Oxidative addition is a chemical reaction that occurs in a large amount of organometallic complexes. It is important to recognise that a transition metal complex is not needed for this reaction to occur.
The oxidation states of complexes are found by the heterolytic bond breaking of the metal centre to the ligands. Assigning oxidation states and electron counting should be kept separately in thought process. They are two very different processes. The complex structure has to be considered carefully for the bonding modes as the same species can have a different oxidation state depending on the ligands.
In hydride species the hydride is shown as being a -1 to the oxidation state. If the metal hydride dissolves then a proton can be formed meaning the metal centre is really a +1 charge now. This shows they are a formalism and only a good approximation.
Oxidative additions and reductive eliminations actually cause a difference in oxidation number when the bond is broken heterolytically. When the most electronegative element carries the negative charge.
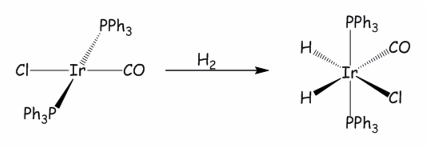
The sum of all the oxidation numbers is equal to the charge of the complex. Alkyl groups are (-1)! Hydrogens are hydride ligands and therefore (-1) also. This means the addition of hydrogen to a complex is oxidative addition.
In hydride species the hydride is shown as being a -1 to the oxidation state. If the metal hydride dissolves then a proton can be formed meaning the metal centre is really a +1 charge now. This shows they are a formalism and only a good approximation.
Oxidative additions and reductive eliminations actually cause a difference in oxidation number when the bond is broken heterolytically. When the most electronegative element carries the negative charge.
The sum of all the oxidation numbers is equal to the charge of the complex. Alkyl groups are (-1)! Hydrogens are hydride ligands and therefore (-1) also. This means the addition of hydrogen to a complex is oxidative addition.
This is the “vascus” compound.
There needs to be a vacant orbital for oxidative addition to occur. This produces two metal A and two metal B bonds. A vanadium (V) compound cannot undergo oxidative addition as this cannot be any more oxidised. If there is a compound which is already six coordinate it is not possible for this compound to undergo oxidative addition.
· Non-bonding electron pair.
· Two vacant coordination sites in.
· An oxidation state two units higher must be energetically accessible.
Always remember that some high oxidation states are not accessible Ni(iv) does not exist as it is so energetically unfavourable. So cannot exist.
These reactions are not restricted to transition metals or organometallics. Tin dichloride can react further with chlorine to become SnCl4. These are all examples of oxidative addition.
Transition metal complexes that undergo these reactions are typically metals in d8 d10 or d0 are seen regularly undergoing these reaction types.
It is possible for a large array of compounds to undergo oxidative addition. Carbon Carbon bonds can be added to a complex, water can be added, carboxylic acids can be added, many many examples of species can be added in oxidative addition.
Reactivity of the metal
With increase of electron density of the metal they are more likely to undergo oxidative addition. This means that electron withdrawing ligands on the metal hinder oxidative addition. This means that PPh3 is a very powerful oxidative reaction complex. Anionic comples are more reactive than neutral ones.
When oxidative addition occurs the ligands are cis to one another in the product.
The opposite of oxidative addition is reductive elimination. Reductive elimination can only occur when the ligands are cis to one another. The catalysis of compounds is important as this allows hydrogenation to occur.
There needs to be a vacant orbital for oxidative addition to occur. This produces two metal A and two metal B bonds. A vanadium (V) compound cannot undergo oxidative addition as this cannot be any more oxidised. If there is a compound which is already six coordinate it is not possible for this compound to undergo oxidative addition.
· Non-bonding electron pair.
· Two vacant coordination sites in.
· An oxidation state two units higher must be energetically accessible.
Always remember that some high oxidation states are not accessible Ni(iv) does not exist as it is so energetically unfavourable. So cannot exist.
These reactions are not restricted to transition metals or organometallics. Tin dichloride can react further with chlorine to become SnCl4. These are all examples of oxidative addition.
Transition metal complexes that undergo these reactions are typically metals in d8 d10 or d0 are seen regularly undergoing these reaction types.
It is possible for a large array of compounds to undergo oxidative addition. Carbon Carbon bonds can be added to a complex, water can be added, carboxylic acids can be added, many many examples of species can be added in oxidative addition.
Reactivity of the metal
With increase of electron density of the metal they are more likely to undergo oxidative addition. This means that electron withdrawing ligands on the metal hinder oxidative addition. This means that PPh3 is a very powerful oxidative reaction complex. Anionic comples are more reactive than neutral ones.
When oxidative addition occurs the ligands are cis to one another in the product.
The opposite of oxidative addition is reductive elimination. Reductive elimination can only occur when the ligands are cis to one another. The catalysis of compounds is important as this allows hydrogenation to occur.