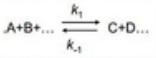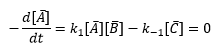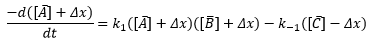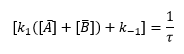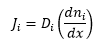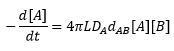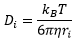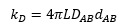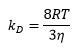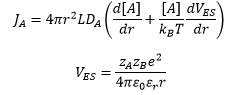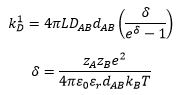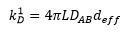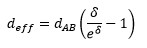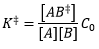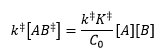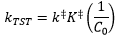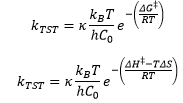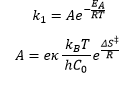|
| ||||||||||||
Theories of Chemical Reactions
Welcome to the Theories of Chemical Reactions revision page. This area is split into five different categories:
- Relaxation Kinetics: Routes to the determination of the rate constants for fast reactions (in equilibrium)
- Methods of measuring fast relaxation kinetics: Flask photolysis etc...
- Diffusion: Mode of motion in liquids and its role in controlling liquid phase reaction rates
- Transition State Theory: Understanding reactions at the microscopic level
- Factors influencing reactivity: The effect of reactant charge, solvent polarity etc...
What do you need to know already?
You need to know:
- The meanings of rate and rate constant, and how to write down a rate equation.
- How to use measured rates to determine the rate constant, and how by measuring rate as a function of concentration you can determine the reaction order.
- About temperature dependence and the Arrhenius equation k = A exp (-Ea / RT)
- Complex multistep reactions and its solvation using steady state approximation SSA.
- How to break the composite rate constant into its elementary contributions.
You need to know:
- The meanings of rate and rate constant, and how to write down a rate equation.
- How to use measured rates to determine the rate constant, and how by measuring rate as a function of concentration you can determine the reaction order.
- About temperature dependence and the Arrhenius equation k = A exp (-Ea / RT)
- Complex multistep reactions and its solvation using steady state approximation SSA.
- How to break the composite rate constant into its elementary contributions.
Why do we need to learn about Theories of Chemical Reactions?
To get a degree?
Well yes, but rate constants vary enormously! We need to understand why the Arrhenius equation works, why reactions in solutions are different to gases, and how we can measure fast reactions and observe intermediates.
To get a degree?
Well yes, but rate constants vary enormously! We need to understand why the Arrhenius equation works, why reactions in solutions are different to gases, and how we can measure fast reactions and observe intermediates.
Relaxation Kinetics
Introduction
Relaxation Kinetics is a general method for determining the elementary rate constants for reactants and products in equilibrium
In general all concentrations are time dependent until equilibrium is established. Therefore we can not use the SSA! DAHUMMMMMM!
There are many experimental methods used to study this kind of reaction, but most share two common features.
Feature 1
The method must impose a definite time zero when the reaction is said to start. This requires an instantaneous perturbation to the concentrations. Instantaneous means much faster than the shortest kinetic relaxation time.
There are many experimental methods used to study this kind of reaction, but most share two common features.
Feature 1
The method must impose a definite time zero when the reaction is said to start. This requires an instantaneous perturbation to the concentrations. Instantaneous means much faster than the shortest kinetic relaxation time.
Laser methods allow the perturbation time shorter than 10^-14 seconds!
Some examples of perturbation methods are:
Feature 2
Observation of the time dependent concentration as a function of time after initial perturbations. Usually some signal proportional to concentration is measured.
The general scheme for relaxation kinetics can be seen through the following cycle.
Equilibrium à Perturbation à Relaxation Equilibrium
Note the final equilibrium can differ from the starting one
Some examples of perturbation methods are:
- Intense Light: Generation of reactive species instantaneously by an intense light pulse. Fastest method, but rather aggressive.
- Temperature: Absorption of infra red light pulse or electrical discharge of a sample. Equilibrium constant is dependent on temperature therefore concentrations need to relax to accommodate new temperature. Rather mild.
- Pressure: Equilibrium constant is also dependent on pressure, provided reaction has a significant change in volume. Basically set off an explosion next to sample. Useful for gas reactions, but slow and complex data is returned.
- Concentration: Mildest method. Useful for enzyme kinetics. Reactants are mixed at t = 0.
Feature 2
Observation of the time dependent concentration as a function of time after initial perturbations. Usually some signal proportional to concentration is measured.
- Absorbance: Most widely used. Beer-Lambert law. Detectors are fast and sensitive.
- Fluorescence: Useful and sensitive, but restricted to fluorescent molecules only.
- Conductivity: Useful for ionic reactions
The general scheme for relaxation kinetics can be seen through the following cycle.
Equilibrium à Perturbation à Relaxation Equilibrium
Note the final equilibrium can differ from the starting one
Analysis of Reaction Kinetics
The methods described above have found particular application in the study of the rate coefficients for equilibrium. This is important for reactions which includes complex formation such as:
Each produces slightly different kinetics but are analysed through identical methods.
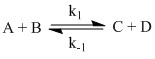
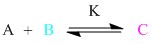
Consider the following bimolecular association reaction:
- Enzyme - Substrate reactions
- Proton transfer reactions
- Isomerisation reactions
Each produces slightly different kinetics but are analysed through identical methods.
Consider the following bimolecular association reaction:

In this case the formation of one molecule of C must mean the destruction of one molecule of A and one molecule of B. The sketch of the time dependent concentrations, [A] [B] and [C] are shown and a few terms are defined.
Similar terms apply for the other species B and C. Defining concentrations as departure from equilibrium turns out to make the analysis a lot easier.
Similar terms apply for the other species B and C. Defining concentrations as departure from equilibrium turns out to make the analysis a lot easier.
From here an expansion of terms can be produced. The important terms are:
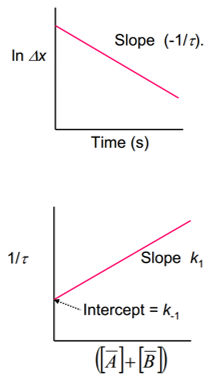
These can be used to plot a graph finding the different rate constants of the reaction:
Diffusion controlled reactions
There is great importance on whether a reaction occuring is diffusion or kinetically controlled.
|
Fick's law of diffusion
The general expression for the flux of molecules is given by Fick's law. By taking into account the movement of two molecules A and B, the diffusion constant DA and the separation of the molecules to react dAB then the following equation is given:
|
The diffusion constant can be calculated by the viscosity of the solution and the radius of the particle:
From the rate equation it can be seen that the rate constant is equal to the constants in front of the concentrations but is also calculable through:
|
Diffusion controlled reaction between ions
|
The presence of ions in the solution causes an increase in interactions between the reactants and the reactants with the solvent.
We assume full diffusion control where [A]=0 and r=dAB.
|
This shows that this is simply the original equation multiplied by an electric constant.
This holds true for most ionic reactions apart from water as this is able to untilise the Grotthus mechanism.
|
Activation controlled reactions
|
The presence of an activation barrier affects the rate of reaction. Transition state theory is a way to predict how a reaction occurs. This is the movement of a reaction through a short lived "transition state". This is based off of a number of assumptions.
Assumption 1: The reactants are in equilibrium with the activated complex: Assumption 2: The activated complex undergoes unimolecular decay with rate constant k(++). C0 is the standard concentration (1M).
|
This leaves the transition state rate constant as:
The use of conventional thermodynamics shows how there is a variation in the Gibbs free energy of the reactants and the activated complex. This means that depending on how the reaction occurs there will be a difference in the entropy of the activated state.
|
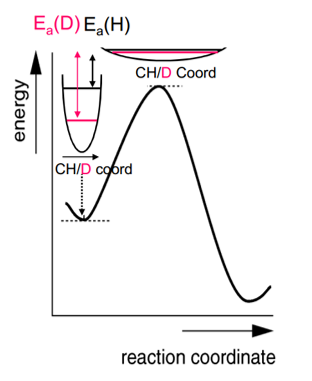
Kinetic isotope effect
The kinetic isotope effect is the ability for the transition state to be predicted by using an isotope in the reaction. The use of an isotope slightly changes the energy levels of each state which in turn causes a difference in entropy and enthalpy. This difference can be measured.