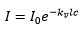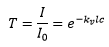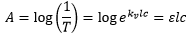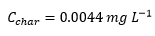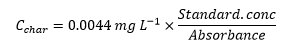Atomic spectroscopy surrounds the use of using radiation to measure for the presence of individual atoms in a sample. There are three types Atomic absorption spectroscopy, atomic emission spectroscopy, atomic fluorescence spectroscopy. (AAS, AES, AFS)
Here the atom is present in its ground state and is excited during atomisation. These excited atoms are then allowed to relax giving a different in energy between the ground and the excited state. The excitation is caused by the atomisation process in many cases.

The sample is atomised and then the absorption of radiation is measured from this atomised sample. The absorption is carried out in the same way as UV-Vis, it is common for noise issues to be present due to low frequencies.
The fluorescence spectrum of an excited sample is taken. This can only be carried out on molecules that are able to fluoresce. Which is limited meaning this method is not used as much as the other two methods.
The difference between this and emission spectroscopy is how the excitation occurs. This is the absorption of radiation and then emission of radiation where the absorption is usually heat in emission spectroscopy.
The difference between this and emission spectroscopy is how the excitation occurs. This is the absorption of radiation and then emission of radiation where the absorption is usually heat in emission spectroscopy.
Sources of atomisation
Atomisation can occur either continuously or discretely. The continuous process involves constantly injecting sample into the atomisation device so there is the constant production of material to be analysed. Discrete methods involve atomising one section of sample and atomising the next section of sample.
This is done by nebulization, the production of mist or spray, and then atomisation.
Solid extraction
An electric spark or a laser can be used to transfer a solid sample into the gas phase. This can occur through the sublimation process.
Nebulisation
Nebulisation works by the production of a mist through the spraying of the sample through a funnel of decreasing size. This causes velocity of the fluid to increase to a maximum as it is sprayed into the air. This uses flame rates 2-5 mL/min. This forms a cone of sample in a carrier gas. This spray can then be passed into the atomiser which can be carried out by a number of different methods:
Flame ionisers
Flame ionisers are the simplest forms of ionisers as they involve passing the sample through a flame causing excitation and oxidation. There are many aspects of the flame that need to be considered when the reaction occurs.
Atomisation can occur either continuously or discretely. The continuous process involves constantly injecting sample into the atomisation device so there is the constant production of material to be analysed. Discrete methods involve atomising one section of sample and atomising the next section of sample.
This is done by nebulization, the production of mist or spray, and then atomisation.
Solid extraction
An electric spark or a laser can be used to transfer a solid sample into the gas phase. This can occur through the sublimation process.
Nebulisation
Nebulisation works by the production of a mist through the spraying of the sample through a funnel of decreasing size. This causes velocity of the fluid to increase to a maximum as it is sprayed into the air. This uses flame rates 2-5 mL/min. This forms a cone of sample in a carrier gas. This spray can then be passed into the atomiser which can be carried out by a number of different methods:
Flame ionisers
Flame ionisers are the simplest forms of ionisers as they involve passing the sample through a flame causing excitation and oxidation. There are many aspects of the flame that need to be considered when the reaction occurs.
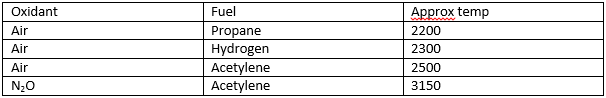
There are a range of
different fuels available allowing a range of different temperatures to be
produced.
Normally the air-acetylene mix is used unless very high temperatures are needed.
Interference
Any effect that changes the signal is an inteferent. This can be helped by adding a blank, there are also physical interferences, chemical interferences ionisation interferences. There are also issues with natural width of the line caused by the Heisenberg uncertainty principle.
The Heisenberg uncertainty principle adds enough uncertainty to the position of the electron to cause a natural broadening in the line taken in the spectrum. The Doppler broadening caused by thermal motion is also a good example of an un avoidable interferent. The gasous atoms will give slightly different wavelength depending if they are travelling towards or away from the detector. The collisional broadening is caused by the collision of atoms causing slightly different energy states to form.
The Heisenberg uncertainty principle adds enough uncertainty to the position of the electron to cause a natural broadening in the line taken in the spectrum. The Doppler broadening caused by thermal motion is also a good example of an un avoidable interferent. The gasous atoms will give slightly different wavelength depending if they are travelling towards or away from the detector. The collisional broadening is caused by the collision of atoms causing slightly different energy states to form.
Intensity
The intensity of emission is linear with concentration until very high concentrations where the intensity begins to level off as there is other absorption affects occurring. With an increase in temperature there are different populations of excited states. Therefore the emission from these levels also increases. Higher levels are less populated so these lines a weaker. Even in hot flames there is still only a very small amount of population of excited states so self-absorption is very common.
Sources
The source used for the radiation need to have a narrow spectral width and have a linear profile with temperature, this means that the temperature must be highly controlled. The intensity needs to be high to allow for good S/N ratio as well as a high durability.
· Cathode Lamp
Graphite furnace
The graphite furnace is one allows all the sample to be atomized the heating can be controlled depending on the sample which is of great use. Sample is usually 5-50 μL or solid. The sample is dried at 110°C. Pyrolysis takes place at 300-1100°C. This breaks up the sample. This is then rapidly heated to 2000-3000°C and atomisation occurs.
Plasma atomisers
Plasma atomisers have been around since the 70s, a plasma is a conducting gasous mixture which contains cations and electrons. This ions can absorb enough power from an external source to maintain a temperature as high as 10000K. There are three power sources that can be used:
· DC source
· Powerful RD generators.
· Powerful microwave generators.
RF or inductively coupled plasma provide the bast advantage, sensitivity and freedom from interference. These are available but these are much more expensive than DC plasma sources, which are cheaper and simpler.
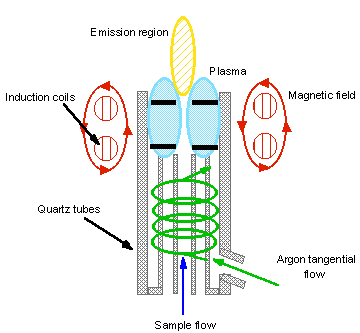
An inductively coupled plasma consists of a analytical zone, an initial radiation zone, an inductive region a load coil and torch.
Any effect that changes the signal is an inteferent. This can be helped by adding a blank, there are also physical interferences, chemical interferences ionisation interferences. There are also issues with natural width of the line caused by the Heisenberg uncertainty principle.
The Heisenberg uncertainty principle adds enough uncertainty to the position of the electron to cause a natural broadening in the line taken in the spectrum. The Doppler broadening caused by thermal motion is also a good example of an un avoidable interferent. The gasous atoms will give slightly different wavelength depending if they are travelling towards or away from the detector. The collisional broadening is caused by the collision of atoms causing slightly different energy states to form.
The Heisenberg uncertainty principle adds enough uncertainty to the position of the electron to cause a natural broadening in the line taken in the spectrum. The Doppler broadening caused by thermal motion is also a good example of an un avoidable interferent. The gasous atoms will give slightly different wavelength depending if they are travelling towards or away from the detector. The collisional broadening is caused by the collision of atoms causing slightly different energy states to form.
Intensity
The intensity of emission is linear with concentration until very high concentrations where the intensity begins to level off as there is other absorption affects occurring. With an increase in temperature there are different populations of excited states. Therefore the emission from these levels also increases. Higher levels are less populated so these lines a weaker. Even in hot flames there is still only a very small amount of population of excited states so self-absorption is very common.
Sources
The source used for the radiation need to have a narrow spectral width and have a linear profile with temperature, this means that the temperature must be highly controlled. The intensity needs to be high to allow for good S/N ratio as well as a high durability.
· Cathode Lamp
Graphite furnace
The graphite furnace is one allows all the sample to be atomized the heating can be controlled depending on the sample which is of great use. Sample is usually 5-50 μL or solid. The sample is dried at 110°C. Pyrolysis takes place at 300-1100°C. This breaks up the sample. This is then rapidly heated to 2000-3000°C and atomisation occurs.
Plasma atomisers
Plasma atomisers have been around since the 70s, a plasma is a conducting gasous mixture which contains cations and electrons. This ions can absorb enough power from an external source to maintain a temperature as high as 10000K. There are three power sources that can be used:
· DC source
· Powerful RD generators.
· Powerful microwave generators.
RF or inductively coupled plasma provide the bast advantage, sensitivity and freedom from interference. These are available but these are much more expensive than DC plasma sources, which are cheaper and simpler.
An inductively coupled plasma consists of a analytical zone, an initial radiation zone, an inductive region a load coil and torch.
The Torch consists of three concentric quartz tubes. Surrounding the end of the torch is a radiofrequency (RF) induction coil which induces a magnetic field. Gases flow in the outer tube to cool the torch. Having the gases flow tangently improves cooling stabilising the plasma. Torch is seeded with electrons using a spark. H accelerates these to energies sufficient to ionise the gases. All of this forms a flame of around 8000-10000 K.
Inductively coupled plasma gives a higher excitation temperature than any flame and has lower detection limits making it more favoured. This can be used for metals or non-metals as the temperatures are high enough to rapidly ionise metals and not allow carbon structures to form for non-metals. A long linear calibration is formed when running ICP which is useful as the rage can be very high. A useful source for both atomic and mass spectrometry. The running costs are high for an ICP machine though.
The main advantages to ICP are that there is a chemically inert environment formed. An optical path is smaller overall so there is less self-absorption The temperature gradient of the flame is also lower meaning it is more uniformly hot throughout making it highly effective. The calibration curve is also much better. The only drawback is large organic materials quickly block the inlets, ICP is quite fragile in this sense as it involves a large amount of “perfect” conditions to be able to form the very high temperatures.
Microwave induced plasmas have a frequency above 300 MHz and they form a standing wave in a cavity, this forms a high energy point which produces a plasma.
Inductively coupled plasma gives a higher excitation temperature than any flame and has lower detection limits making it more favoured. This can be used for metals or non-metals as the temperatures are high enough to rapidly ionise metals and not allow carbon structures to form for non-metals. A long linear calibration is formed when running ICP which is useful as the rage can be very high. A useful source for both atomic and mass spectrometry. The running costs are high for an ICP machine though.
The main advantages to ICP are that there is a chemically inert environment formed. An optical path is smaller overall so there is less self-absorption The temperature gradient of the flame is also lower meaning it is more uniformly hot throughout making it highly effective. The calibration curve is also much better. The only drawback is large organic materials quickly block the inlets, ICP is quite fragile in this sense as it involves a large amount of “perfect” conditions to be able to form the very high temperatures.
Microwave induced plasmas have a frequency above 300 MHz and they form a standing wave in a cavity, this forms a high energy point which produces a plasma.
Detectors
Multi-element determination is not possible if the detector used scans through the wavelengths slowly as there is only a very small amount of time when the sample is present in a way that can be analysed.
The absorption of a species is given by the Beer-Lambert law.
Multi-element determination is not possible if the detector used scans through the wavelengths slowly as there is only a very small amount of time when the sample is present in a way that can be analysed.
The absorption of a species is given by the Beer-Lambert law.
Which can be related to the transmission by:
The absorbance is given by:
The characteristic concentration is the concentration with 1% absorbance for a certain element.
The spectral responses are from either the emission or absorption between ground and excited states. Gas phase causes vary narrow widths of atomic lines compared to typical output from a mono/poly-chromator.
Background corrections
There can be many different interferences in the spectrum.
Chemical interference
Complex compounds like blood can form a large number of interfering compounds which can cause radiation absorption. The use of protective agents such as EDTA can help. Chemical interferenes arise from formation of chemical species that promote dissociation giving rise to depressed signals.
Continuum source corrections
This is pretty self explanatory, the source may have a non-linear decrease in radiation with temperature causing a non-uniform decrease in the wavelength.
Zeeman effect
The Zeeman effect is a strong magnetic field splits degenerate atomic spectral lines in several lines according to their Mj values. These have different polarization characteristics compared to which atom is being analysed. This non-degeneracy causes a σ- π σ* lines to form. The emission line is only found from the π transition but there are absorptions at the σ- and σ* which give negative movements in the background.
Ionisation interferences
There are some compounds that form ions that give a different spectra completely. These can make a spectrum much more complex which is not favourable. Ion suppressors can be added. These stop the ions forming, these are usually similar, but more easily ionised, structures. These form ions preferentially affecting the samples ability to ionise.
Background corrections
There can be many different interferences in the spectrum.
Chemical interference
Complex compounds like blood can form a large number of interfering compounds which can cause radiation absorption. The use of protective agents such as EDTA can help. Chemical interferenes arise from formation of chemical species that promote dissociation giving rise to depressed signals.
Continuum source corrections
This is pretty self explanatory, the source may have a non-linear decrease in radiation with temperature causing a non-uniform decrease in the wavelength.
Zeeman effect
The Zeeman effect is a strong magnetic field splits degenerate atomic spectral lines in several lines according to their Mj values. These have different polarization characteristics compared to which atom is being analysed. This non-degeneracy causes a σ- π σ* lines to form. The emission line is only found from the π transition but there are absorptions at the σ- and σ* which give negative movements in the background.
Ionisation interferences
There are some compounds that form ions that give a different spectra completely. These can make a spectrum much more complex which is not favourable. Ion suppressors can be added. These stop the ions forming, these are usually similar, but more easily ionised, structures. These form ions preferentially affecting the samples ability to ionise.