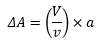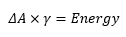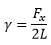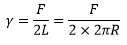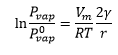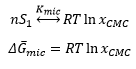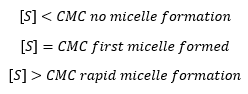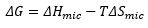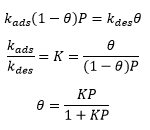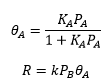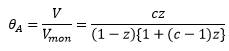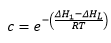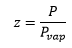The key points and equations of surface chemistry
Energy and surface tension
|
The energy change when there is a change in the surface is represented by the equation:
|
The measurement of surface tension can be by two methods, either a a stretching of a material or the ring in a liquid experiment.
|
The change of vapour pressure caused by the presence of a porous substance can be given by the following equation
Ir the radius is negative is can mean that the radius is curved the other way.
|
Micelle formation
|
Micelle formation
Micelle formation is directly linked to the surface concentration of the surfactant present which is linked to solution concentration. Micelles are amalgamations of monomer species. |
At low monomer concentrations there are no micelles present.
The number of monomers in a micelle is normally between 80-100.
|
The free energy of micelle formation is represented by:
The entropy of micelle formation is still positive as
|
Adsorption
|
Physisorption is where molecules adsorb onto a solid surface with weak van der Waals interaction. This follows the usual potential energy surface.
Chemisorption is where the molecules are initially physisorbed but then under go a chemical bond to the solid surface. All of the adsorption models discussed deal with chemisorption because the life times of the adsorption structures formed are much higher. The discussion of coverage is base on the differences of the bonding of molecules in an adsorption model. |
Chemisorption passes through a high energy transition state during the process, this is where the bond formation takes place.
|
Chemisorption models.
|
Langmuir isotherm model
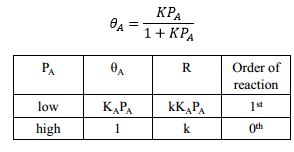
As this isotherm is used for chemisorption it can be used regularly for prediction of the effect of pressure on the reaction rate. This is carried out simply by changing the pressure value in the equation.
Bimolecular reactions
Where there is a reaction between an already adsorbed molecule and another molecule that has yet reacted the following equations can be used: |
Type II isotherm
As the pressure is increased it is possible for multiple layers to form. These can form in a number of different ways. The layering of the molecules has three different growth models.
BET
The BET isotherm model shows how the at higher concentrations there are more multiplayers produced. The Langmuir isotherm assumptions are used. The model has quite a complex equation represented by: |