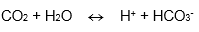Summary of electrodes
Sadly it is very common for highly specific questions of certain electrodes to come up in exams. This page should help give the main details and some key tips on explanation of the electrodes.
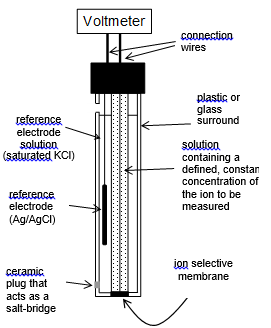
Ion selective electrodes
|
Key points
There needs to be:
It is really important to not get an amperometric sensor and ISEs mixed up. ISEs are not amperometric sensors. There are no marks for the description of one against another. This is very important. |
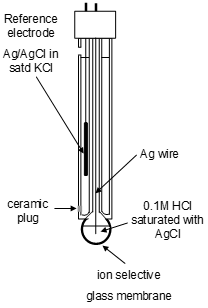
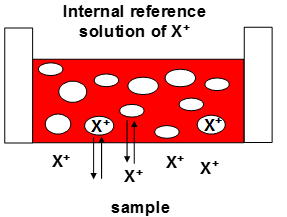
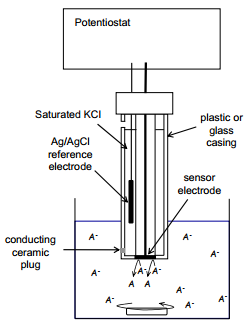
A top level non-specific ISE design is shows below. This is a good starting point with acknowledging that the standard electrode is almost always a silver solid with silver chloride salt in a saturated potassium chloride solution.
|
There are three main types of electrode:
- Glass electrodes: consiting of silica membranes measure: pH, Na+.
- Solid state electrodes: F-, Cl-.
- Polymer electrodes: K+, Ca2+.
|
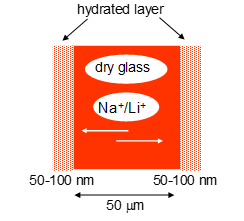
Glass electrode (pH)
Movement of ions in a silica membrane causes a potential difference to form depending on how many hydrogen ions are on either side of the membrane. Sensitive to other cations such as Na+ at high pHs. |
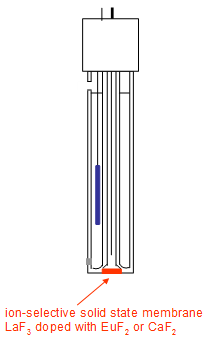
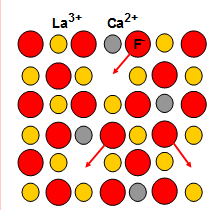
Solid State Electrode (F-)
The fluoride selective electrode measures the difference in potential caused by Fluoride ions. This has CaF2 and BaF2 doped LaF3. The movement of flouride ions through the solid surface is caused by doping agents. OH- is the only interfering ion so these electrodes cannot be used in alkaline pHs. At low pHs hydrogen ions bond to flouride causing a smaller voltage to be measured. TISAB is used. |
Polymer electrodes
Polymer electrodes are used to coordinate to metals. This is known as ionophore interaction and is selective to a particular ion. It is important to keep in mind the chemistry of these structures. It is very likely that the different ioniphores can interact with different structures.
|
There are three types of other electrodes that need to be considered. These are mentioned regularly in exam papers.
|
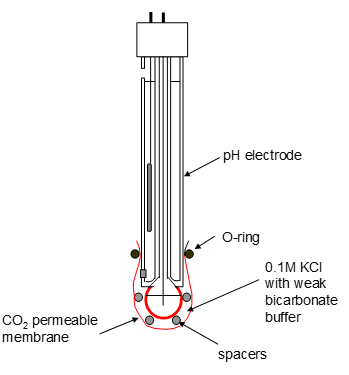
Gas sensing electrodes
An example of a gas sensing electrode is the carbon dioxide detector this uses the relationship between carbon dioxide and pH to measure concentration. |
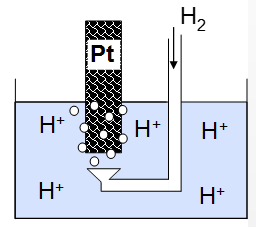
Hydrogen standard electrode
This is the electrode that all electrode potentials are based from. This is the reaction of hydrogen ions with electrons forming hydrogen gas catalysed by Platinum. This is not used as much due to the complications of hydrogen storage etc. |
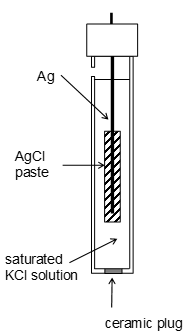
Silver chloride standard electrode
The electrode reaction is between silver ions and eleectrons. The AgCl paste provides Ag+ ions this is jept in a KCl solution. This is cheap and robust compared to the hydrogen standard. It is frequenctly attached to other analytical devices. |
Amperometric sensors
These sensors actually carry out a chemical reaction at their surface. The change in current is measured.
|
The design of amperometric sensors involve these common points:
|
|
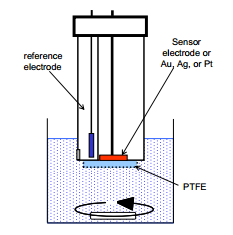
The Clarke Electrode
This is an oxygen sensor electrode that measures oxygen concentration by reducing oxygen forming water. The selectivity of this electrode is given by the presence of PTFE. |
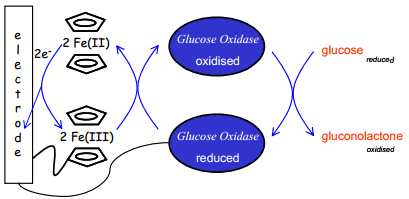
The Ferrocence Based Glucose Biosensor
Glucose can be analysed using a Ferrocene detector. This sensor uses a silver chloride reference electrode. |