Pericyclic reactions
A pericyclic reaction is a concerted (meaning that the bond breaking and making occurs at the same time) reaction that takes place as a cyclic rearrangement of electron density. These reactions occur as a constant electron flow, meaning that there is no intermediate formed. The transition state formed is of a higher energy than the reactants. This forms a smooth curve so there is no point in which a lower energy intermediate forms. It is also important to note that there are no radical or ionic species formed in these reactions.
It is important to remember that there are three different types of pericyclic reaction: Cycloaddition, sigmatropic and electrocyclic reactions. These are three distinct reactions and they should not be used interchangeably.
Cycloaddition
A cycloaddition reaction is a reaction between a di-unsaturated group with a unsaturated group forming a cyclic system. This shows that cycloaddition forms two σ bonds from 3 π bonds.
The Diels Alder reaction
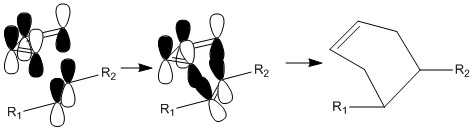
The Diels-Alder reaction is the most used cycloaddition reaction. It is one of the most simple ways to create a cyclic structure. This reaction is thermodynamically favoured as there is the formation of two σ bonds which are lower energy than the π bonds that were present. Notice the intermediate is aromatic and is therefore reasonably well stabilized.
A cycloaddition reaction is a reaction between a di-unsaturated group with a unsaturated group forming a cyclic system. This shows that cycloaddition forms two σ bonds from 3 π bonds.
The Diels Alder reaction
The Diels-Alder reaction is the most used cycloaddition reaction. It is one of the most simple ways to create a cyclic structure. This reaction is thermodynamically favoured as there is the formation of two σ bonds which are lower energy than the π bonds that were present. Notice the intermediate is aromatic and is therefore reasonably well stabilized.
Orientation of reactants
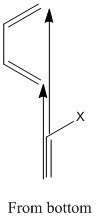
The reaction does not occur in a two dimensional plane. These is a great importance in the fact that the reactants need to approach from the top and bottom of one another. This means that the π bonds that are present can interact in the correct way to form σ bonds. This is possible from the top-bottom angle. This can be easily seen below as the π bonds can interact in a parallel fashion forming σ bonds.
The reaction does not occur in a two dimensional plane. These is a great importance in the fact that the reactants need to approach from the top and bottom of one another. This means that the π bonds that are present can interact in the correct way to form σ bonds. This is possible from the top-bottom angle. This can be easily seen below as the π bonds can interact in a parallel fashion forming σ bonds.
It is important to remeber that the Diels-Alder reaction can be carried out with hetero atoms. This makes this reaction highly useful when coupling reagents or producing heterocyclic ring systems.
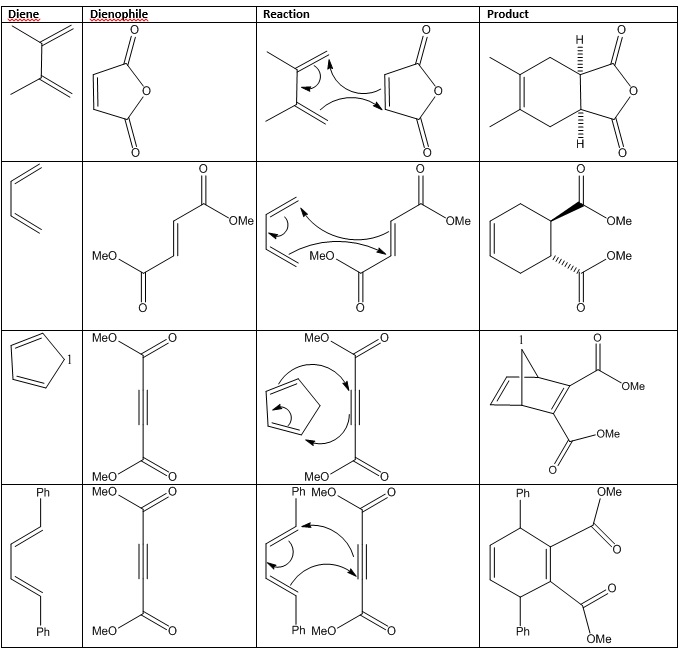
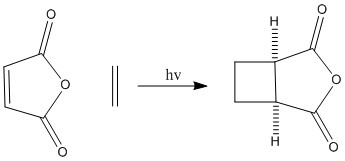
Examples of cycloaddition reactions, the mechanisms and their products are shown below.
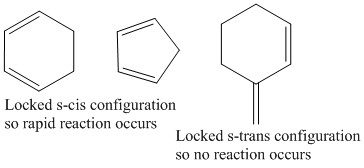
These reactions show how the stereochemistry varies during a reaction and also how the atoms can reconfigure themselves during their reaction. It also gives the names for the reactants: The diene and dienophile are easy to find during one of these reactions. It is though important to be able to recognise how effective a diene will be at reacting with a dienophile depending on what arrangement it is in. Cis or Trans.
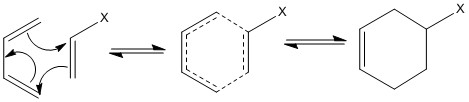
The reaction can only occur when the diene is in the correct cis position. No reaction can occur when the molecule is in the trans position. This causes locked configuration molecules to either react rapidly or not react at all.
This can be explained by looking at the bonding orbitals during the reaction as to why a certain structure needs to be present for the reaction to occur.
This can be explained by looking at the bonding orbitals during the reaction as to why a certain structure needs to be present for the reaction to occur.
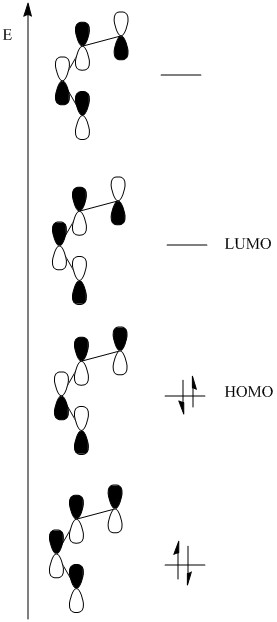
It is seen that reactions only occur when there are a number of electrons that follow Hückel’s law. This can be explained by the bonding orbitals that are formed with the π electrons that are present during the reaction.
Shown here are the comparable energies present in a conjugated diene. These show that the highest occupied molecular orbital has one orbital configuration and the LUMO has another.
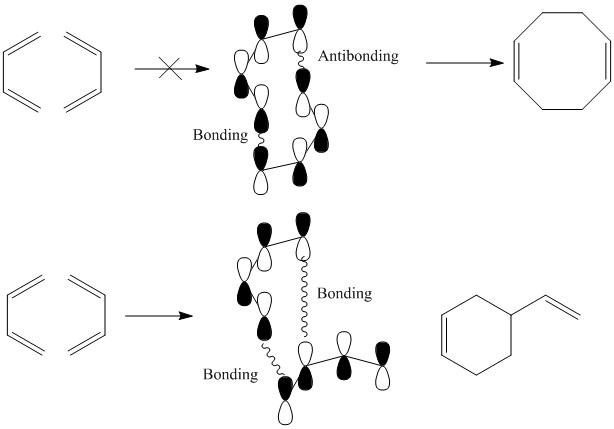
This is important when an alkene is brought into the molecules presence causing the formation of the σ bonds. This formation cannot occur if the orbitals of the alkene and the diene are out of phase with one another. If one antibonding orbital is present then the reaction does not occur.
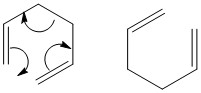
This is seen to good effect when two molecules of butadiene are reacted with one another. There is one product in this reaction.
Shown here are the comparable energies present in a conjugated diene. These show that the highest occupied molecular orbital has one orbital configuration and the LUMO has another.
This is important when an alkene is brought into the molecules presence causing the formation of the σ bonds. This formation cannot occur if the orbitals of the alkene and the diene are out of phase with one another. If one antibonding orbital is present then the reaction does not occur.
This is seen to good effect when two molecules of butadiene are reacted with one another. There is one product in this reaction.
One of the easiest ways this can be overcome is the use of photochemical excitation to change the HOMO and LUMO orbitals.
Sigmatropic rearrangement
In sigma tropic rearrangement no σ bonds are formed overall. There is a loss of a σ bond followed by a movement of two π bonds which then form another σ bond. This does not form any ionic or radical intermediates and bonds are made and broken simultaneously.
[3,3]- Sigmatropic Rearrangements
A [3,3] rearrangement occurs through a six membered cyclic transition state this means that the reactions occur with three points on a cyclic structure available for bonding. The numbering occurs from the σ bond that is being broken.
In all Sigmatropic rearrangements it is important to look at any possible stabilisation factors that may drive the reaction, such as the favored formation of a C=O bond over a C-O bond. The substitution of alkenes can also help with this.
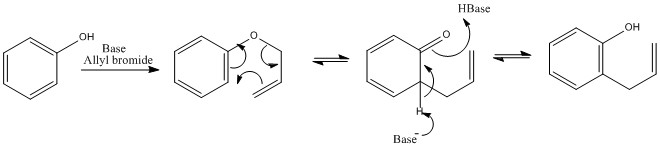
Clasien rearrangement
Clasien rearrangemet is the rearrangement that takes place on a oxygen group this can be aromatic or aliphatic. This utilises the benzene ring to form a more stable. The driving force behind this reaction is the formation of the more stable C=O bond than a C=C bond being present in the aliphatic systems.
In sigma tropic rearrangement no σ bonds are formed overall. There is a loss of a σ bond followed by a movement of two π bonds which then form another σ bond. This does not form any ionic or radical intermediates and bonds are made and broken simultaneously.
[3,3]- Sigmatropic Rearrangements
A [3,3] rearrangement occurs through a six membered cyclic transition state this means that the reactions occur with three points on a cyclic structure available for bonding. The numbering occurs from the σ bond that is being broken.
In all Sigmatropic rearrangements it is important to look at any possible stabilisation factors that may drive the reaction, such as the favored formation of a C=O bond over a C-O bond. The substitution of alkenes can also help with this.
Clasien rearrangement
Clasien rearrangemet is the rearrangement that takes place on a oxygen group this can be aromatic or aliphatic. This utilises the benzene ring to form a more stable. The driving force behind this reaction is the formation of the more stable C=O bond than a C=C bond being present in the aliphatic systems.
The reactions that occur proceed via a chair like transition state, a chair being the most stable conformation for the six-membered ring. This means that the reaction can only form an E isomer.
This also follows the favourable rules with the conformation of cyclohexane. If any substituents can be placed into the equatorial position it is highly likely that they will be.
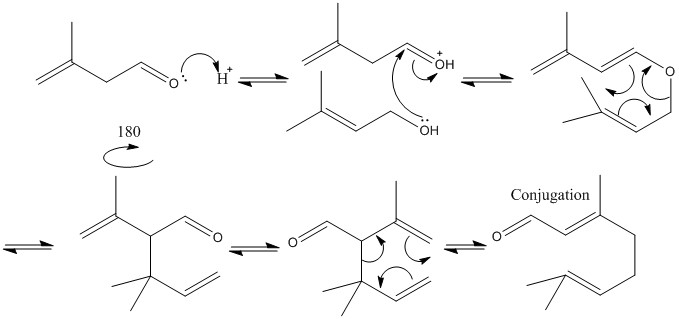
Cope rearrangement
Cope rearrangement is the same as the Clasien rearrangement but without the presence of a oxygen group. This means that the reaction is usually stabalised by the addition of carbon groups to allow one reactant to form over another.
This also follows the favourable rules with the conformation of cyclohexane. If any substituents can be placed into the equatorial position it is highly likely that they will be.
Cope rearrangement
Cope rearrangement is the same as the Clasien rearrangement but without the presence of a oxygen group. This means that the reaction is usually stabalised by the addition of carbon groups to allow one reactant to form over another.
An example of the two rearrangements being used in the formation of a single product is shown below:
Other sigmatropic rearrangements
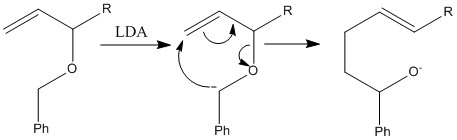
[2,3]- Sigmatropic rearrangement
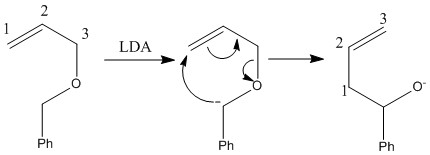
A [2,3] rearrangement proceeds via a 5 membered transition state. This can only occur when there is a negative charge present that can therefore allow the movement of 6 electrons to still form an aromatic state. This can occur by the reaction of a base with a compound removing an acidic compound worming a negative charge. The reaction can then occur maintaining the negative charge. The favourable reaction is the one that allows the negative charge to be placed on the more stable molecule.
[2,3]- Sigmatropic rearrangement
A [2,3] rearrangement proceeds via a 5 membered transition state. This can only occur when there is a negative charge present that can therefore allow the movement of 6 electrons to still form an aromatic state. This can occur by the reaction of a base with a compound removing an acidic compound worming a negative charge. The reaction can then occur maintaining the negative charge. The favourable reaction is the one that allows the negative charge to be placed on the more stable molecule.
The interconversion between an allyic ether into a homoallylic alcohol is known as the [2,3] Wittig rearrangement.
Introduction of an extra R substituent results in the formation of the E product as part of the five membered cyclic transition state. This is explained through the fact that the phenyl group and the R group will move into equatorial positions and will therefore try and be as far away from one another forming an E isomer.
Introduction of an extra R substituent results in the formation of the E product as part of the five membered cyclic transition state. This is explained through the fact that the phenyl group and the R group will move into equatorial positions and will therefore try and be as far away from one another forming an E isomer.
This only leaves electrocyclic reactions:
Electrocyclic reactions
A ring is always formed (or broken), but there is only one σ bond that is lost or broken. This also only occurs across the ends of a conjugated π system.
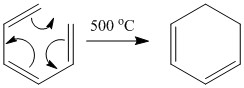
For the hexatriene molecule there is the ability for the formation of a σ bond through the loss of a π bond. This is also seen for the cyclobutene molecule where a σ bond is lost and a π bond formed as the angle strain allows this to be more stable.
Electrocyclic reactions
A ring is always formed (or broken), but there is only one σ bond that is lost or broken. This also only occurs across the ends of a conjugated π system.
For the hexatriene molecule there is the ability for the formation of a σ bond through the loss of a π bond. This is also seen for the cyclobutene molecule where a σ bond is lost and a π bond formed as the angle strain allows this to be more stable.
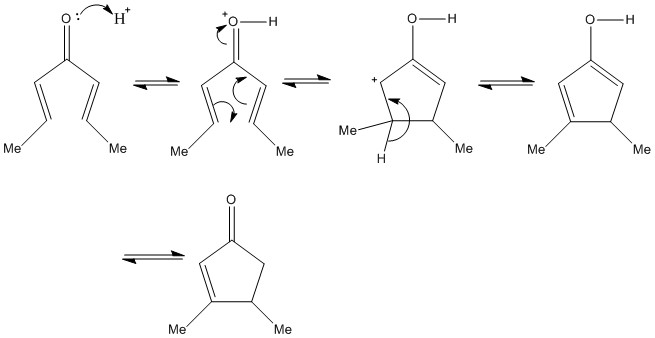
Nazarov cyclisation
This is a cyclisation reaction that involves a ring closure of an unsaturated ketone under acidic conditions. It is a highly effective reaction that involves the formation of a σ bond and then further loss of proton and then a tautomerisation step.
This produces a final cyclopentanone product.
This is a cyclisation reaction that involves a ring closure of an unsaturated ketone under acidic conditions. It is a highly effective reaction that involves the formation of a σ bond and then further loss of proton and then a tautomerisation step.
This produces a final cyclopentanone product.
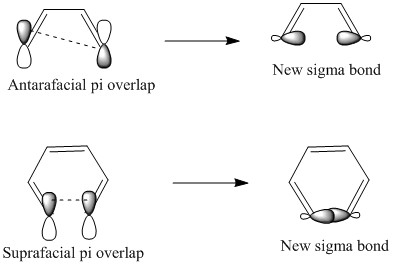
This involves something called antarafacial overlap. This is where there is a harder formation of the reaction due to the σ bond having to form across the plane of the molecule. This is hard to explain but easy to show: