Electron counting and oxidation states almost always come up here are the key points and any less common examples of electron counts.
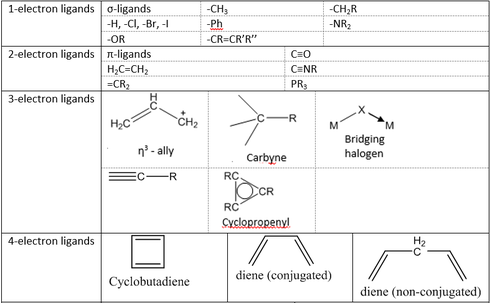
Electron counting
|
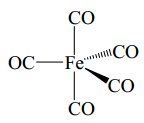
This complex below has 5 carbonyl groups each donating 2 electrons and the iron is in group 8 so donates 8 electrons. This means this is an 18 electron species.
|
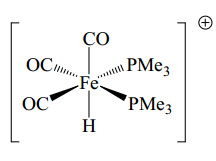
This complex has 5 two electron donating species. The hydride ligand donates one electron this means there are 19 electrons present. Once the positive charge is taken into account one electron is removed making this and 18 electron species.
|
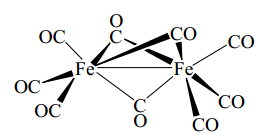
For the more complex complexes such as this one it is important to work though systematically. Each iron has 3 un-bridged CO groups which equates to 6 electrons. The bridging CO groups are one electron each. This leaves 6+8+3=17 electrons.
This can be overcome by the two iron atoms sharing electrons this is a donation of one electron bring both complexes to 18 electron species. |