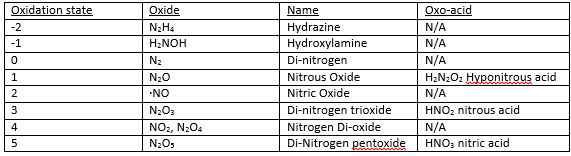
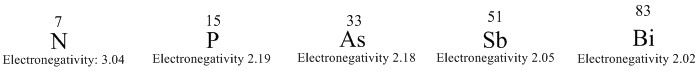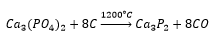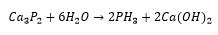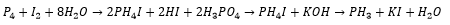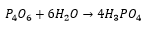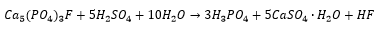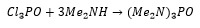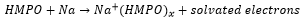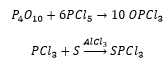The group 15 elements are important in the periodic table groups these consist of the important elements of nitrogen and phosphorus that are found a great deal in inorganic chemistry.
Nitrogen
Reactivity
The reactivity of nitrogen is strikingly un-reactive as nitrogen is only found as one allotrope; di-nitrogen. Di-nitrogen has a triple bond that forms due to the size of the atoms and the electron excess that they have. Bonds can form from one p-orbital to the other p-orbital and the lone pairs left do not allow bonding of more nitrogen atoms, so no chain can form.
This leaves nitrogen being an inert gas with an extremely strong bond ∆H=+944 kJ mol-1. In nature the fixation of nitrogen in the atmosphere is done with a very complex organo-metallic system based around molybdenum. Industrially it is carried out in the Haber-Bosch process.
Reactivity
The reactivity of nitrogen is strikingly un-reactive as nitrogen is only found as one allotrope; di-nitrogen. Di-nitrogen has a triple bond that forms due to the size of the atoms and the electron excess that they have. Bonds can form from one p-orbital to the other p-orbital and the lone pairs left do not allow bonding of more nitrogen atoms, so no chain can form.
This leaves nitrogen being an inert gas with an extremely strong bond ∆H=+944 kJ mol-1. In nature the fixation of nitrogen in the atmosphere is done with a very complex organo-metallic system based around molybdenum. Industrially it is carried out in the Haber-Bosch process.
Phosphorus Chemistry
Phosphorus in stark contrast with nitrogen forms many different allotropes.
Phosphorus can be found in the white phosphorus form: P4 this is found to be a trigonal pyramidal structure, this structure has highly strained bonds and has lone pairs of electrons surrounding it. This makes it very likely for this molecule to react with other molecules with the large amount of electron density it has around the outside. This molecule is highly reactive in the air and ignites instantaneously.
With intense heating (1800˚C) P2 does form this is similar to N2 but only forms due to large excitation of the molecules.
Red phosphorus
Red phosphorus is much less reactive and does not ignite spontaneously it is a solid powder with a 3-dimensional structure on heating of this compound black phosphorus is formed this forms trigonal pyramidal coordination on its atoms but in a puckered form.
Phosphines
Metallic Phosphines can be produced by the following reaction mechanism.
Metal rich phosphine’s are hard, brittle and have metallic conductivity although they are mostly chemically inert. An example of this is the monophosphide InP which is mostly found in the zinc blende structure. There can also be phosphorus rich structures which are stabalised with cations such as P73- and P113-.
Hydrolysis of these compounds can give phosphines or alkylated phosphines.
Hydrolysis of these compounds can give phosphines or alkylated phosphines.
Alkaline hydrolysis of PH4I can also be used:
Phosphines have the same shape and structure of ammonia except phosphorus in the place of nitrogen. PR3 where R is an alkyl group is important in many different coordination chemistry reactions and as a substituent ligand.
The importance of phosphines as ligands is due to the electronegativity of phosphorus it has an electronegativity of nearly exactly the same as hydrogen’s. This means that constituents added to the phosphorus can cause the phosphine group to be either electron rich or electron poor and the ligand to be electron withdrawing or donating based on the electron richness of the phosphine group.
The use of bidentate phosphine ligands can also control the isomerism of the metal centre. This can be very useful when creating different compounds that may need to follow a set isomerism themselves.
Phosphorus oxides
When white phosphorus is exposed to oxygen the oxygen reacts removing some of the strain from the bonds. When reacting in excess oxygen multiple bonds forming P4O10.
The importance of phosphines as ligands is due to the electronegativity of phosphorus it has an electronegativity of nearly exactly the same as hydrogen’s. This means that constituents added to the phosphorus can cause the phosphine group to be either electron rich or electron poor and the ligand to be electron withdrawing or donating based on the electron richness of the phosphine group.
The use of bidentate phosphine ligands can also control the isomerism of the metal centre. This can be very useful when creating different compounds that may need to follow a set isomerism themselves.
Phosphorus oxides
When white phosphorus is exposed to oxygen the oxygen reacts removing some of the strain from the bonds. When reacting in excess oxygen multiple bonds forming P4O10.
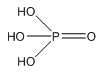
Phosphoric acid
Phosphoric acid is a tri-basic acid and therefore has three different pKas this is important to note and should be understood when discussing multi-basic acids. The phosphorus acids are poruced but hydrolysing the phosphorus oxides.
Phosphoric acid is a tri-basic acid and therefore has three different pKas this is important to note and should be understood when discussing multi-basic acids. The phosphorus acids are poruced but hydrolysing the phosphorus oxides.
The structure of phosphoric acid shows how delocalisation of the oxygen around the molecule can stabilise the negative charge formed.
With an increase in E=O groups there is a decrease in the pKa so a increase in acidity.
Paulings rule is a way to predict the pKa of an acid by counting the number of E=O bonds there are present and using the equation:
8 – 5P
Where P is the number of E=O bonds. Pauling also states that the pKa increases by five units for each successive proton transfer.
These can be produced by the addition of calcium phosphate fluoride with acid and water. The HF produced can be removed as Na2SiF6. In the following reaction:
Paulings rule is a way to predict the pKa of an acid by counting the number of E=O bonds there are present and using the equation:
8 – 5P
Where P is the number of E=O bonds. Pauling also states that the pKa increases by five units for each successive proton transfer.
These can be produced by the addition of calcium phosphate fluoride with acid and water. The HF produced can be removed as Na2SiF6. In the following reaction:
The phosphate salts can form extremely alkali solutions such as the dissociation of Na3PO4. The salts of phosphates are often used as buffers often used is Na2HPO4 this has the capability to accept and donate protons.
Phosphorus-Nitrogen compounds
PIII and PV are very similar to the phosphorus oxide compounds. The Lewis acidity of the phosphate group can be used to convert halide groups to amine groups.
Phosphorus-Nitrogen compounds
PIII and PV are very similar to the phosphorus oxide compounds. The Lewis acidity of the phosphate group can be used to convert halide groups to amine groups.
This forms hexamethylphosphoramide which is a highly polar strongly coordinating solvent. This has the ability to form solvated electrons which have a large number of unique properties.
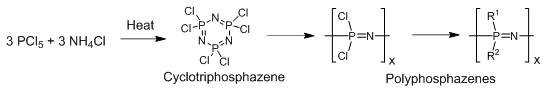
The Phosphazenes are a significant brand of chemistry as there is great amount of substitution reactions that can be found from these compounds:
As shown these phosphazenes can undergo a large amount of substitution forming a large number of different possible compounds.
Phosphorus halides
PIII
- PF3 is a colourless gas of boiling point -101.8°C.
- PCl3 is a liquid and is an important starting material in most ligand formation reactions.
- PBr3 is a liquid.
- PI3 is a crystalline solid.
PV
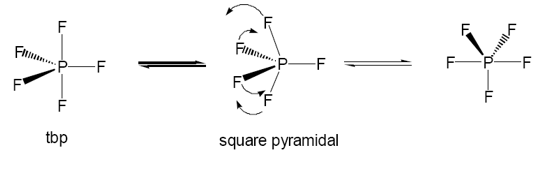
PF5 is a gas with boiling point -84.5°C, it maintains a trigonal bipyrimadal structure. Which can undergo Berry rotation. There is an importance of this rotation as the NMR shows only one electron environment due to the usual two.
Phosphorus halides
PIII
- PF3 is a colourless gas of boiling point -101.8°C.
- PCl3 is a liquid and is an important starting material in most ligand formation reactions.
- PBr3 is a liquid.
- PI3 is a crystalline solid.
PV
PF5 is a gas with boiling point -84.5°C, it maintains a trigonal bipyrimadal structure. Which can undergo Berry rotation. There is an importance of this rotation as the NMR shows only one electron environment due to the usual two.
PCl5
PCl5 is a liquid solution in non-polar solvents it can form a gas as monomeric PCl5 molecules. An important point to make is when placed into a polar solvent solution these monomers break down forming an adduct of [PCl4]+[PCl6]-. This is also able to be easily distorted and when in the gas phase it often breaks down forming PCl3 and the production of chlorine.
P(III) halides are readily oxidised to phosphorus oxyhalides.
PCl5 is a liquid solution in non-polar solvents it can form a gas as monomeric PCl5 molecules. An important point to make is when placed into a polar solvent solution these monomers break down forming an adduct of [PCl4]+[PCl6]-. This is also able to be easily distorted and when in the gas phase it often breaks down forming PCl3 and the production of chlorine.
P(III) halides are readily oxidised to phosphorus oxyhalides.
Arsenide
Arsenide is found in a large number of different ores and has a number of allotropes.
Two crystal structures that are formed are realgar red crystals and orpiment yellow. These are both formed with arsenide bonding to sulphides.
Realgar: As4S4.
Orpiment yellow: As2S3
Arsenate: AsO4- very similar to phosphate PO43-.
As, Sb Bi
All of these can form trivalent molecules which can act as ligands for metal centres. This is very similar to phosphines capabilities. These can all act as Lewis bases as they have a lone pair much like nitrogen and phosphorus.
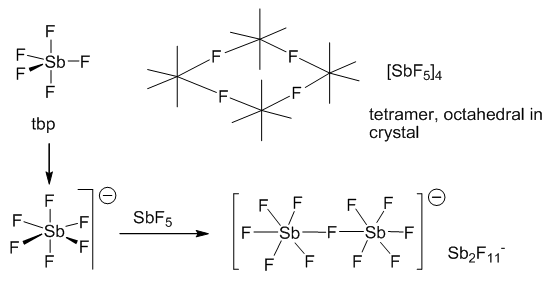
They can also form pentalvalent structures. These only form with the electronegative fluorine and chlorine atoms and not bromine or iodine. They act as Lewis acids and will normally gain another fluoride atom to become negatively charged. These are very stable non-coordinating atoms. These can be seen below:
Arsenide is found in a large number of different ores and has a number of allotropes.
Two crystal structures that are formed are realgar red crystals and orpiment yellow. These are both formed with arsenide bonding to sulphides.
Realgar: As4S4.
Orpiment yellow: As2S3
Arsenate: AsO4- very similar to phosphate PO43-.
As, Sb Bi
All of these can form trivalent molecules which can act as ligands for metal centres. This is very similar to phosphines capabilities. These can all act as Lewis bases as they have a lone pair much like nitrogen and phosphorus.
They can also form pentalvalent structures. These only form with the electronegative fluorine and chlorine atoms and not bromine or iodine. They act as Lewis acids and will normally gain another fluoride atom to become negatively charged. These are very stable non-coordinating atoms. These can be seen below:
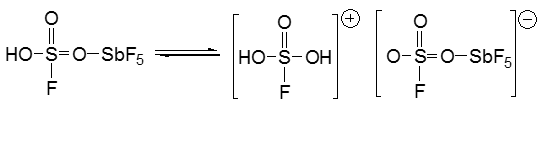
These stabilised anion structures have been put to good use by being used for super acid chemistry, the non-aqueous conditions that need to be set make these harder to produce although they are such a strong acid they can protonate methane, this makes them a very strong acid.