Principles of aromaticity
[If you're looking for Aromatic reactions]
The principle of aromaticity is based on how electrons in symmetrical p orbitals can interact with one another forming π bonds. When these π interact with on another in the same plane and can form a fully cyclic system the molecule becomes highly stabilized. The stabilization produced from these π systems is known as aromaticity and produces some very distinct properties.
Aromaticity and anti-aromaticity
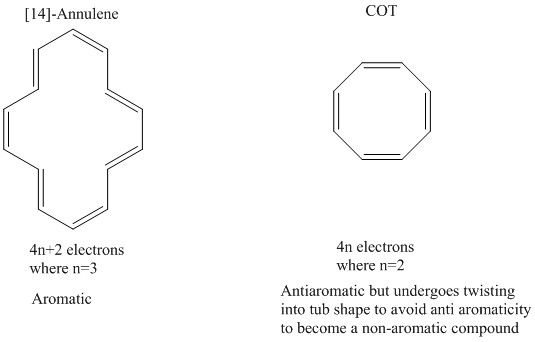
An aromatic compound can only form when Hückel’s rules have been obeyed. This is where there are 4n+2 electrons present. When this occurs it can be favourable for a planar aromatic state to form.
Remember:
When the number of electrons present only equate to 4n electrons then the compound is known to be anti-aromatic and will distort itself to become tub shaped. COT (cyclooctatetraene) is a great example of this as it pulls itself up on either side to not have alternating bonds.
An aromatic compound can only form when Hückel’s rules have been obeyed. This is where there are 4n+2 electrons present. When this occurs it can be favourable for a planar aromatic state to form.
Remember:
- Molecule needs to obey Huckel's rule (4n+2).
- Molecule needs to be planar.
- All atoms in the ring need to be Sp2 hybridized.
When the number of electrons present only equate to 4n electrons then the compound is known to be anti-aromatic and will distort itself to become tub shaped. COT (cyclooctatetraene) is a great example of this as it pulls itself up on either side to not have alternating bonds.
So why are compounds like COT so unstable compared to aromatic molecules? Study of the orbital energies can give the answer to this question:
The orbital diagram for benzene and COT are shown below. This shows that benzene's HOMO is fully occupied meaning the electrons are highly stabilized in this configuration. In the COT diagram it is obvious that two radicals have formed. These radicals are highly reactive and are very unfavorable in a molecule. This shows the importance of the molecule obeying Huckel's rule as this allows the formation of a fully occupied HOMO.
The orbital diagram for benzene and COT are shown below. This shows that benzene's HOMO is fully occupied meaning the electrons are highly stabilized in this configuration. In the COT diagram it is obvious that two radicals have formed. These radicals are highly reactive and are very unfavorable in a molecule. This shows the importance of the molecule obeying Huckel's rule as this allows the formation of a fully occupied HOMO.
With this good predictions can be made surrounding the relative stability of different aromatic groups depending on how many electrons are present.
So that covers Huckel's rule,
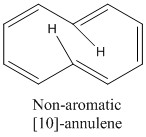
The molecule also needs to be able to become planar. An interesting example of this is [10]-annulene, this obeys Huckel's rule and it would be expected for it to have aromatic properties but it does not!
The presence of the central hydrogen atoms cause the molecule to twist slightly meaning it does not have aromatic properties.
So that covers Huckel's rule,
The molecule also needs to be able to become planar. An interesting example of this is [10]-annulene, this obeys Huckel's rule and it would be expected for it to have aromatic properties but it does not!
The presence of the central hydrogen atoms cause the molecule to twist slightly meaning it does not have aromatic properties.
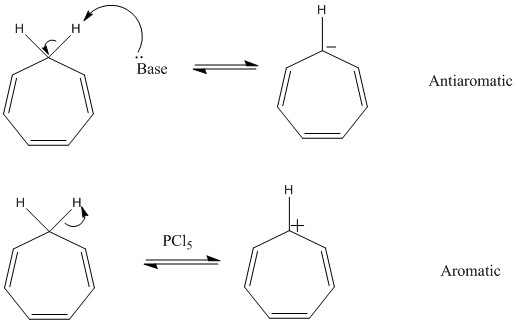
This now leaves cases where hybridization is important. This is very clear in cycloheptatriene. This molecule can undergo loss of a hydrogen forming either a +ve or -ve ion depending on the conditions. This leaves either an Sp2 hybridized negatively charged molecule which does not obey Huckel's rule and actually becomes anti-aromatic. The positivley charged ion is actually aromatic, it obeys Huckel's rule, has the ability to be planar and has all Sp2 hybridized carbon atoms.
NMR
One does not simply avoid NMR in a topic.
Due to the aromatic nature of the compounds it could be predicted that there will be a significant change in the NMR. A lot of the aromatic stabilization is based on the fact that there is a higher electron density surrounding the nuclei in the ring. This would cause the proton NMR to be affected.
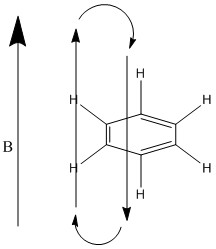
The fact that the electrons in the ring are free to move around it is very easy for them to interact with a magnetic field that is being induced across them. This initial field is then countered by the induced magnetic field caused by the current of the electrons. This causes the protons on the outside of the ring to be further de-shielded as the induced magnetic field works with the initial magnetic field. This is shown below:
This causes the electrons in the ring to be further down shifted.
For larger annulene groups such as [18]-annulene there is a larger down shift for the outside electrons but also the electrons in the centre have a larger shielding effect. This causes the ppm values to be around -3 for the centre protons.
For larger annulene groups such as [18]-annulene there is a larger down shift for the outside electrons but also the electrons in the centre have a larger shielding effect. This causes the ppm values to be around -3 for the centre protons.
Heteroaromatic compounds
There are some aromatic compounds that are made from different compounds. The importance when it comes to analysing whether these compounds are aromatic or not is to work out if they obey Hückel’s rule and if there are enough free p orbitals to allow a full cyclic ring of electrons.
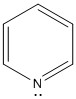
Pyridine is a good example of this:
There are some aromatic compounds that are made from different compounds. The importance when it comes to analysing whether these compounds are aromatic or not is to work out if they obey Hückel’s rule and if there are enough free p orbitals to allow a full cyclic ring of electrons.
Pyridine is a good example of this:
Here the lone pair of electrons can easily bond to a hydrogen ion making it act as a base. This shows that there is no need for the lone pair of electrons to bond in the aromatic π ring.
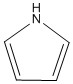
In the example of Pyrrole in its natural state there is a hydrogen atom bonded to it. This is because the lone pair is bonded in the π ring. This means there are 3 pairs of electrons present and therefore enough electrons for the ring to be aromatic.
In the example of Pyrrole in its natural state there is a hydrogen atom bonded to it. This is because the lone pair is bonded in the π ring. This means there are 3 pairs of electrons present and therefore enough electrons for the ring to be aromatic.
Larger molecules can be harder to spot and not always obvious if the electrons are being added to the aromatic ring or are free to bond to the other molecules. A quick test can be to see whether the molecule is basic or not. If there are many lone pairs then the molecule will be basic. If all of these electrons are being used in to maintain aromaticity then the molecule will not be basic.