Gas Chromatography is a very powerful technique but is not without its limitations. It is an important technique that is still in use today. Although not as common place as it once was GC has some important uses and is still common.
GC
Gas chromatography is a very powerful technique that is used for the separation of volatile chemical species. It involves the adsorption of species onto a solid phase from a mobile gas phase.
Gas chromatography is a very powerful technique that is used for the separation of volatile chemical species. It involves the adsorption of species onto a solid phase from a mobile gas phase.

Gas chromatography uses the presence of a inert carrier gas through a capillary or packed column. The flow of analytes through the column is measured by detectors.
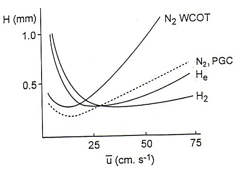
Carrier gasesThe carrier gas is important it is normally N2, He, H2 and must be of high purity. It is very important oxygen is not present in the column stream.
The flow rate for the gases are high at around 25-200 mL/ min for a packed column. This is why N2 is normally used due to the expense of the other gases. |
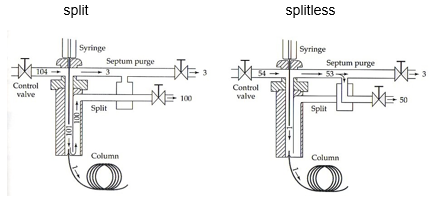
InjectorsThere are two different methods of injection systems these are direct injection and split injection systems. Split injection only utilises 0.1-1% of sample and the rest goes to column waste. This is used when there is a high concentration of analyte that is likely to overload the column.
In the event of small analyte concentrations direct injection is used. The temperature of the injector is high so that samples partition into the gas phase quickly. |
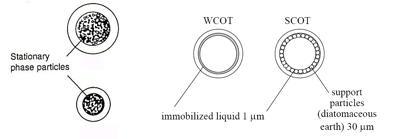
ColumnsThe columns that are present can be two different types, packed or capillary/ open tubular.
Immobilized "liquid" stationary phases are oils that have a low volatility and a high decomposition temperature. These are chemically inert so they do not react at high temperatures and are chemically attached to the support.
Stationary phases are covalently bonded groups which are bonded directly to the column. All of these have the same like attracts like properties of a HPLC column. |
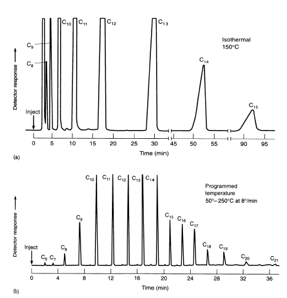
Elution by temperatureTghe temperature of the oven that the column is contained in is very important. This temperature must be kept stable throughout the analysis and at a certain minimum the analytes cannot be present in the vapour phase so therefore cannot be separated.
It is important for temperature to be high so the analytes can run fast and separation is efficent although this can be too fast and cause analytes to run too close to one another. Temperature programming can be used which works a lot like gradient elution. This increase in speed at certain temperatures can help bring species off of the column at different rates which is highly effective at controlling separation factors. |
Detectors
|
Thermal conductivity detector
A thermal conductivity detector measures the difference in thermal conductivity of different components as they travel pass a detector. This detector is a series of heated coils. It is not very sensitive but is good for detecting gases such as O2 and N2O. The use of a carrier gas with a very large thermal conductivity is important so the detector is at its maximum sensitivity. |
Flame ionisation detector.
This is a simple instrument that burns organic compounds and has a very high detector limit of around 50 ppb. The response from the detector is proportional to the number of carbons present in the compound. It is inversely proportional to the amount of oxygen in the compound such that: |
Sample preparation
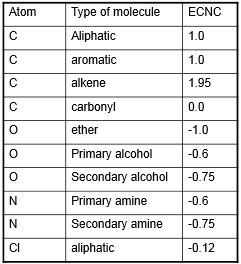
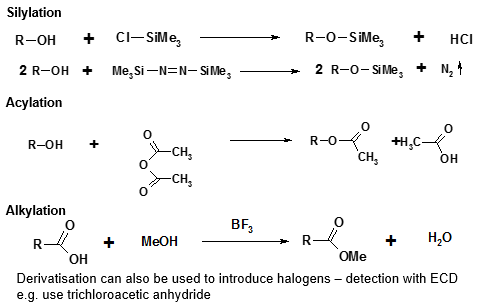
Derivitisation is an important process used in GC and in HPLC. This is the act of adding a compound that can help with turning compounds into volatile ones which can then be separated by GC. The examples of this are shown below: