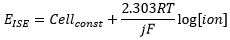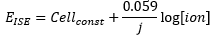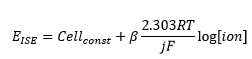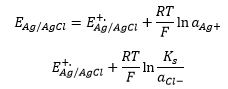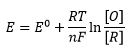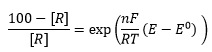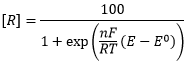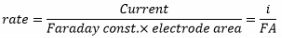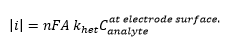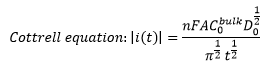This is a short summary of the equations present in the ion analysis topic.
Nernst equation
The Nernst equation is an equation that relates the electrochemical potential measured to the concentration of the ion present.
The Nernst equation is an equation that relates the electrochemical potential measured to the concentration of the ion present.
· F – Faraday’s constant 96484 C mol-1
· R – Gas constant 8.314 J K-1 mol-1.
· T – Temperature in Kelvin.
· J – Charge of ion.
· R – Gas constant 8.314 J K-1 mol-1.
· T – Temperature in Kelvin.
· J – Charge of ion.
This is the importance in the Nernst equation in finding the electrode potential of an ion in solution. The only other aspects to consider are:
- An electrode constant is present (β) but this is normal equal to one and only changes close to detection limits or with issues with the electrode itself.
Interfering ions and selectivity coefficients
Standard electrode
A standard hydrogen electrode measures the potential formed of the silver chloride interactions.
A standard hydrogen electrode measures the potential formed of the silver chloride interactions.
This is where Ks is the solubility product. This is found by the addition of the concentration of ions in the solution.
Amperometric sensors
The change in potential difference should move across the oxidation/reduction barrier causing reduced material to become oxidised or vice versa.
There eqautions are where [O] and [R] are the concentrations of oxidised and reduced material E is the electrode potential and E0 is the standard electrode potential (compared to the hydrogen electrode.
This obviously only takes into account the concentration of the ions at the electrode surface, this means that any changes in concentration caused by the movement of the analyte into or away from the electrode needs to be accounted for. This can be represented by the Cottrell equation: