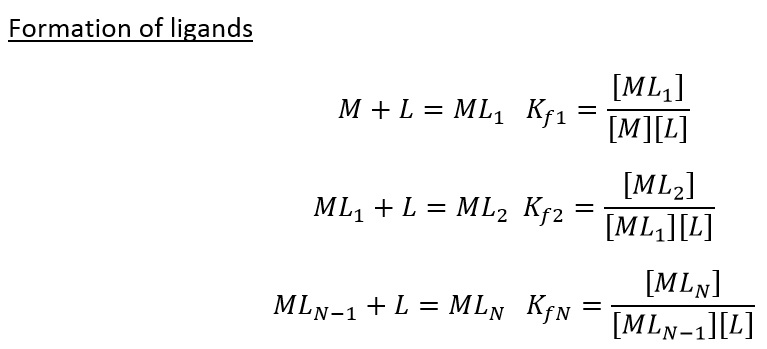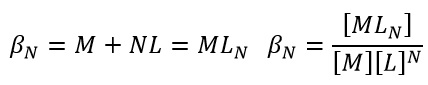Crystal field splitting
Crystal field splitting is an important concept in transition metal chemistry. Crystal field theory explains many of the complex properties that transition metal complexes show and why they hold many unique properties.
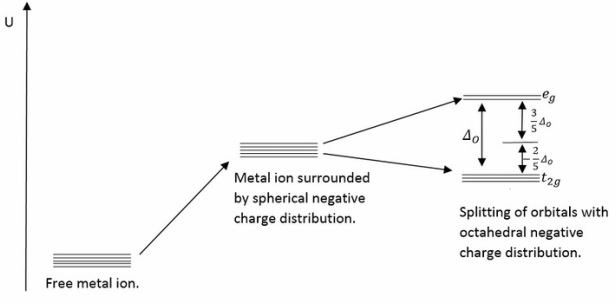
The splitting of the d orbitals is due to the negative charge of ligands increasing the overall energy of the metal atom. When the ligand charge is arranged in an octahedral fashion around the molecule it can be easily seen that the and are both excited equally to a higher energy level. It is also important to note that different arrangements can be formed due to these factors and the effects of Jahn Teller distortion. This creates tetrahedral ligand field splitting and octahedral ligand field splitting.
Crystal field splitting is an important concept in transition metal chemistry. Crystal field theory explains many of the complex properties that transition metal complexes show and why they hold many unique properties.
The splitting of the d orbitals is due to the negative charge of ligands increasing the overall energy of the metal atom. When the ligand charge is arranged in an octahedral fashion around the molecule it can be easily seen that the and are both excited equally to a higher energy level. It is also important to note that different arrangements can be formed due to these factors and the effects of Jahn Teller distortion. This creates tetrahedral ligand field splitting and octahedral ligand field splitting.
Tetrahedral splitting
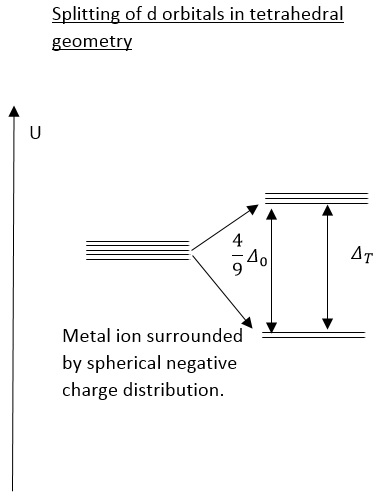
Tetrahedral splitting is 4/9 that of the octahedral splitting distance. It also has a different amount of energy levels in each area. This is an important aspect of how different geometries form in chemistry. Due to the lower splitting energy the tetrahedral complex it is normal to find these in the high spin state. This is due to the pairing energy remaining the same yet the splitting energy decreasing.
Tetrahedral splitting is 4/9 that of the octahedral splitting distance. It also has a different amount of energy levels in each area. This is an important aspect of how different geometries form in chemistry. Due to the lower splitting energy the tetrahedral complex it is normal to find these in the high spin state. This is due to the pairing energy remaining the same yet the splitting energy decreasing.
Square planar splitting
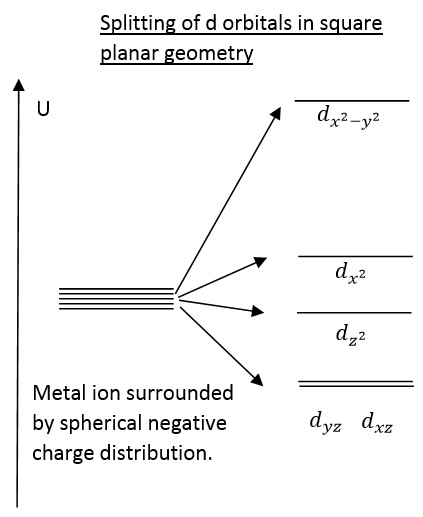
Square planar splitting can be seen as almost an extreme Jahn-Teller distortion. It can be seen that the elongation of the bonds in the z axis have caused complete removal of the ligands leaving the orbitals at completely different energy states.
Square planar splitting can be seen as almost an extreme Jahn-Teller distortion. It can be seen that the elongation of the bonds in the z axis have caused complete removal of the ligands leaving the orbitals at completely different energy states.
Classical complexes
The charge density around the metal is equal even when the oxidation number of the metal varies. This leads to the assumption that the ligands have the main effect on electron density and properties not the metal.
This leads to different types of ligands being accepted as giving properties such as:
· Bonding type: σ and π donor, σ donor only, π donor only, π acceptor.
This is based on the spectrochemical series.
Spectrochemical series
The ordering of ligands in terms of the frequency of the absorptions seen in the UV/vis spectrum. Low frequency, (long wavelength on the left). At the extreme left where the weaker ligands are found are σ and π donors. This causes a large amount of electron addition to the molecule meaning the fields are pushed closer together at the far right σ donors and π acceptors are found. These can end up withdrawing a large amount of density. This difference increases with charge allowing a very large difference in 2nd and 3rd row transition metals.
I− < Br− < S2− < SCN− < Cl− < NO3− < N3− < F− < OH− < C2O42− < H2O <NCS− < CH3CN < py < NH3 < en < 2,2'-bipyridine < phen < NO2− < PPh3 < CN− < CO
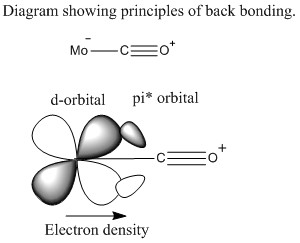
The larger halogen atoms can bond using both sigma and pi donation. This is because they are large with a large number of lone pairs. This middle compounds such as pyridine usually bond with only sigma bonds. Carbon monoxide ligands bond through sigma donation but withdraw electron density from the central atom through anti-bonding pi orbitals.
The charge density around the metal is equal even when the oxidation number of the metal varies. This leads to the assumption that the ligands have the main effect on electron density and properties not the metal.
This leads to different types of ligands being accepted as giving properties such as:
· Bonding type: σ and π donor, σ donor only, π donor only, π acceptor.
This is based on the spectrochemical series.
Spectrochemical series
The ordering of ligands in terms of the frequency of the absorptions seen in the UV/vis spectrum. Low frequency, (long wavelength on the left). At the extreme left where the weaker ligands are found are σ and π donors. This causes a large amount of electron addition to the molecule meaning the fields are pushed closer together at the far right σ donors and π acceptors are found. These can end up withdrawing a large amount of density. This difference increases with charge allowing a very large difference in 2nd and 3rd row transition metals.
I− < Br− < S2− < SCN− < Cl− < NO3− < N3− < F− < OH− < C2O42− < H2O <NCS− < CH3CN < py < NH3 < en < 2,2'-bipyridine < phen < NO2− < PPh3 < CN− < CO
The larger halogen atoms can bond using both sigma and pi donation. This is because they are large with a large number of lone pairs. This middle compounds such as pyridine usually bond with only sigma bonds. Carbon monoxide ligands bond through sigma donation but withdraw electron density from the central atom through anti-bonding pi orbitals.
High spin low spin
The difference of orbital energies is imperitive to the understanding of high spin low spin complexes. If the splitting energy is greater than the coupling energy then that movement cannot be carried out and a low spin complex is formed.
Magnitude and nature of field splitting
It has been known for a long time that the splitting of the field is mostly due to the simpliest property found which is the covalent charcter found in most of these compounds. This is based on the spectrochemical series on how powerful the ligand is to cause the splitting to occur.
Denticity
Denticity is the number of donor atoms on the ligand which bind to the central metal ion. This is the principle of momodentate and bidentate ligands.
Some compounds are ambidentate such as NO2 which either bonds from one of the Ns or the Os this leads to different linking isomers.
Polydentate ligands can both chelate and bridge. This is important to the entropy of the system. The bidentate ligands can be seen as forming a ring with the metal centre. This can lead onto geometric analysis forwhich is more favoured as strain can take a factor in the four ringed atoms.
Tridentate ligands can either form mer- or fac-isomers this depends on whether the ligands are found in the plane or found on the face to one another. This is an interesting process and can lead to different stabilities.
Quadridentate ligands can form large macrocyclic molecules and are usually seen in the tripod formation, it is possible for these to be planar and surround the entire molecule. This is found regularly in natural cases.
Hexadentate ligands are found in EDTA this is a very strong ligand due to the chelate effect.
Isomerism
Different formations of the ligands can cause different isomerism to form. It must be remembered that there can be rapid equallibrium between the compounds causing one colour to be seen of the most favourable state.
This also leaves the simple process of cis trans isomers depending if they are on the same side as one another or the opposite sides to one another.
The difference of orbital energies is imperitive to the understanding of high spin low spin complexes. If the splitting energy is greater than the coupling energy then that movement cannot be carried out and a low spin complex is formed.
Magnitude and nature of field splitting
It has been known for a long time that the splitting of the field is mostly due to the simpliest property found which is the covalent charcter found in most of these compounds. This is based on the spectrochemical series on how powerful the ligand is to cause the splitting to occur.
Denticity
Denticity is the number of donor atoms on the ligand which bind to the central metal ion. This is the principle of momodentate and bidentate ligands.
Some compounds are ambidentate such as NO2 which either bonds from one of the Ns or the Os this leads to different linking isomers.
Polydentate ligands can both chelate and bridge. This is important to the entropy of the system. The bidentate ligands can be seen as forming a ring with the metal centre. This can lead onto geometric analysis forwhich is more favoured as strain can take a factor in the four ringed atoms.
Tridentate ligands can either form mer- or fac-isomers this depends on whether the ligands are found in the plane or found on the face to one another. This is an interesting process and can lead to different stabilities.
Quadridentate ligands can form large macrocyclic molecules and are usually seen in the tripod formation, it is possible for these to be planar and surround the entire molecule. This is found regularly in natural cases.
Hexadentate ligands are found in EDTA this is a very strong ligand due to the chelate effect.
Isomerism
Different formations of the ligands can cause different isomerism to form. It must be remembered that there can be rapid equallibrium between the compounds causing one colour to be seen of the most favourable state.
This also leaves the simple process of cis trans isomers depending if they are on the same side as one another or the opposite sides to one another.
This shows how with a higher K value the addition of a ligand is preferred to the current state. It is usually seen that initially the rates of formation are high but as the metal centre becomes more saturated the K value drops below 1. This can also be summerised as the overall formation force constant:
This is the basis of crystal field theory. Move onto ligand field theory for a more in depth discussion of Ligand field theory.