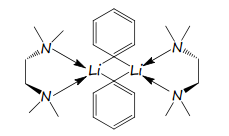
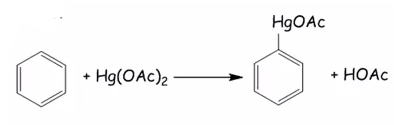
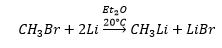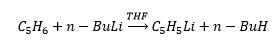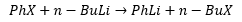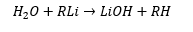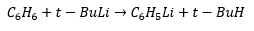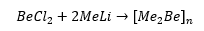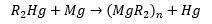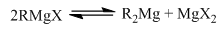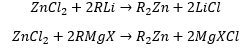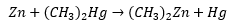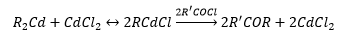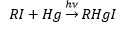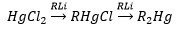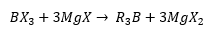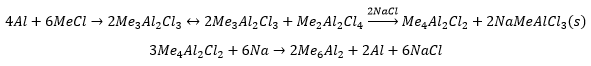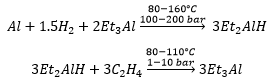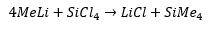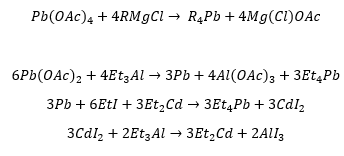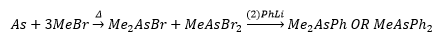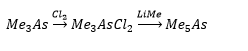Main group organometallics
This chemistry is based on the main group chemistry discussed earlier.
A compound is regarded as an organometallic if it contains at least on metal-carbon bond, this also includes metalloids such as B, Si and As. There is a great importance on these different organometallic chemistry is important.
These are regularly used in organic chemistry in the form of Grignard reagents, there are many more reactants that are found. Phosphine ligands are often used as a ligand as a supporting ligand than they are other compounds. There are many varieties of phosphine ligands. Other compounds such as ferrocene which involve large delocalised structures with a metal centre. It is important to remember that an organometallic compound HAS to have an organometallic bond.
There can be any number of carbon containing compounds can be bonded. This means that the entire variety of the carbon containing compounds is amazing due to the variation of the different metals that can be coupled to any metal. There is such a large variety of different bonding modes which allows a number of different bonding situations which change the properties of many different compounds. This allows a large number of highly useful compounds to be designed and placed in a large number of applications.
How are they used?
Organometallic chemistry is very important in different industrial processes as 90% of catalytic processes. These normally use organometallic bonds in an intermediate step. The organometallic materials chemistry is important as precursors to further reactions. Most of these can be volatile which can be purified and changed.
Synthesis of organometallic compounds
Group 1 organometallic reagents
Organometallic lithium is produced through the reaction of methylbromide and lithium.
Direct synthesis
This is the direct reaction between an organic compound directly reacting with a metal.
A compound is regarded as an organometallic if it contains at least on metal-carbon bond, this also includes metalloids such as B, Si and As. There is a great importance on these different organometallic chemistry is important.
These are regularly used in organic chemistry in the form of Grignard reagents, there are many more reactants that are found. Phosphine ligands are often used as a ligand as a supporting ligand than they are other compounds. There are many varieties of phosphine ligands. Other compounds such as ferrocene which involve large delocalised structures with a metal centre. It is important to remember that an organometallic compound HAS to have an organometallic bond.
There can be any number of carbon containing compounds can be bonded. This means that the entire variety of the carbon containing compounds is amazing due to the variation of the different metals that can be coupled to any metal. There is such a large variety of different bonding modes which allows a number of different bonding situations which change the properties of many different compounds. This allows a large number of highly useful compounds to be designed and placed in a large number of applications.
How are they used?
Organometallic chemistry is very important in different industrial processes as 90% of catalytic processes. These normally use organometallic bonds in an intermediate step. The organometallic materials chemistry is important as precursors to further reactions. Most of these can be volatile which can be purified and changed.
Synthesis of organometallic compounds
Group 1 organometallic reagents
Organometallic lithium is produced through the reaction of methylbromide and lithium.
Direct synthesis
This is the direct reaction between an organic compound directly reacting with a metal.
Methyl chloride is a gas so is not as easily used as methyl bromide. It is important to remember that lithium metal cannot make contact with water, lithium can even react with nitrogen which means argon must be used. This is a two electron process.
Metallation:
Metallation:
Butyl lithium is a base and will dissociate any acid which is stronger than butane. This means that any compound a stronger acid than butane will react with butyl lithium. This happens in the above reaction due to the acidity of the cyclopentadiene ligand.
Metal Halogen Exchange:
Metal Halogen Exchange:
When an organohalide is reacted with an organolithium there is an exchange of the reactants that takes place. These three methods do not just apply to methods they form in many different reactions.
Structure of organolithiums
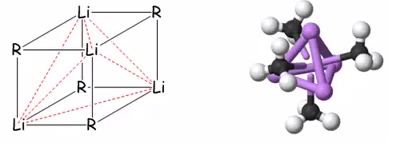
The lithium carbon bond is polar as the alkyl group is partially negatively charged making it a very polar solvents. Organolithiums are not found as typical compounds but rather form aggregates consisting of in weakly-polar solvents such as diethyl ether the compound can stabilise itself by forming aggregates of tetrahedral Li atoms with methyl groups on each face. The reason why this molecule dissolves even though it is highly polar is due to the fact that these aggregates help to stabalise the charge over the structure. There is very little bonding structure through this molecule.
Structure of organolithiums
The lithium carbon bond is polar as the alkyl group is partially negatively charged making it a very polar solvents. Organolithiums are not found as typical compounds but rather form aggregates consisting of in weakly-polar solvents such as diethyl ether the compound can stabilise itself by forming aggregates of tetrahedral Li atoms with methyl groups on each face. The reason why this molecule dissolves even though it is highly polar is due to the fact that these aggregates help to stabalise the charge over the structure. There is very little bonding structure through this molecule.
This is rather odd which is explained by the electron deficient bonding mode, this is in the same context of boranes. Instead of the bridging hydride groups there is a different bonding group:
The bonding here can be described in terms of a set of localised molecular orbitals formed by three hybridised lithium orbitals on each face of the tetrahedron and one hybrid orbital from giving an orbital that accommodates one electron pair. This can be seen as a 4-centre two electron bond. This can be seen simply as the carbon is on the face and therefore adjacent to three different lithium atoms. On average there will be two electrons per bond and therefore 2 electrons spread across four centres.
TWO ELECTRONS FOUR CENTRES.
This explains why these structures are soluble in organic solvents. The tetrahedral structures can bond together forming an oligomeric polyhedral with no overall dipole. This means that these polyhedral materials can be made soluble in non-polar solvents.
In the solid state these compounds can aggregate to even larger compounds which creates a very large polyhedra, the fact that these species are electron deficient. Thinking back to boron, the borane still doesn’t increase in electron count, although it does increase the number of orbitals being “used”. This stabalises the molecule although it is still very favourable to act as a base. The organolithium compounds can be broken with good donor ligands into smaller units. The breaking up of the aggregate causes an increase of reactivity as the aggregate is somewhat broken up.
The bonding here can be described in terms of a set of localised molecular orbitals formed by three hybridised lithium orbitals on each face of the tetrahedron and one hybrid orbital from giving an orbital that accommodates one electron pair. This can be seen as a 4-centre two electron bond. This can be seen simply as the carbon is on the face and therefore adjacent to three different lithium atoms. On average there will be two electrons per bond and therefore 2 electrons spread across four centres.
TWO ELECTRONS FOUR CENTRES.
This explains why these structures are soluble in organic solvents. The tetrahedral structures can bond together forming an oligomeric polyhedral with no overall dipole. This means that these polyhedral materials can be made soluble in non-polar solvents.
In the solid state these compounds can aggregate to even larger compounds which creates a very large polyhedra, the fact that these species are electron deficient. Thinking back to boron, the borane still doesn’t increase in electron count, although it does increase the number of orbitals being “used”. This stabalises the molecule although it is still very favourable to act as a base. The organolithium compounds can be broken with good donor ligands into smaller units. The breaking up of the aggregate causes an increase of reactivity as the aggregate is somewhat broken up.
The compound above shows how there are 6 electrons around the lithium. This is still electron deficient.
Due to the how the bonding occurs there is a significant amount of electron deficiency across the tetrahedral structure. This means that it is highly favourable for these compounds to take on one electron. This means that strong donor ligands can break these oligomers into smaller units leaving a small number of lithium carbon bonds.
Reactivity of organolithium compounds
Lithium reagents are pyrophoric (meaning they ignite spontaneously in air) and hydrolyse vigorously.
Alkyl lithiums have very polar bonds and are also electron deficient. This makes them highly reactive to any polar molecules. The R- section bonds forming an alkane and the lithium forms a hydroxide group.
Due to the how the bonding occurs there is a significant amount of electron deficiency across the tetrahedral structure. This means that it is highly favourable for these compounds to take on one electron. This means that strong donor ligands can break these oligomers into smaller units leaving a small number of lithium carbon bonds.
Reactivity of organolithium compounds
Lithium reagents are pyrophoric (meaning they ignite spontaneously in air) and hydrolyse vigorously.
Alkyl lithiums have very polar bonds and are also electron deficient. This makes them highly reactive to any polar molecules. The R- section bonds forming an alkane and the lithium forms a hydroxide group.
This can be seen as they readily react with any protons present in solution and can also be used as a base when sterically hindered. This makes them very strong bases, this acts almost totally as an electrophile.
It is possible to use organolithium reactants to form any organometallic compounds further to the right than lithium on the periodic table. This is incredibly important in metathesis reactions where a metal halide is reacted with an organolithium reactant forming a different organometallic compound.
t-Butyl lithium is a better base than n-butyl lithium. Due to the fact that the carboanion is more electron rich in t-butyl lithium making it a very strong base.
This is important in the process of the production of the organometallic compounds. As in all cases the lithium atom has a high positive charge and the carbon bond has a high negative charge. This can be forced even further by changing the metal that is bonded to the group.
Reactivity of the heavier alkali metals
The heavier metals are more electropositive meaning they are less attractive to electrons. This means that the bond becomes increasingly more polar. The increase in polarity can be seen in great detail as the bond is even more polar and more reactive. This is accentuated by the fact that is an ionic lattice and therefore fully ionic in character. As these compounds become heavier they become incredibly ionic and therefore incredibly reactive due to the pronounced carbanionic character.
The methyine anion is incredibly reactive, although most of these are so reactive they are almost impossible to use.
Summary
A compound is regarded as oranometallic if it contains at least on metal-carbon bond.
Synthesis structure, applications and reacitivity of organolithiums.
Aggregate formation
Electron deficient bonding (4c,2e)
Oligomeric polyhedral,
Strong bases and nucleophiles.
Higher alkali metal organometallic are more ionic and more reactive.
Alkaline earth metals
The alkaline earth metals are less electropositive and show very different properties to the alkali metals. The importance of this can be seen as there is a large difference between the compounds formed compared to the compounds formed in the alkali metal reagents.
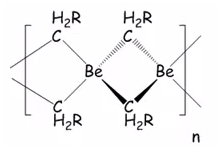
Beryllium reagents
Beryllium reagents are rarely used due to the fact that beryllium is a highly toxic material it is also in very low abundance this means there is little to no beryllium chemistry. In the reaction of beryllium (II) chloride with methyllithium a linear polymer is formed. This is a metathesis route for the formation of the chemical reaction.
t-Butyl lithium is a better base than n-butyl lithium. Due to the fact that the carboanion is more electron rich in t-butyl lithium making it a very strong base.
This is important in the process of the production of the organometallic compounds. As in all cases the lithium atom has a high positive charge and the carbon bond has a high negative charge. This can be forced even further by changing the metal that is bonded to the group.
Reactivity of the heavier alkali metals
The heavier metals are more electropositive meaning they are less attractive to electrons. This means that the bond becomes increasingly more polar. The increase in polarity can be seen in great detail as the bond is even more polar and more reactive. This is accentuated by the fact that is an ionic lattice and therefore fully ionic in character. As these compounds become heavier they become incredibly ionic and therefore incredibly reactive due to the pronounced carbanionic character.
The methyine anion is incredibly reactive, although most of these are so reactive they are almost impossible to use.
Summary
A compound is regarded as oranometallic if it contains at least on metal-carbon bond.
Synthesis structure, applications and reacitivity of organolithiums.
Aggregate formation
Electron deficient bonding (4c,2e)
Oligomeric polyhedral,
Strong bases and nucleophiles.
Higher alkali metal organometallic are more ionic and more reactive.
Alkaline earth metals
The alkaline earth metals are less electropositive and show very different properties to the alkali metals. The importance of this can be seen as there is a large difference between the compounds formed compared to the compounds formed in the alkali metal reagents.
Beryllium reagents
Beryllium reagents are rarely used due to the fact that beryllium is a highly toxic material it is also in very low abundance this means there is little to no beryllium chemistry. In the reaction of beryllium (II) chloride with methyllithium a linear polymer is formed. This is a metathesis route for the formation of the chemical reaction.
This forms a linear polymer. It is important to note that this polymer is insoluble which suggests that it is not electron deficient and cannot be broken up with compounds high in electron density. This material has a very low volatility, and also a very poor solubility. These compounds are electron deficient due to them only having four electrons one from each methyl group and two from the beryllium.
It is important to remember this reaction as the metathesis reaction is used for almost all structures ever.
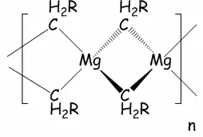
Magnesium reagents
Dialkyl magnesium compounds can be prepared by transmetallation.
There is great importance in magnesium reagents due to their use as Grignard reagents.
Magnesium reagents
Dialkyl magnesium compounds can be prepared by transmetallation.
There is great importance in magnesium reagents due to their use as Grignard reagents.
Dialkyl mercury compounds are very toxic and form a magnesium alkyl polymer.
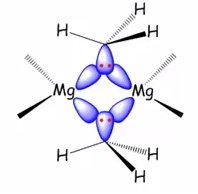
This is clearly 2 electron 3 centre bonding. Again only four electrons are present in the following bond:
These vacant orbitals form (3c, 2e) bonds the same found in diborane.
These compounds involve the reaction between an organometallic with the magnesium to produce a organomagnesium reagent. This produces a three centre two electron bond between the methyl groups and two magnesium centres.
These compounds involve the reaction between an organometallic with the magnesium to produce a organomagnesium reagent. This produces a three centre two electron bond between the methyl groups and two magnesium centres.
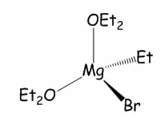
This can be carried out under nitrogen although the diethyl ether needs to be rigorously dried. It would be expected that two diethyl ether molecules will be bonded to the Grignard reagent in a four corrdinate compound.
It is possible for this crystal to be produced under the right conditions.
When producing Grignard reagents it is important there are only a few solvents that can be used to allow successful production of the Grignard reagent. This is due to the relative stability of the Grignard reagent formed, the magnesium metal centre is a strong Lewis base and will react rapidly with any protic solvents that are present. This means any solvents with a slightly acidic proton will react readily and rapidly with a Grignard reagent.
This means there are still many solvents that can still possibly be used, but the reaction doesn’t occur in many solvents such as toluene or cyclohexane. Why is this? The magnesium centre formed in the reaction has two empty p orbitals. For the reaction equilibrium to be favourable these need to be stabilised in some way, the best way of doing this is the presence of an oxygen that can donate electron density, such as that in an ether group.
So that leaves the common solvents; diethyl ether and tetrahydrofuran. Tetrahydrofuran is preferred of these two as the oxygen has much more directionality compared to the diethyl ether group. This directionality allows a more focussed stability of the magnesium group.
The other way to produce Grignard reagents is through the direct synthesis of magnesium with an alkylhalide. This is can be carried out in any aprotic solvent such as diethyl ether. In solution the RMgX species is solvated effectively. It is possible to crystallise monomeric four co-ordinate magnesium complexes from dry ether solutions. This is where magnesium is surrounded and maintains an electron octet.
Grignard reagents can also have a Schlenk equilibrium. This is where there is a rapid movement of a dialkyl magnesium group and a magnesium dihalide. When in a dioxane solution the dihalide complex becomes a solid allowing the removal of the dialkyl magnesium group. This shows how in a solvent there is a equilibrium between a dialkyl magnesium and a dialkyl halide. The chemistry occurring here has one alkyl group and a halide with a metal is found in almost all organometallics in group two.
When producing Grignard reagents it is important there are only a few solvents that can be used to allow successful production of the Grignard reagent. This is due to the relative stability of the Grignard reagent formed, the magnesium metal centre is a strong Lewis base and will react rapidly with any protic solvents that are present. This means any solvents with a slightly acidic proton will react readily and rapidly with a Grignard reagent.
This means there are still many solvents that can still possibly be used, but the reaction doesn’t occur in many solvents such as toluene or cyclohexane. Why is this? The magnesium centre formed in the reaction has two empty p orbitals. For the reaction equilibrium to be favourable these need to be stabilised in some way, the best way of doing this is the presence of an oxygen that can donate electron density, such as that in an ether group.
So that leaves the common solvents; diethyl ether and tetrahydrofuran. Tetrahydrofuran is preferred of these two as the oxygen has much more directionality compared to the diethyl ether group. This directionality allows a more focussed stability of the magnesium group.
The other way to produce Grignard reagents is through the direct synthesis of magnesium with an alkylhalide. This is can be carried out in any aprotic solvent such as diethyl ether. In solution the RMgX species is solvated effectively. It is possible to crystallise monomeric four co-ordinate magnesium complexes from dry ether solutions. This is where magnesium is surrounded and maintains an electron octet.
Grignard reagents can also have a Schlenk equilibrium. This is where there is a rapid movement of a dialkyl magnesium group and a magnesium dihalide. When in a dioxane solution the dihalide complex becomes a solid allowing the removal of the dialkyl magnesium group. This shows how in a solvent there is a equilibrium between a dialkyl magnesium and a dialkyl halide. The chemistry occurring here has one alkyl group and a halide with a metal is found in almost all organometallics in group two.
The formation of carbon carbon bonds to form a carbanion is very important the Grignard reagent is a nucleophile which attacks the carbonyl groups etc.
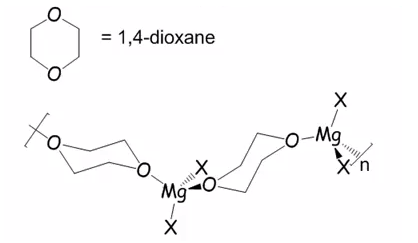
The use of dioxane can also be used to form a large coordination structure. 1,4 dioxane does not form a bidentate ligand being perfect for this functionality.
The use of dioxane can also be used to form a large coordination structure. 1,4 dioxane does not form a bidentate ligand being perfect for this functionality.
The electron count of eight here forms a much more stable polymer which is insoluble which can be precipitated from the solution. The equation them shifts meaning a very large amount of dialkyl magnesium to be produced. This can form halide free Grignard reagents.
Group 12 Organometallics
Divalent group 12 organometallics have some similarities to but do not participate in 2e3c bonds. This is important because these compounds tend to form linear geometries as they are held stable enough by their own electron densities. This are not considered transition metals as they do not have a partially filled orbital.
This can be seen in Metathesis- where zine chloride forms a dialkylated using an organolithium reagent or a Grignard reagent.
Group 12 Organometallics
Divalent group 12 organometallics have some similarities to but do not participate in 2e3c bonds. This is important because these compounds tend to form linear geometries as they are held stable enough by their own electron densities. This are not considered transition metals as they do not have a partially filled orbital.
This can be seen in Metathesis- where zine chloride forms a dialkylated using an organolithium reagent or a Grignard reagent.
This is an important reaction and can create a large amount of different alkylating agents.
And in transmetallation:
And in transmetallation:
R2Zn can be used as mild alkylating agents. This is important as these can only alkylate certiain groups over others.
The dialkyl zinc reagent formed is a linear molecule. It is not a polymer. Although zinc is an electropositive element it is not as electropositive as magnesium. This is still quite a polar bond with a significant amount of polarity of the bond.
Organocadmum compounds
This is synthesised through metathesis, the C-Cd bond is not as polar as the zinc carbon bond and only reacts with acyl chlorides and do not react with carbonyl bonds. The diminished Lewis acidity (ability to accept electrons) of organocadmium compounds can be used to synthesis ketones from acid chlorides.
This procedure is selective with other functional groups present eg. COOR.
The dialkyl zinc reagent formed is a linear molecule. It is not a polymer. Although zinc is an electropositive element it is not as electropositive as magnesium. This is still quite a polar bond with a significant amount of polarity of the bond.
Organocadmum compounds
This is synthesised through metathesis, the C-Cd bond is not as polar as the zinc carbon bond and only reacts with acyl chlorides and do not react with carbonyl bonds. The diminished Lewis acidity (ability to accept electrons) of organocadmium compounds can be used to synthesis ketones from acid chlorides.
This procedure is selective with other functional groups present eg. COOR.
Organomercury compounds
Direct synthesis and metathesis is the same way, although these specific compounds can be produced:
Direct synthesis and metathesis is the same way, although these specific compounds can be produced:
A reaction that only occurs in organomercury compounds is the following reaction this occurs in the opposite direction for all other organometallics apart from this mercury compound:
The hydrolysis of this compound reacts with water which is highly soluble and leads to mercury poisoning. Metal lined rubber gloves are needed for dimethyl mercury as this is a small compound. It is believed to have the same toxicity of other mercury compounds. The problem with dimethyl mercury is highly toxic and volatile.
Group 13
Organoboron compounds undergo metathesis:
Group 13
Organoboron compounds undergo metathesis:
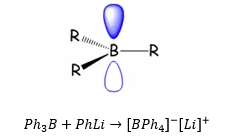
These are trigonal compounds that do not form dimers. These do act as lewis acids but these do not form dimers.
There are known as “ate” compounds:
There are known as “ate” compounds:
This can be used as a non-coordinating anion.
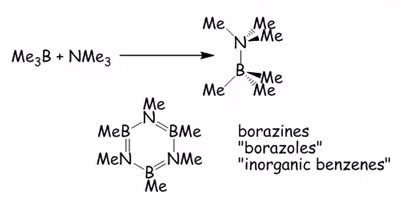
The organoboron compounds do form borazines and borazoles. This can be seen in the two compounds:
The organoboron compounds do form borazines and borazoles. This can be seen in the two compounds:
Borazine is planar but π electron are more localised than in benzene and doesn’t have the same stabilisation.
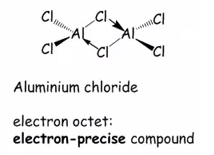
Organoaluminium compounds
The compounds are formed in the same way as many other compounds. These are very expensive and can be made by direct synthesis. This does not always go through into trialkyl species. The dimeric compound have three methyl groups and three chloride ligands.
Organoaluminium compounds
The compounds are formed in the same way as many other compounds. These are very expensive and can be made by direct synthesis. This does not always go through into trialkyl species. The dimeric compound have three methyl groups and three chloride ligands.
This can dimerise into bridging chloride groups this is important as an electron precise compound can be produced, this is due to the stabalisation of the lone pair being donated.
The dimerization with the methyl groups only forms a sextet of electrons.
The formation of aluminium alkyl compounds, these can be used for the synthesis of polymer chains. This is known as Ziegler’s synthesis.
Triethyl aluminium is much more preferred due to the easier conditions to be produced.
These are mostly used in the formation of detergents. The compounds are added to ethene which form much longer chains on the aluminium, when oxygen is added the aluminium carbon bonds can undergo insertion of the oxygen. These can be reacted with water to form aluminium hydroxide and alcohol. Long chained alcohols are used in detergents.
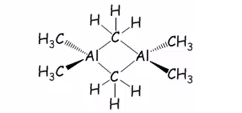
2 electron three centre bonding is therefore seen in aluminium and many other structures, this is still electron deficient but forms a more favourable system, it is important to remember that normal sigma bonds will form across this groups before any sharing bonding will form.
Not all organo metallic molecules form this bonds it is based on how Lewis acidic the compound is and how its steric demands allow it to form. This is not seen in trimethyl gallium for example, this makes it much more volatile as it is only a monomer. Bridging halides will form over alkyl groups as there are free electron donations to these and not electron deficient bonding systems. Hydride ligands bridge preferentially although this is due to steric effects, they still form 2 electron 3 centre bonding. This means the dimerization forms between the hydride group.
This means chlorides preferred to hydride which is preferred to alkyl.
These are mostly used in the formation of detergents. The compounds are added to ethene which form much longer chains on the aluminium, when oxygen is added the aluminium carbon bonds can undergo insertion of the oxygen. These can be reacted with water to form aluminium hydroxide and alcohol. Long chained alcohols are used in detergents.
2 electron three centre bonding is therefore seen in aluminium and many other structures, this is still electron deficient but forms a more favourable system, it is important to remember that normal sigma bonds will form across this groups before any sharing bonding will form.
Not all organo metallic molecules form this bonds it is based on how Lewis acidic the compound is and how its steric demands allow it to form. This is not seen in trimethyl gallium for example, this makes it much more volatile as it is only a monomer. Bridging halides will form over alkyl groups as there are free electron donations to these and not electron deficient bonding systems. Hydride ligands bridge preferentially although this is due to steric effects, they still form 2 electron 3 centre bonding. This means the dimerization forms between the hydride group.
This means chlorides preferred to hydride which is preferred to alkyl.
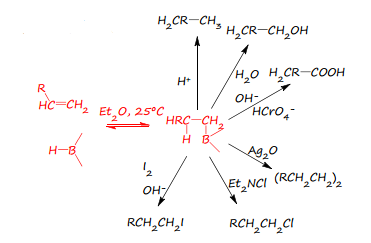
Hydroboration
Hydroboration is the formation of antimarkovnikov addition this bonds to the less steric carbon group which means cis addition is preferred and therefore occurs. The carbon boron functionality is very reactive.
Hydroboration is the formation of antimarkovnikov addition this bonds to the less steric carbon group which means cis addition is preferred and therefore occurs. The carbon boron functionality is very reactive.
Summary
Dialkyl magnesium complexes are linear polymers with 2e3c bonds.
RMgX Grignard reagents are solvate and participate in Schlenk equilibrium.
Group 12 complexes have similarities with group 2 but do not form 2e3c bonds.
Synthesis and structure of organoborons involving vacant orbitals and hydroboration.
Synthesis, structure and reactivity of organoaluminiums, electron deficient bonding/ dimeric structure. Pyrophoric.
Group 14
Group 14 organometalic compounds are very stable due to the formation Si-C bonds etc. This is seen due to the fact that TMS is so unreactive, this shows how the sample is so heavily electron stable. It is not based on thermodynamic principles due to the fact that the silicon carbon bonds will react down to water carbondioxide and silicon dioxide. This means that the kinetic explaination due to the difference in the lewis acidity. Trimethyl aluminium is a strong Lewis acid as it has a lone p orbital, the polarity of the aluminium carbon bond is larger difference in polarity making it more reactive. This is not an issue in silicon carbon bonds as much.
The main group alkyls that do not fit in the bonding orbitals are electron deficient.
The difference in electronegativity forms the different bond strength.
Organometallic reactivity
When faced with a reaction mechanism between any species with an organometallic species there are a number of considerations:
· Where the polarisation on the bonds lie?
· How polarised are these bonds?
· What vacant orbitals are present and what is the electron deficiency of the compound?
· What are the relative polarisabilities of the species that it is reacting with?
The polarisability of the bonds can easily be found by predicting the relative electronegativity’s of the elements present. The greater the difference in electronegativity’s the more reactive the species.
When the structures have been drawn it is important to recognise which areas of both the reactants and the products are polarised and therefore how they will react. A driving force for these reactions are the relative electronegativity’s of the two metals present in a transmetallation reaction.
It is also worth remembering that all of these organometallic complexes discussed are thermodynamically unfavourable with respect to oxidation to MOn H2O and CO2.
When these organometallics compounds have a vacant orbital and highly polarised bonds reactions with oxygen can also occur.
Preparation of organosilicon compounds
These occur in sol-gel chemistry type reactions. These react with water in silanols. This forms a silicon oxygen silicon linkage which forms a polymer eliminating HCl, this forms a silicone.
Dialkyl magnesium complexes are linear polymers with 2e3c bonds.
RMgX Grignard reagents are solvate and participate in Schlenk equilibrium.
Group 12 complexes have similarities with group 2 but do not form 2e3c bonds.
Synthesis and structure of organoborons involving vacant orbitals and hydroboration.
Synthesis, structure and reactivity of organoaluminiums, electron deficient bonding/ dimeric structure. Pyrophoric.
Group 14
Group 14 organometalic compounds are very stable due to the formation Si-C bonds etc. This is seen due to the fact that TMS is so unreactive, this shows how the sample is so heavily electron stable. It is not based on thermodynamic principles due to the fact that the silicon carbon bonds will react down to water carbondioxide and silicon dioxide. This means that the kinetic explaination due to the difference in the lewis acidity. Trimethyl aluminium is a strong Lewis acid as it has a lone p orbital, the polarity of the aluminium carbon bond is larger difference in polarity making it more reactive. This is not an issue in silicon carbon bonds as much.
The main group alkyls that do not fit in the bonding orbitals are electron deficient.
The difference in electronegativity forms the different bond strength.
Organometallic reactivity
When faced with a reaction mechanism between any species with an organometallic species there are a number of considerations:
· Where the polarisation on the bonds lie?
· How polarised are these bonds?
· What vacant orbitals are present and what is the electron deficiency of the compound?
· What are the relative polarisabilities of the species that it is reacting with?
The polarisability of the bonds can easily be found by predicting the relative electronegativity’s of the elements present. The greater the difference in electronegativity’s the more reactive the species.
When the structures have been drawn it is important to recognise which areas of both the reactants and the products are polarised and therefore how they will react. A driving force for these reactions are the relative electronegativity’s of the two metals present in a transmetallation reaction.
It is also worth remembering that all of these organometallic complexes discussed are thermodynamically unfavourable with respect to oxidation to MOn H2O and CO2.
When these organometallics compounds have a vacant orbital and highly polarised bonds reactions with oxygen can also occur.
Preparation of organosilicon compounds
These occur in sol-gel chemistry type reactions. These react with water in silanols. This forms a silicon oxygen silicon linkage which forms a polymer eliminating HCl, this forms a silicone.
There are many different uses for silicones.
Tin alkyls are very toxic. These are very useful in antifouling paints which stops the growths on the bottom of boats. Lead alkyls are also very toxic this is formed by the following:
Tin alkyls are very toxic. These are very useful in antifouling paints which stops the growths on the bottom of boats. Lead alkyls are also very toxic this is formed by the following:
The lead carbon bond is not very polar and this forms a homolytic splitting, forming two radicals. It is also important to note that the difference in stability is not thermodynamic but rather kinetic. The alkyl lead bond will not react easily as all avaliable orbitals are occupied. By contrast transition metals pocess vacant 3d orbitals which can be filled allowing facile reactions to occur. There is also a second kinetic argument to this where the size of transition metal ligands can block empty orbitals making it much harder for reactions to occur through a certain route.
Arsenic and bismuth and phosphorus are able to be classed as organometallic compounds. Group 15 organometallic chemistry is very complicated.
Preparation of R3E and R5E
Direct synthesis and metathesis are possible to the formation of the compound.
Preparation of R3E and R5E
Direct synthesis and metathesis are possible to the formation of the compound.
These form pseudotetrahedral structures. As there is a lone pair present and are therefore found as pyramidal structures and also as bases. Usually present as ligands.
Group 11 is considered as these are copper and silver, there are not many similarities between these and others. Although they are almost always in oxidation state 1. This has a large amount of organic industry. These are organo cuprates which can be used in conjugated enones, this is in a (Michael addition) reaction.
The insertion of silver into a bond cuases the formation of multiple radicals which can then move onto form a polymer chain and silver metals. This is very useful.
· Remember the differences and importance of kinetic and thermodynamic stability.
· The reactivity of main group organometallics is based on polarisability of the bonds.
· Group 14 organometallics do not have vacant orbitals and are therefore much more stable.
· The heavier group 15 elements form states of III and IV.
· Almost exclusively the organometallic chemistry of group 11 is of oxidation state I.
The insertion of silver into a bond cuases the formation of multiple radicals which can then move onto form a polymer chain and silver metals. This is very useful.
· Remember the differences and importance of kinetic and thermodynamic stability.
· The reactivity of main group organometallics is based on polarisability of the bonds.
· Group 14 organometallics do not have vacant orbitals and are therefore much more stable.
· The heavier group 15 elements form states of III and IV.
· Almost exclusively the organometallic chemistry of group 11 is of oxidation state I.