Welcome to Thermodynamics I!
Here you can study the first, second and third laws of thermodynamics. If you don't know what entropy is then you have no clue what you're in for!
This page contains revision videos, useful documents and a written transcript of what you need to know!
Feel I have done something awesome, not-so awesome, or absolutely useless (or even worse - wrong!)? Let me know at the feedback point at the relevant section so that i can investigate it. All feedback is greatly appreciated!
Enjoy Thermodynamics as much as we do :)!
Here you can study the first, second and third laws of thermodynamics. If you don't know what entropy is then you have no clue what you're in for!
This page contains revision videos, useful documents and a written transcript of what you need to know!
Feel I have done something awesome, not-so awesome, or absolutely useless (or even worse - wrong!)? Let me know at the feedback point at the relevant section so that i can investigate it. All feedback is greatly appreciated!
Enjoy Thermodynamics as much as we do :)!
Basic Entropy Revision
|
The Awesome Song of Dr. Ashworth!Skip to 2:00 to listen to the song, or if you have already lost the will to live; try to survive the whole 4:21!
|
Learn These! NOW!
The Second Law of Thermodynamics:
"The entropy of an isolated system increases in the course of a spontaneous change."
The Third Law of Thermodynamics:
"The entropies of all perfect crystals are the same at the absolute zero of temperature."
Documents you may find useful!
A mini-quiz i thought i decided to create! Play full screen and just progress through slides. You'll get how it works :)
| ||||||||
Thermodynamics: The Second and Third Laws
What you need to know:
Why does one reaction occur and another doesn't?
Why does one reaction occur and another doesn't?
- Entropy and Free Energy
- Absolute Values from the Third Law
- Predict effects of changes of T and P on processes
- Thermodynamic foundations of equilibrium
Lecture One
|
Lecture 1
Entropy is a driving force of a reaction. First Law of Thermodynamics: "The total energy of the universe remains the same." Therefore we need to know more than the conservation of energy to explain why some reactions happen. Spontaneous Change is a change that tends to occur without needing to be driven by an external influence. For an example: 2H2 + O2 --> 2H2O or Diamond --> Graphite Or how about connecting an empty flask with one full of gas, the gas dissociating is a spontaneous process. It is 'near' impossible that all the gas particles will end in the flask it began in. Whats the pattern then? Well for a block of metal cooling vibrational energy spreads out. For gas diffusing or expanding the gas molecules are spreading out. The reverse is VERY unlikely. Entropy and Disorder Low entropy = little disorder High entropy = lots of disorder The Second Law of Thermodynamics can be expressed as "the natural progression of the universe is from order to disorder." Definition of Entropy Entropy is given the sign S Given that the temperature is constant, the entropy change is given by: △S = (qrev / T) △S: Change in entropy (J/K); qrev: Quantity of reversible heat (J); T: Temperature (K) |
Try one yourself...
A large flask of water is placed on a heater and 100J of energy is transferred reversibly to the water at 25'C. What is the change in entropy of the water? Answer △S = (qrev / T) = (100 J / 298 K) = + 0.336 J/K |
|
Entropy is a state function!
Also what if the temperature is not constant, or the volume changes with entropy? Well we have to deal with change in temperature on heating by using heat capacity Cp and CV at constant pressure and volume respectively. These are values which are constants for each substance or scenario. Entropy change on heating
You do not need to necessarily know the derivation but... 1. dS = dqrev/T 2. dqrev = CdT Substitute 2 into 1 3. dS = CdT / T Integrate 3 between two temperatures 4. △S = ∫ CdT / T (between T2 and T1) Assume C constant with T so it can come out the integral 5. △S = C ∫dT / T (between T2 and T1) = Cln(T2/T1) △S = Cln(T2/T1) |
Try one yourself...
A sample of N2 gas of volume 20.0L at 5.00 kPa is heated from 20.'C to 400.'C at constant volume. What is the change in entropy of the nitrogen? CV,m(N2) = 20.81 J/K/mol Answer T1 = 293 K T2 = 673 K use PV=nRT equation n = (5.00 kPa x 20.0 L) / (8.31447 LkPa/K/mol x 293 K) n = (5.00 x 10^3 Pa x 20.0 x 10^-3 m^3) / (8.31447 LkPa/K/mol x 293 K) △S = (5.00 x 20.0 / (8.31447 x 293)) mol x 20.81 J/K/mol x ln (673 / 293) △S = + 0.710 J/K |
Key Points
MOST IMPORTANT!
The Second Law of Thermodynamics
The entropy of an isolated system increases in the course of a spontaneous change.
- A process is spontaneous if it requires no external intervention
- Entropy is a measure of disorder
- Entropy is a state function
MOST IMPORTANT!
The Second Law of Thermodynamics
The entropy of an isolated system increases in the course of a spontaneous change.
Lecture Two
|
Lecture 2
Entropy changes with volume Why is there entropy change with volume you ask? Well a greater volume means more positional disorder. Remember the equation for isothermal expansion? No you say? Well here is a reminder... wrev = -nRTln(V2/V1) Well for an ideal gas △U = 0 and therefore qrev = -wrev . Therefore we can rearrange this equation to give the following expression. △S = qrev / T △S = nRln(V2/V1) Entropy changes with pressure Well long story short for this one, just use the derivation for volume and use Boyle's law to substitute pressure from temperature. △S = nRln(V2/V1) Boyle's Law At constant temperature... (V2/V1) = (P1/P2) So another big red equation coming now... △S = nRln(P1/P2) Notice that the order you place pressure in the natural log expression is inverse to how you would for volume or temperature! Make sure you get it the right way around! |
Try one yourself...
What is the change in entropy of the gas when 1.00 mol N2 (g) expands isothermally from 22.0 L to 44.0 L? Answer △S = nRln(V2/V1) △S = 1.00 mol x 8.31447 J/K/mol x ln ( 44.0 / 22.0 ) △S = + 5.76 J/K Try one yourself...
Calculate the change in entropy when the pressure of 0.321 mol O2 (g) is increased from 0.300 atm to 12.00 atm at constant temperature. Answer △S = nRln(P1/P2) △S = 0.321 mol x 8.31447 J/K/mol x ln (0.300 / 12.00) △S = - 9.85 J/K |
|
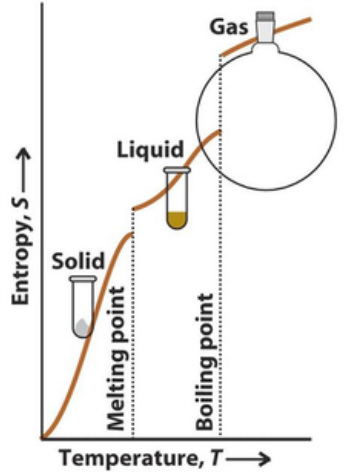
Entropy changes with state
Now imagine entropy change on vaporisation (going from liquid to gas). What do we expect and how the **** do we calculate it? Well! We benefit from the use of a phase transition. Why?
And now for Ashworth's "One of six things you need to accept before breakfast" sarcastic approach for what we need to understand! Although we already have created an equation for constant temperature right...? △S = (qrev / T) Well we can take it a step further and introduce it in a phase transition sense... △Svap = △Hvap / Tvap |
Try one yourself...
What is the entropy of vaporisation of acetone at its normal boiling point of 56.2 'C? △Hvap = 29.1 kJ/mol Answer △Svap = △Hvap / Tvap △Svap = 2.91 x 10^4 J/mol / 329.4 K △Svap = + 88.4 J/K/mol |
|
Trouton's Rule
Did you know that the increase in disorder is the same for most liquids? Well now you know! On top of this, i suppose some guy called Trouton actually managed to quantify this? This is classed as troutons rule... △Svap ≈ 85 J/K/mol Liquids with high ordering, such as water, have vaporisation entropies > 85 J/K/mol |
Try one yourself...
Use Trouton's rule to estimate the enthalpy of vaporisation of liquid bromine which boils at 59.0 'C. Answer △Svap = △Hvap / Tvap Troutons rule: △Svap ≈ 85 J/K/mol Therefore by rearranging the top equation to make enthalpy of vaporisation the subject (thats what the question is asking for) △Hvap = △Svap x Tvap △Hvap = 85 J/K/mol x 332 K △Hvap = + 28 kJ/mol (The literature value is 29.45 kJ/mol. Nice one Trouton! *High five*) |
Entropy changes at other temperatures
Fusion is when a substance goes from solid to liquid (melting). Again, same as vaporisation, we expect an increase in entropy but smaller than that during vaporisation. Therefore use the same equation but change vap to fus. Whatever you do do not use troutons rule for fusion *face palm* (its only for vaporisation).
Other times you may have to use your initiative is to break calculations down into steps.
Say we wanted to know △Svap for a substance at 25'C ? Well we are awesome and can work it out! Assume substance boils at 100'C.
Key Points
Fusion is when a substance goes from solid to liquid (melting). Again, same as vaporisation, we expect an increase in entropy but smaller than that during vaporisation. Therefore use the same equation but change vap to fus. Whatever you do do not use troutons rule for fusion *face palm* (its only for vaporisation).
Other times you may have to use your initiative is to break calculations down into steps.
Say we wanted to know △Svap for a substance at 25'C ? Well we are awesome and can work it out! Assume substance boils at 100'C.
- △Svap (25 'C) = △S (Heating liquid from 25 'C to 100 'C) Which we can do!
- + △Svap (100 'C) Which we can do!
- + △S (Cooling vapour from 100'C to 25 'C) Which we can do!
Key Points
- We now know how to account for entropy change on:
- Volume change
- Pressure change
- Change of state
- We now know how to use Trouton's rule to estimate enthalpies of vaporisations
- The entropy of a substance increases on fusion or vaporsation
Lecture Three
|
Molecular Interpretation
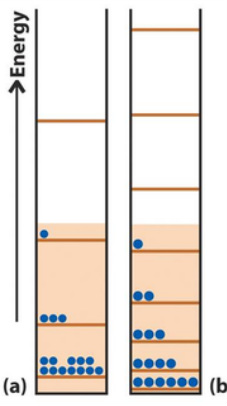
We have thought of entropy as disorder. So... Imagine a perfectly ordered substance. What is the best we can hope for? - Zero positional disorder - Zero thermal disorder This suggests there is a natural zero for entropy. The Third Law of Thermodynamics The entropies of all perfect crystals approach zero as the absolute temperature approaches zero Mathematically: S --> 0 as T --> 0 Statistical Entropy and the Boltzmann Equation S = kBlnW W is the number of ways that the atoms or molecules in the sample may be arranged to give the same energy, we say that W is the amount of microstates. Ensembles are large numbers of replicas: if W = 1: there is only one way of getting a given energy if W = 1000: there are lots of ways of getting a given energy Residual Entropy CO at T = 0 K; S = 4.6 J/K HCl at T = 0 K; S = 0 J/K HCl is more ordered than CO at 0K |
Try one yourself:
Calculate the statistical entropy of a solid made of four CO molecules at T = 0 when all molecules are perfectly alligned. Answer W = 1 S = kBln1 = 0 Try one yourself: Calculate the statistical entropy of a solid made of four CO molecules at T = 0 when the four molecules lie in random orientations Answer W = 2 x 2 x 2 x 2 = 2^4 S = kBln(2^4) = 3.8281 x 10^-23 J/K Try one yourself: Calculate the entropy of 1.00 mol of CO assuming each molecule may be orientated in one of two ways Answer S = kBlnW = kBln(2^6.02x10^23) = 5.76 J/K Try one yourself: The entropy of 1.00 mol of FClO3 at T = 0 is 10.1 J/K. Suggest an interpretation. Answer: Assume that FClO3 can adopt four different orientations in the crystal W = 4^6.02x10^23 S = kBlnW =kBln(4^6.02x10^23) =11.5 J/K/mol |
Equivalence of statistical and thermodynamic entropies
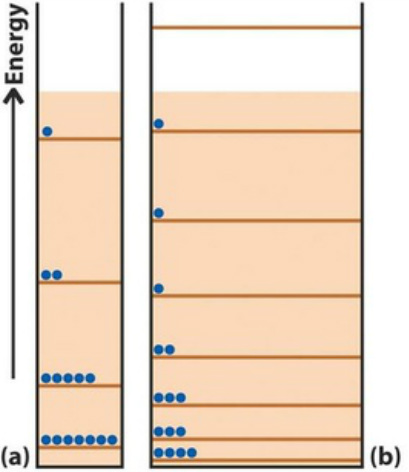
Regard a gas as lots of particles in a box
What happens when we increase the length of our box?
Regard a gas as lots of particles in a box
What happens when we increase the length of our box?
Equivalent of taking up more volume and therefore more positional disorder.
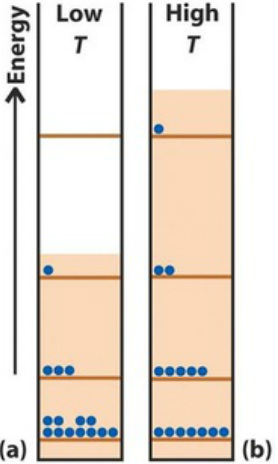
So what happens if we increase the temperature of the box?
So what happens if we increase the temperature of the box?
Statistical & Thermodynamic entropies
In both cases, more energy levels are made availiable...
In both cases, the entropy is higher...
Our lack of information has increased...
The amount of disorder has increased...
How do we show the equivalence?
In an isothermal expansion:
△S = nRln(V2/V1)
Assume the number of microstates for one molecule is proportional to volume...
then...
W = (constant x V)^N
△S = kBln(constant x V2)^N - kBln(constant x V1)^N
△S = NkBln{(constant x V2) / (constant x V1)}
△S = NkBln(V2/V1)
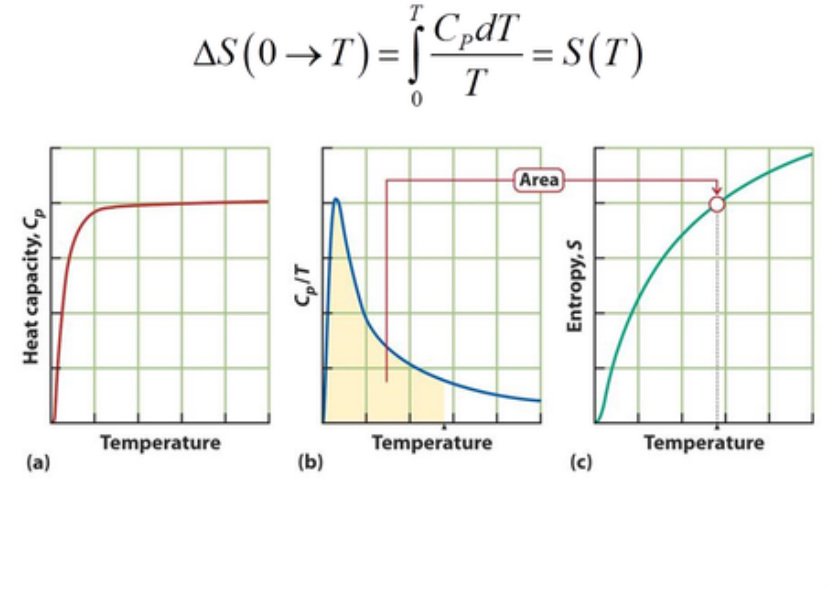
Standard molar entropies
The third law and Boltzmann formula allows us to calculate the absolute entropy of a substance:
S(T) = S(0) + △S(0 --> T)
= △S(0 --> T)
In both cases, more energy levels are made availiable...
In both cases, the entropy is higher...
Our lack of information has increased...
The amount of disorder has increased...
How do we show the equivalence?
In an isothermal expansion:
△S = nRln(V2/V1)
Assume the number of microstates for one molecule is proportional to volume...
then...
W = (constant x V)^N
△S = kBln(constant x V2)^N - kBln(constant x V1)^N
△S = NkBln{(constant x V2) / (constant x V1)}
△S = NkBln(V2/V1)
Standard molar entropies
The third law and Boltzmann formula allows us to calculate the absolute entropy of a substance:
S(T) = S(0) + △S(0 --> T)
= △S(0 --> T)
Consider Br2 and H2
The heavier the molecules, the more closely spaced energy levels
The heavier the molecules, the more closely spaced energy levels
Lecture Four
|
Systems and Surroundings
Water spontaneously forms ice at low temperature (increase in order) Cold packs for athletes (become cold spontaneously) Living systems (appear to increase order spontaneously) These go against what we have thought about entropy, but these do not take into account the total entropy change! Sneeze in the street ΔS = ΔH / T Overall Entropy Change You can judge the direction of a spontaneous change Entropy change of system: chemical change very difficult to judge Entropy change of surroundings: heat output easy to judge |
Entropy change of surroundings
Try one yourself Calculate the change in entropy of the surroundings when the following reaction goes to completion at 298 K. N2(g) + 3H2 --> NH3(g) ΔH = -92.22 kJ at 298 K Answer ΔS(surr) = -ΔS(system) = -ΔH/T = 309 J/K Try one yourself Assess whether the combustion of magnesium is spontaneous at 25'C 2Mg(s) + O2(g) --> 2MgO(s) ΔS = -217 J/K ΔH = -1202 kJ Answer ΔS(surr) = -ΔH(surr)/T ΔS(total) = ΔS + ΔS(surr) = 3.81 x 10^3 J/K |