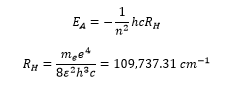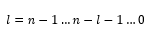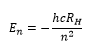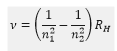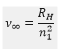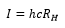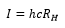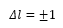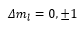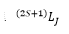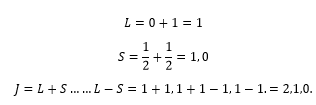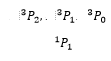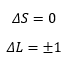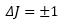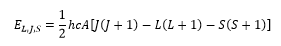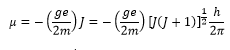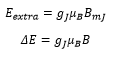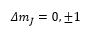When electrons are excited from one energy state to another there are a number of selection rules that are allowed and therefore can occur and a number that are forbidden. This can be used effectively to find the presence of atoms in a sample. Term symbols can be used as a short hand to describe the electronic structure of the atom.
What needs to be understood?
A lot of this is discussed in the quantum mechanics lecture notes. But this will just give a quick run over of what needs to be understood. This should also be in slightly more layman’s terms over what should be understood.
The hydrogen atom
The hydrogen atom has distinct solutions for the Schrodinger equation. This gives excact values for the energy levels according to the Bohr model and how the different orbitals lie at different energies. These can be represented by the equations:
A lot of this is discussed in the quantum mechanics lecture notes. But this will just give a quick run over of what needs to be understood. This should also be in slightly more layman’s terms over what should be understood.
The hydrogen atom
The hydrogen atom has distinct solutions for the Schrodinger equation. This gives excact values for the energy levels according to the Bohr model and how the different orbitals lie at different energies. These can be represented by the equations:
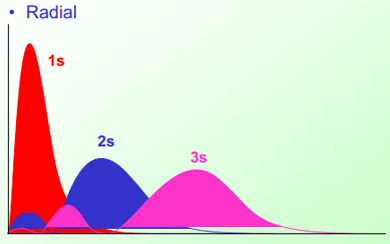
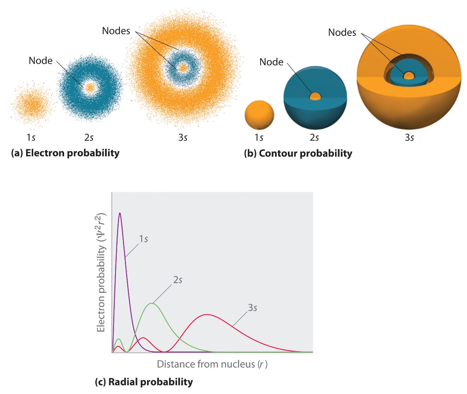
This diagram shows how the 1s orbital is held much closer to the nucleus than the 2s and 3s orbitals although there is orbital penetration. This is where there is probability of finding the electrons of the 3s and 2s in the 1s orbital range.
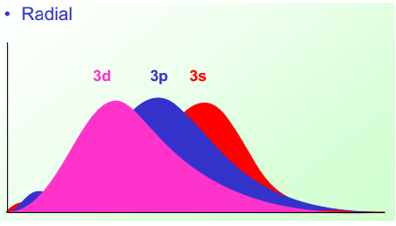
This diagram shows how the 3d orbital is held closer to the shell than the 3s orbital althought there are sections of the 3s and 3p orbital closer than the 3d. This means that all orbitals are of the same energy in an hydrogenic atoms so long as they are at the same energy shell or principle quantum number n.
When the atom is no longer hydrogenic electronic repulsions causes a difference in orbital energies. This difference in orbital energies means that the electronic spectrum becomes more complex.
Exchange integrals
The exchange integrals- the movement of electrons from one energy level to another level is a purely quantum mechanical effect. At the macroscopic level this is purely the movement of electrons from one energy state (high or low) to another energy state (low or high).
The properties of an electron can be mapped using quantum numbers. These are derived from the Schrodinger equation for the hydrogenic atom. This can then be used to form the radial distribution function which can then be used to calculate the probability of an electron being found in each place.
Principle quantum number
The principle quantum number is a discrete value which describes which electron shell the electron is in. The difference between principle quantum numbers is the largest energy difference that is found in the atomic spectra. There is great importance that when a hydrogenic atom is looked at, an atom with just one electron, that the only energy difference is found between the principle quantum number as all other energy states are degenerate.
The principle quantum number is represented by the letter “n”.
Where n directly relates to the electron shell. Where n=1 the first energy shell is being discussed. The total number of nodes that are present are equal to the principle quantum number minus one.
Azimuthal quantum number
The azimuthal quantum number is closely related to the value of the principle quantum number:
When the atom is no longer hydrogenic electronic repulsions causes a difference in orbital energies. This difference in orbital energies means that the electronic spectrum becomes more complex.
Exchange integrals
The exchange integrals- the movement of electrons from one energy level to another level is a purely quantum mechanical effect. At the macroscopic level this is purely the movement of electrons from one energy state (high or low) to another energy state (low or high).
The properties of an electron can be mapped using quantum numbers. These are derived from the Schrodinger equation for the hydrogenic atom. This can then be used to form the radial distribution function which can then be used to calculate the probability of an electron being found in each place.
Principle quantum number
The principle quantum number is a discrete value which describes which electron shell the electron is in. The difference between principle quantum numbers is the largest energy difference that is found in the atomic spectra. There is great importance that when a hydrogenic atom is looked at, an atom with just one electron, that the only energy difference is found between the principle quantum number as all other energy states are degenerate.
The principle quantum number is represented by the letter “n”.
Where n directly relates to the electron shell. Where n=1 the first energy shell is being discussed. The total number of nodes that are present are equal to the principle quantum number minus one.
Azimuthal quantum number
The azimuthal quantum number is closely related to the value of the principle quantum number:
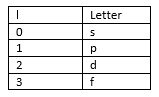
This shows how there are multiple values of l for each energy level. These values of l are given a number of different letters:
These electrons have radial distribution functions:
This shows the s functions which can then be used to explain why when there are more than one electron the orbitals are no longer degenerate in the energy levels. This is due to the electrons having a non-zero probability of being found at the nucleus.
The magnetic quantum number
The magnetic quantum number specifies the component of orbital angular momentum of an electron through and with n and l this gives a full one electron wavefunction. The values of the magnetic quantum number range from +l to –l.
This gives the vector that the magnetic quantum number is going in. This is explained by the complex equations that surround the electron motions.
Although all of these orbitals are orthogonal it can be easy to spot why the degeneracy occurs due to the identical symmetry that all of the orbitals maintain.
Molecular electronic transitions
When an electron is being considered in a hydrogenic ion and ignoring the spin state the electron can be described by the three quantum numbers, n, l and ml. The energy level can be represented as:
The magnetic quantum number
The magnetic quantum number specifies the component of orbital angular momentum of an electron through and with n and l this gives a full one electron wavefunction. The values of the magnetic quantum number range from +l to –l.
This gives the vector that the magnetic quantum number is going in. This is explained by the complex equations that surround the electron motions.
Although all of these orbitals are orthogonal it can be easy to spot why the degeneracy occurs due to the identical symmetry that all of the orbitals maintain.
Molecular electronic transitions
When an electron is being considered in a hydrogenic ion and ignoring the spin state the electron can be described by the three quantum numbers, n, l and ml. The energy level can be represented as:
The frequency of the transitions can be calculated by the Rydberg equation:
The ionization energy can be taken from the Rydberg constant.
The ionisation energy of the atom is given as:
This shows how for a hydrogen atom the ionisation enthalpy is a constant.
The selection rules that are present for electronic transitions these are based on how the parity must change, and that the change must only be one higher or lower as that is all that is possible.
The selection rules that are present for electronic transitions these are based on how the parity must change, and that the change must only be one higher or lower as that is all that is possible.
This shows how for a hydrogen atom the ionisation enthalpy is a constant.
The selection rules that are present for electronic transitions these are based on how the parity must change, and that the change must only be one higher or lower as that is all that is possible.
The selection rules that are present for electronic transitions these are based on how the parity must change, and that the change must only be one higher or lower as that is all that is possible.
This leaves the change in the angular momentum will also change in the transition:
The transition can only be equal to 0 when there is already angular momentum present in the atom.
Spin
Electron spin is one of the other ways in which the electrons properties can be described, although an up spin and a down spin are degenerate unless placed in an electric field. Each electron has a spin of s = ±1/2
Term symbols
Term symbols are a spectroscopers way of describing the electron orbitals that are present.
Which can further lead to electron states being described and ordered in energy as of Hund’s rule.
Spin
Electron spin is one of the other ways in which the electrons properties can be described, although an up spin and a down spin are degenerate unless placed in an electric field. Each electron has a spin of s = ±1/2
Term symbols
Term symbols are a spectroscopers way of describing the electron orbitals that are present.
Which can further lead to electron states being described and ordered in energy as of Hund’s rule.
This is where S is the total spin of the atom and L is the total azimuthal quantum number and J is the total orbital angular momentum present. These are the addition of the orbitals that can then be described by a series of states.
This is given by the integers between the values being taken and the L value being represented as the letter or the orbital.
This can be explained by the following example:
He-1s12p1
Here a helium atom has an electron in the 1s orbital and the 2p orbital. The electronic state of this example is shown as:
This is given by the integers between the values being taken and the L value being represented as the letter or the orbital.
This can be explained by the following example:
He-1s12p1
Here a helium atom has an electron in the 1s orbital and the 2p orbital. The electronic state of this example is shown as:
This gives the possible values as being:
Using these three electron states as examples Hund’s rules can be used to explain energy levels present of these states.
Using these term symbols it is easy to identify which transitions are allowed by the following selection rules:
Using these term symbols it is easy to identify which transitions are allowed by the following selection rules:
Hund’s rules
Rule 1: The state with the maximum multiplicity lies lowest in energy. A singlet has a higher energy than a triplet.
Rule 2: For a given multiplicity the term with the largest value of L has the lowest energy.
Rule 3: For atoms with less than ½ filled shells the energy with the lowest value of J lies lowest in energy.
Spin-orbit coupling
Rule 1: The state with the maximum multiplicity lies lowest in energy. A singlet has a higher energy than a triplet.
Rule 2: For a given multiplicity the term with the largest value of L has the lowest energy.
Rule 3: For atoms with less than ½ filled shells the energy with the lowest value of J lies lowest in energy.
Spin-orbit coupling
This gives the total energy of the state of the energy. When there is an energy transition both have a given energy and the difference in energies can be calculated.
The Zeeman effect
The Zeeman effect can be given as the electrons possess a magnetic moment that as a result of the spin angular momenta. These moments will interact with an externally applied magnetic field and the resulting shifts in energy should be apparent in the spectrum of the atom.
The Zeeman effect can be given as the electrons possess a magnetic moment that as a result of the spin angular momenta. These moments will interact with an externally applied magnetic field and the resulting shifts in energy should be apparent in the spectrum of the atom.
For an electron the g value is approximately 2.
The z component is the axis in which the electric field is acting. This is closely related to the Bohr magneton. This shows the difference in energies depending on the difference in electrons. The splitting between states is given as:
The z component is the axis in which the electric field is acting. This is closely related to the Bohr magneton. This shows the difference in energies depending on the difference in electrons. The splitting between states is given as:
The Zeeman effect does impose the extra selection rule which is: