Electrophillic Aromatic Substitution Reactions
Click here to view last years material
Remember EDG are Activating and direct attack to Ortho and Para positions
Remember EWG are Deactivating and direct attack to the Meta position
Effect of Substituents to Orientation of Electrophillic Aromatic Substitution
EDG Very Activating
|
OH (Phenols)
|
Ortho and Para (Conjugation)
|
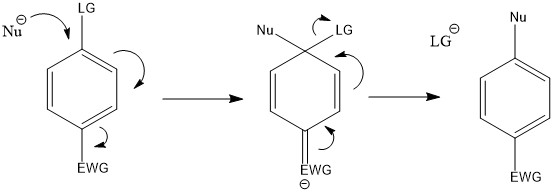
Nucleophillic Aromatic Substitution Reactions
Goes through an addition-elimination mechanism
Requires:
- Nucelophile (Nu-) such as a molecule with oxygen, nitrogen or cyanide that contains a lone pair
- Leaving Group (LG) such as a halide
- Electron Withdrawing Group (EWG) such as a carbonyl, nitro or cyanide group ortho or para to the leaving group.
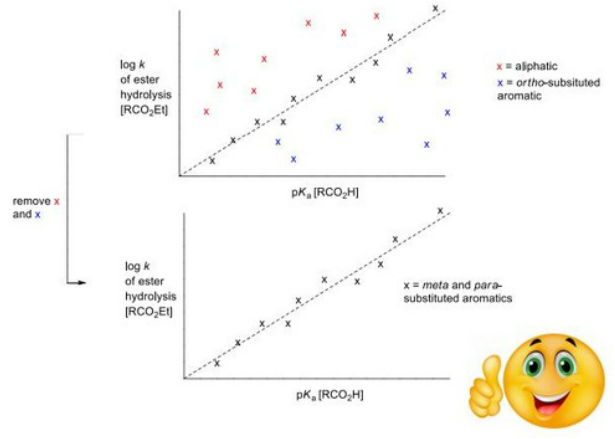
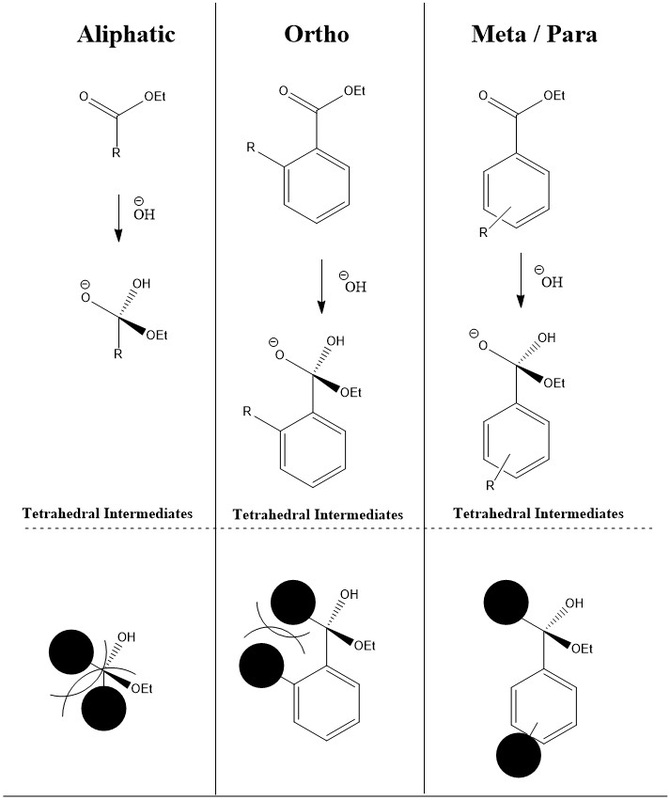
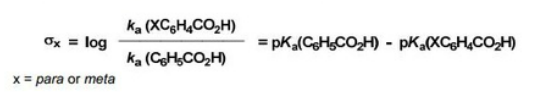
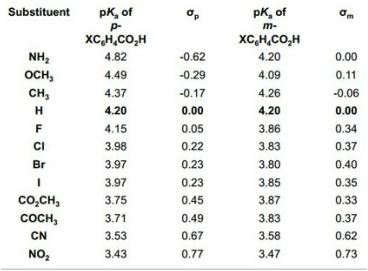
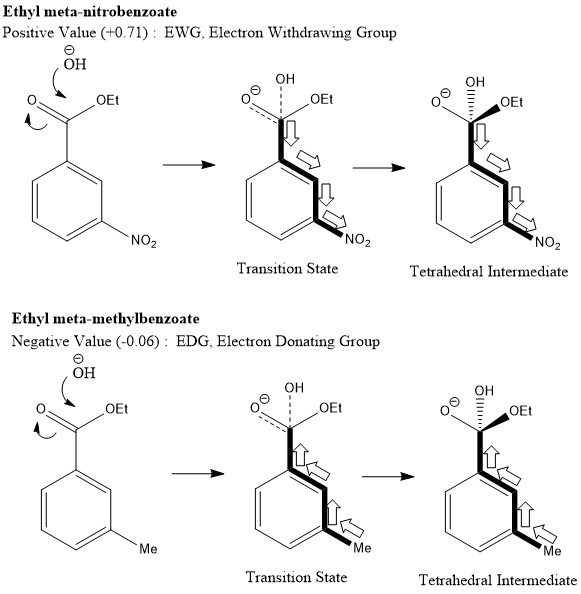
Hammett Relationship
"The effect of a substituent on the intermediate or transition state created in the course of a reaction."
Hammett quantified a value for this by comparing the rate of hydrolysis of esters (kinetic) with the pKa of the corresponding acid (thermodynamic). The following was discovered.
A plot of rate, k (y axis), against pKa (x axis) shows overall no correlation. But if the side group of the ester was aliphatic (no aromatic) then it would like above a 'trend line' and if it was ortho-substituted then it would lie below the trend line.
|