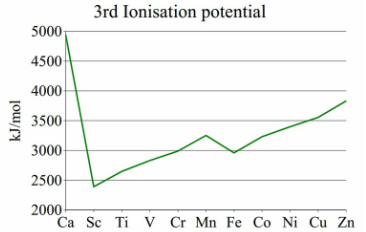
The ionisation energies vary throughout the group and although there is definite raise of enthalpy across the d-electron group there are certainly some peaks and anomalies that need to be discussed. The best way of doing this is comparing the three ionisation enthalpy graphs.
These compounds are important as they have a very good ability to both gain and lose electrons allowing them to act as reducing agents or oxidising agents. These compounds can also be magnetic depending on how many different electrons are present and in what state the crystal field theory explains the orbitals surrounding the metal.
The metals that are present have a large variety of properties and have a large importance in how they can fill a number of different oxidation states.
The main importance with understanding the presence of the orbitals involved in the bonding of the transition metals is the fact that shielding and effective nuclear charge make the largest noticeable differences to how a reaction occurs and how it proceeds.
The key points are:
· As one moves along the d-block:
· Adding to the nuclear charge.
· Poor shielding of d-electrons.
· Increase in the effective nuclear charge.
· Contraction of the d-orbitals into the core.
All of these key points can be represented by the group of properties that the transition metals have, this is: The variation in ionisation enthalpy, ionic radii and hydration enthalpies.
Ionisation enthalpies
The ionisation energies vary throughout the group and although there is definite raise of enthalpy across the d-electron group there are certainly some peaks and anomalies that need to be discussed. The best way of doing this is comparing the three ionisation enthalpy graphs.
This diagram shows that the trend isn’t perfect and there is a large amount of plateaus present. This is due to the fact that all the elements have 4s orbitals that may be filled. This means that there is a large variation in orbital energies, which in turn, leads to a variation in the ionisation enthalpies that are present.
The 2nd ionisation potential ALWAYS results in a 3dn formation. This means that the ionization potential follows a definite trend. The main example here is the peak at the chromium. This is due to their being an electron lost from a 3d5 forming a 3d4 orbital. This is not spin favourable due to an electron being taken out of a symmetrical system. This means that this is more stabalised in energy.
This shows the calcium is now in the 3p orbital so has a massive energy increase. The rest show similar properties except manganese is now moving from the 3d5 to the 3d4 configuration.
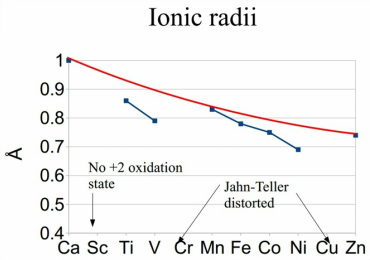
Ionic radii
The ionic radii of the +2 elements should be able to follow a distinct pattern in loss of size of the groups. This is simple as the same subshell and orbital is being filled and by that logic the effective nuclear charge should increase across the period. This does not occur and there are some significant drops in ionic radii in certain areas.
This is due to the orbtials being directional causing a decrease in the structure size. As the orbtials do not all have the same amount of electron density when placed into a chemical atmosphere some of the bonding groups can get closer bringing the radii down. This is not true for manganese which maintains a fully filled electron orbital this means there is circular symmetry present meaning they can all fill the same orbitals.
Ionic radii
The ionic radii of the +2 elements should be able to follow a distinct pattern in loss of size of the groups. This is simple as the same subshell and orbital is being filled and by that logic the effective nuclear charge should increase across the period. This does not occur and there are some significant drops in ionic radii in certain areas.
This is due to the orbtials being directional causing a decrease in the structure size. As the orbtials do not all have the same amount of electron density when placed into a chemical atmosphere some of the bonding groups can get closer bringing the radii down. This is not true for manganese which maintains a fully filled electron orbital this means there is circular symmetry present meaning they can all fill the same orbitals.
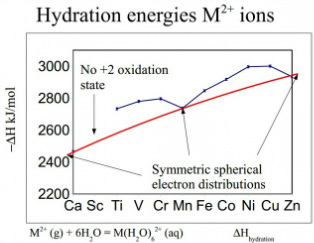
Hydration energies for M(2+) ions
The hydration energies although a very different graph see the same movements away from the centre. This is due to the there being a very obvious relation between the energy needed to remove water to the atomic radii. This means the lower energy is seen at the Maganese and Zinc due to an obvious spherical symmetry for the water to surround.
The hydration energies follow the formula below:
The hydration energies although a very different graph see the same movements away from the centre. This is due to the there being a very obvious relation between the energy needed to remove water to the atomic radii. This means the lower energy is seen at the Maganese and Zinc due to an obvious spherical symmetry for the water to surround.
The hydration energies follow the formula below:
Electrode potential
The electrode potential is a measure of the dissociation of a solid to a aqueous ion. The electrode potential increases across the group which seem to be simply explained by the change in ionisation energies that are present. This is not true however as:
Ionisation energies refer to:
The electrode potential is a measure of the dissociation of a solid to a aqueous ion. The electrode potential increases across the group which seem to be simply explained by the change in ionisation energies that are present. This is not true however as:
Ionisation energies refer to:
So this is the removal of electrons to a gas phase atom.
The electrode potentials are represented as:
The electrode potentials are represented as:
This means that there are two very different processes occurring here. The electrode potential can only be found by either direct measurement of by an energy cycle that allows calculation of the strength of the bonding between all states.
It is very important to remember how to form this energy cycle. It is needed for a large number of exam answers involving how the ionic radii effects the different enthalpies involved in a reaction.
This table shows how it is important to note that the ionisation enthalpies and the changes in enthalpy of hydration and sublimation make a large difference to the enthalpy of oxidation. This can be seen in the equation:
This table shows how it is important to note that the ionisation enthalpies and the changes in enthalpy of hydration and sublimation make a large difference to the enthalpy of oxidation. This can be seen in the equation:
Electrode potentials have a greater relation to the Ionisation enthalpy as these are inherently larger energy transitions. This means that earlier in the group it is favourable for the ion to be in the +3 state where as later in the group it is more favourable for it to be in the +2 oxidation state.
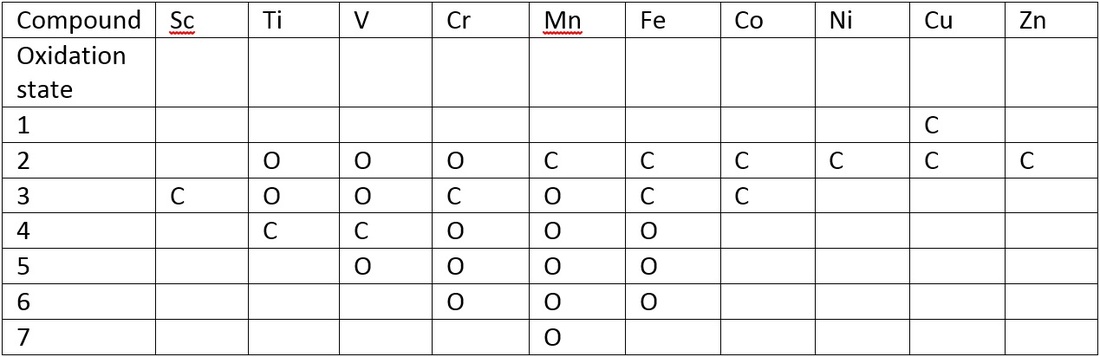
Oxidation states
The oxidation states of the transition metals follow a definite pattern moving from high oxidation to low oxidation seen below:
This is where C is the common oxidation state at O is an observed oxidation state.
Oxidation states
The oxidation states of the transition metals follow a definite pattern moving from high oxidation to low oxidation seen below:
This is where C is the common oxidation state at O is an observed oxidation state.
When these higher oxidation states are present in water there is a strongly oxidising effect. With the higher oxidising effect there is a greater chance that the transition metal will behave as an acid. Forming an acid-base equilibrium.
Descriptive chemistry
Oxides
The oxides have similar structures seen for the same stoichiometry. This is testament for the electronegativity of oxygen only being stabilised in certain formations. With a higher oxidation state of metal the oxide is more likely to act covalently rather than ionic compound. Due to this it is best to remember certain examples surrounding each stoichiometry rather than all of the different examples.
MO
Ionic type behaviour found in all transition metal forms takes NaCl structure although the Copper takes a square planar unit due to Jahn-Teller distortion.
M2O3
Oxide that maintains ionic like behaviour in a hexagonal close packed array of oxide ions the 2/3rds of the octahedral site filled by M3+. This is seen for Ti, V, Cr, Fe found in these oxidation states.
MO2
The rutile structure is seen as a six-fold coordination for the metal, three for the oxide ions. This shows a higher covalent character as the coordination of the oxygen begins to reduce. This is seen in the square planar structure. This is seen for Ti, V, Cr, Mn.
M2O5
Only seen for V, similar to P2O5, this is a molecular type structure which can be seen by the oxygen being two coordinate and the layers of atoms being held by van der waals forces. The fact that this sturcutre is similar to the highly covalent P2O5 suggests a large amount of electron sharing begins to form. This can only be seen in V.
MO3
Only seen for Cr, infinite chains of CrO4 tetrahedra, the structure is similar to SeO3. It is worth noting that Cr is at its maximum oxidation state so is a highly positive ion now. This shows why there is a large similarity between this and the covalent selenium bonding.
M2O7
Only seen for Mn, this is an oily material that is soluble in organic solvents this is at its group valent state. The fact that this material is soluble in organic solvents show that the bonds are only polar and are in no way ionic.
Chlorides
MCl2
The different chlorides can be either Cr, Mn, Fe, Co, Ni or CdI2 the layer structures are either ccp or hcp Cl- and ½ octahedral sites filled by M2+.
MCl3
This carries a BI3 structure which is based on the hcp close packing chloride ions with V3+ in the octahedral sites. Found in Ti, V, Cr, Mn, Fe, Vcl3 FeCl3.
MCl4
Only Ti, V, and Cr, have this molecular structure, the reason why there is much less similarities between chloride and oxide metals is the lack of ability for multiple bonds to form in covalent like chloride.
Oxides
The oxides have similar structures seen for the same stoichiometry. This is testament for the electronegativity of oxygen only being stabilised in certain formations. With a higher oxidation state of metal the oxide is more likely to act covalently rather than ionic compound. Due to this it is best to remember certain examples surrounding each stoichiometry rather than all of the different examples.
MO
Ionic type behaviour found in all transition metal forms takes NaCl structure although the Copper takes a square planar unit due to Jahn-Teller distortion.
M2O3
Oxide that maintains ionic like behaviour in a hexagonal close packed array of oxide ions the 2/3rds of the octahedral site filled by M3+. This is seen for Ti, V, Cr, Fe found in these oxidation states.
MO2
The rutile structure is seen as a six-fold coordination for the metal, three for the oxide ions. This shows a higher covalent character as the coordination of the oxygen begins to reduce. This is seen in the square planar structure. This is seen for Ti, V, Cr, Mn.
M2O5
Only seen for V, similar to P2O5, this is a molecular type structure which can be seen by the oxygen being two coordinate and the layers of atoms being held by van der waals forces. The fact that this sturcutre is similar to the highly covalent P2O5 suggests a large amount of electron sharing begins to form. This can only be seen in V.
MO3
Only seen for Cr, infinite chains of CrO4 tetrahedra, the structure is similar to SeO3. It is worth noting that Cr is at its maximum oxidation state so is a highly positive ion now. This shows why there is a large similarity between this and the covalent selenium bonding.
M2O7
Only seen for Mn, this is an oily material that is soluble in organic solvents this is at its group valent state. The fact that this material is soluble in organic solvents show that the bonds are only polar and are in no way ionic.
Chlorides
MCl2
The different chlorides can be either Cr, Mn, Fe, Co, Ni or CdI2 the layer structures are either ccp or hcp Cl- and ½ octahedral sites filled by M2+.
MCl3
This carries a BI3 structure which is based on the hcp close packing chloride ions with V3+ in the octahedral sites. Found in Ti, V, Cr, Mn, Fe, Vcl3 FeCl3.
MCl4
Only Ti, V, and Cr, have this molecular structure, the reason why there is much less similarities between chloride and oxide metals is the lack of ability for multiple bonds to form in covalent like chloride.