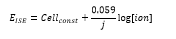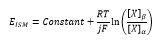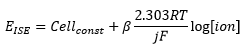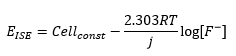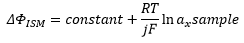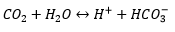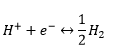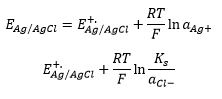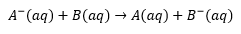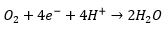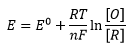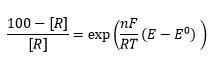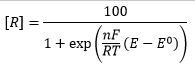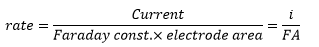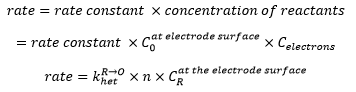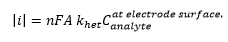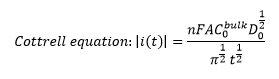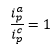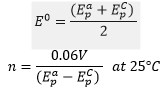These are an abridged form of the lecture notes all in one place. I have tried to remove any unnecessary information from Profs lecture note's.
Analytical chemistry
Electroanalytical chemistry: Ion selective electrodes
Electroanalytical chemistry uses the relationship between a charged ion or a redox compound and an electrode to quantitatively analyse the analyte.
The principles are based on the measure of the potential difference of the current.
The electrochemical cell
There are a number of examples of electrochemical cells, the simpliest is shown below, all electrochemical cells have:
· Two electrodes
· Electrolytes
· Inclusion of a device to measure current or voltage.
· A saltbridge if the two solutions cannot mix.
Electroanalytical chemistry: Ion selective electrodes
Electroanalytical chemistry uses the relationship between a charged ion or a redox compound and an electrode to quantitatively analyse the analyte.
The principles are based on the measure of the potential difference of the current.
The electrochemical cell
There are a number of examples of electrochemical cells, the simpliest is shown below, all electrochemical cells have:
· Two electrodes
· Electrolytes
· Inclusion of a device to measure current or voltage.
· A saltbridge if the two solutions cannot mix.
This shows how the formation of AgCl occurs on the surface of the silver metal.
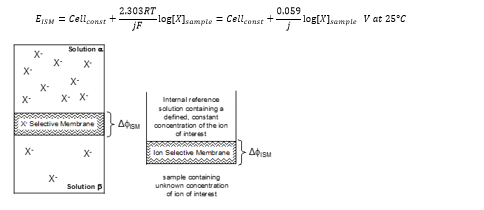
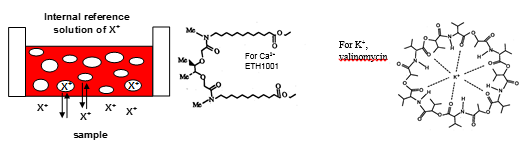
Ion selective electrodes
Definition: Electroanalytical devices that allow measurement of the concentration of a selected ion in a sample (solution) of interest by virtue of the electric potential difference (voltage difference) due to the concentration of that ion.
There are many uses for ion electrodes specialising in water and food analysis. As well as biomedical applications in blood, plasma etc.
These have an analytical advantage:
· They have a fast response time as they are continuously measuring of the sample.
· Simple to use.
· Cheap, small, robust and portable.
· High sensitivity and selectivity for the ion.
· No interference from colour changes.
· Passive measurement (some exceptions).
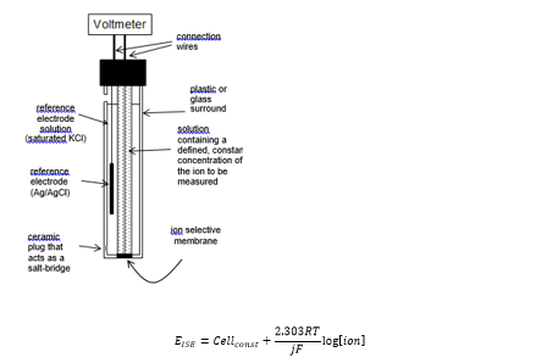
Design of ISEs
All ISEs follow a similar construction to one another. Known as a combination electrode.
The sensing electrode is specific although the reference is normally the same Ag, AgCl and KCl mix. The potential difference is measured from this electrode which is related to the concentration of the ions present.
Ion selective electrodes
Definition: Electroanalytical devices that allow measurement of the concentration of a selected ion in a sample (solution) of interest by virtue of the electric potential difference (voltage difference) due to the concentration of that ion.
There are many uses for ion electrodes specialising in water and food analysis. As well as biomedical applications in blood, plasma etc.
These have an analytical advantage:
· They have a fast response time as they are continuously measuring of the sample.
· Simple to use.
· Cheap, small, robust and portable.
· High sensitivity and selectivity for the ion.
· No interference from colour changes.
· Passive measurement (some exceptions).
Design of ISEs
All ISEs follow a similar construction to one another. Known as a combination electrode.
The sensing electrode is specific although the reference is normally the same Ag, AgCl and KCl mix. The potential difference is measured from this electrode which is related to the concentration of the ions present.
· F – Faraday’s constant 96484 C mol-1
· R – Gas constant 8.314 J K-1 mol-1.
· T – Temperature in Kelvin.
· J – Charge of ion.
This means at 25°C:
· R – Gas constant 8.314 J K-1 mol-1.
· T – Temperature in Kelvin.
· J – Charge of ion.
This means at 25°C:
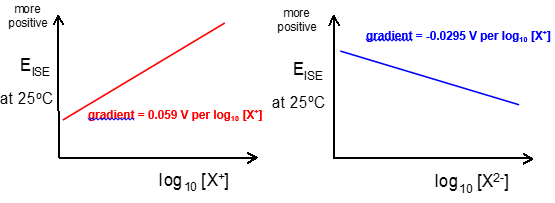
· Double charge:- ½ gradient.
· Positive gradient:- Positive charge.
· Negative gradient:- Negative charge.
How do ISE’s work:
When there is a difference in ion concentration, there is a potential difference formed across the membrane:
· Positive gradient:- Positive charge.
· Negative gradient:- Negative charge.
How do ISE’s work:
When there is a difference in ion concentration, there is a potential difference formed across the membrane:
There is no longer a potential difference when the electrochemical potential μ is equal to zero.
Incorperating this equation into the electrode potential:
Incorperating this equation into the electrode potential:
By defining [X]α as the constant concentration of X in the internal reference solution of the ISE:
ISEs Theory and practice
Deviations from the Nernst equation need to be taken into account for analysis. The electromotive efficiency is used to describe this behaviour. This is represented by the β.
Deviations from the Nernst equation need to be taken into account for analysis. The electromotive efficiency is used to describe this behaviour. This is represented by the β.
Most of the time β=1 but not always. The equation shows that there is a direct relationship between the concentration of ions at the surface and the electrode potential. This means that:
· Measurements with ISEs should be stirred to allow equilibriation.
Most of the time β=1 but not always. The equation shows that there is a direct relationship between the concentration of ions at the surface and the electrode potential. This means that:
· Measurements with ISEs should be stirred to allow equilibriation.
· Calibration of ISEs may be performed by taking readings of in two solutions defined by concentration and assuming linear behaviour.
Selectivity coefficients
There is always some interference by an interfering ion which leads to the selectivity coefficient kA,B:
· Measurements with ISEs should be stirred to allow equilibriation.
Most of the time β=1 but not always. The equation shows that there is a direct relationship between the concentration of ions at the surface and the electrode potential. This means that:
· Measurements with ISEs should be stirred to allow equilibriation.
· Calibration of ISEs may be performed by taking readings of in two solutions defined by concentration and assuming linear behaviour.
Selectivity coefficients
There is always some interference by an interfering ion which leads to the selectivity coefficient kA,B:
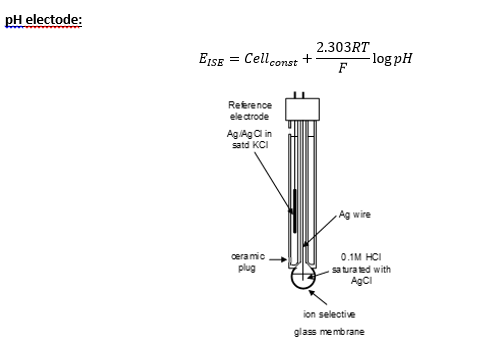
Types of ion electrode
· Glass electrodes: pH, NH4+, Na+.
· Solid state electrodes F-. Cl-, S2-.
· Polymer electrodes K+, Ca2+.
· Glass electrodes: pH, NH4+, Na+.
· Solid state electrodes F-. Cl-, S2-.
· Polymer electrodes K+, Ca2+.
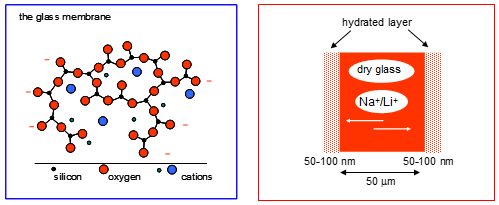
This uses a silica glass membrane which hydrogen ions can pass through.
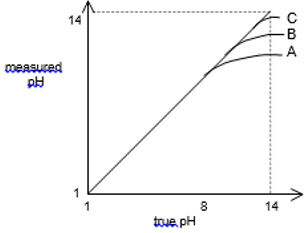
At higher pH levels the alkaline effect begins to take place, this is when positive counter ions such as Na in NaOH, can begin to interfere this can be seen to a large amount coming to maximum pH levels;
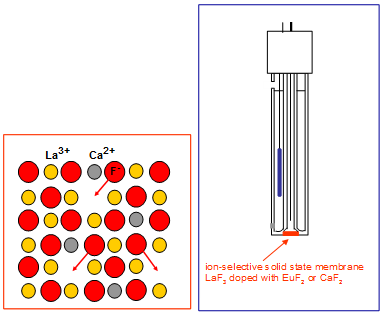
Flouride selective electrode
The ISM is a crystal of LaF3 doped with CaF2 and BaF2 this is a schottkey defect.
The pH needs to be considered as OH- ions can cause a large change in measured concentration.
· OH- ions need to be avoided.
· pH < 3 causes HF formation and should be avoided.
· Al(III) and Fe (III) complex strongly to F-.
Then using TISAB flouide electrodes have a nerstien response from 10-5 to 1 M.
The ionic strength of the solution needs to be kept constant. At high activities of other ions there can be large variations in the potential difference:
· OH- ions need to be avoided.
· pH < 3 causes HF formation and should be avoided.
· Al(III) and Fe (III) complex strongly to F-.
Then using TISAB flouide electrodes have a nerstien response from 10-5 to 1 M.
The ionic strength of the solution needs to be kept constant. At high activities of other ions there can be large variations in the potential difference:
This is why TISAB is used as a buffer.
Polymer electrodes
The ion selective membrane is a thin ionexchange PVC film. This is flexible and robust.
Polymer electrodes
The ion selective membrane is a thin ionexchange PVC film. This is flexible and robust.
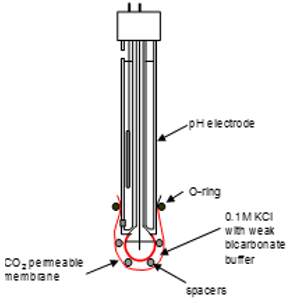
Gas sensing electrodes
These exploit ISEs to allow acid base chemistry to detect gases. For the CO2 selective electrode:
These exploit ISEs to allow acid base chemistry to detect gases. For the CO2 selective electrode:
There is a pH electrode standardising the equilibrium in water:
Reference electrodes
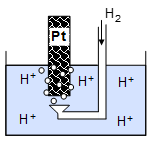
Standard hydrogen electrode is the most commonly known although it is hardly used due to the lack of portability of the electrode and the accessibility of hydrogen.
Here:
Standard hydrogen electrode is the most commonly known although it is hardly used due to the lack of portability of the electrode and the accessibility of hydrogen.
Here:
It is catalysed using platinum and the reaction is always in equilibrium.
The Ag/ AgCl reference electrode involved the reaction with silver and chloride ions in a saturated potassium chloride solution. The wire is coated with AgCl which provides Ag+ ions.
The solubility product Ks = aAg+ + acl- to keep the activity of these constant the potential of the reference electrode at a defined value the Ag/AgCl wire is kept immersed in a saturated KCl solution.
This is a cheap and robust electrode to use.
This is a cheap and robust electrode to use.
Amperometric sensors
Definition: Electrochemical devices that allow the measurement of the concentration of a selected redox active species througmeasure of flow of current due to oxidation or reduction of species.
Applications There are many applications such as dissolved oxygen, glucose and neurotransmitters.
Analytical advantages: Same as the ISE but the analyte concentration is detected at the electrode surface so can allow many physiological uses.
Electron transfer:
Applications There are many applications such as dissolved oxygen, glucose and neurotransmitters.
Analytical advantages: Same as the ISE but the analyte concentration is detected at the electrode surface so can allow many physiological uses.
Electron transfer:
A- Is being oxidised here as it loses and electron. B is being reduced as it is gaining an electron.
The above is an example of a homogenous reaction this can occur on a solid surface and is therefore a heterogenous reaction.
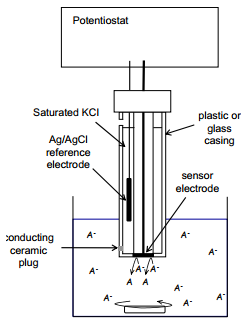
Design of amperometric sensors
The potentiostat is a device that controls the potential applied to the sensor electrode relative to the reference electrode. It also measures the current flow between the reference and sensor electrode due to oxidation of reduction of the analyte.
The above is an example of a homogenous reaction this can occur on a solid surface and is therefore a heterogenous reaction.
Design of amperometric sensors
The potentiostat is a device that controls the potential applied to the sensor electrode relative to the reference electrode. It also measures the current flow between the reference and sensor electrode due to oxidation of reduction of the analyte.
Heterogenous redox reactions only occur at the surface which means that the magnitude of the current flow is only proportional to the analyte at the surface.
The Clarke electrode
This is widely used for the measurement of dissolved here is reduced.
The Clarke electrode
This is widely used for the measurement of dissolved here is reduced.
The selectivity is maintained by using a PTFE cover on the sensor electrode which only allows oxygen through.
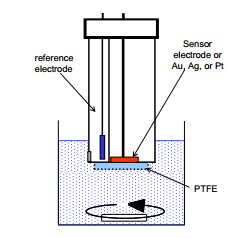
The ferrocene based glucose biosensor
This is the primary tool used for routine analysis of glucose.
· Graphite electrode enhanced with ferrocene.
· Ag/AgCl reference.
· Glucose is oxidised to form glucolacton.
This is the primary tool used for routine analysis of glucose.
· Graphite electrode enhanced with ferrocene.
· Ag/AgCl reference.
· Glucose is oxidised to form glucolacton.
This gives a linear response between 5 and 12 mM a physiologically important range.
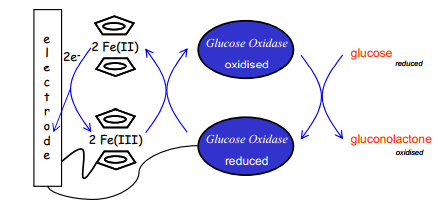
Electron flow
It is important to remember in which direction electron flow takes place. The movement of electrons from a sensor electrode causes a positive current. Away from the electrode a negative current:
Electron flow
It is important to remember in which direction electron flow takes place. The movement of electrons from a sensor electrode causes a positive current. Away from the electrode a negative current:
Key uses and features
· Flow of current directly related to analyte.
· Redox chemistry causes current flow.
· Concentration of analyte is lowered by the measurement. It has to be stirred.
· Analyte can be an ion or have no charge, only redox ability is needed although it needs to be able to be oxidised or reduced quickly.
In principle a molecule that reaches the electrode should react although:
· Physical exclusion can stop analytes reaching the sensor electrode.
· Selectivity can be obtained by enzyme use.
· Electrostatic repulsion of other analytes from the sensor electrode can occur.
What potential should be used?
The reactions effectiveness is purely based on the potential difference used.
· Flow of current directly related to analyte.
· Redox chemistry causes current flow.
· Concentration of analyte is lowered by the measurement. It has to be stirred.
· Analyte can be an ion or have no charge, only redox ability is needed although it needs to be able to be oxidised or reduced quickly.
In principle a molecule that reaches the electrode should react although:
· Physical exclusion can stop analytes reaching the sensor electrode.
· Selectivity can be obtained by enzyme use.
· Electrostatic repulsion of other analytes from the sensor electrode can occur.
What potential should be used?
The reactions effectiveness is purely based on the potential difference used.
Where [O] is concentration of oxidised substance and [R] is concentration of reduced substance.
When the concentration of one of these compounds is known then the following relationships can be used:
When the concentration of one of these compounds is known then the following relationships can be used:
This means the concentration of R can be given as a sample percentage:
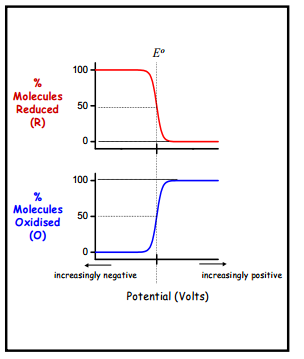
The curvature seen here is related to the different selectivities, at high reduced concentration there is a large amount of un-reacted species but the electrode potential is not yet positive enough to rapidly cause oxidation this means that kHET the rate constant is the limiting factor. At the most positive potentials all of the molecules are now oxidising rapidly which causes the diffusion constant to take more of an effect which then becomes the limiting factor.
Current and electrodes reactions
· 1 Amp= passage of 1 coloumb of charge per second.
· The faraday constant = 96484 colombs of charge per mole of electrons.
Therefore 1 Amp = 1x10-5 electrons.
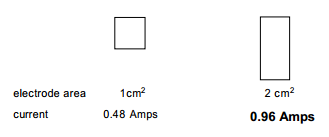
The electrode area is directly related to the current:
· 1 Amp= passage of 1 coloumb of charge per second.
· The faraday constant = 96484 colombs of charge per mole of electrons.
Therefore 1 Amp = 1x10-5 electrons.
The electrode area is directly related to the current:
So the rate of electrode reaction can be represented as:
This is related to the concentration of the analyte by:
Combing the two definitons of electron transfer this can form:
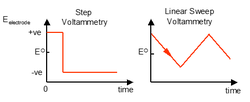
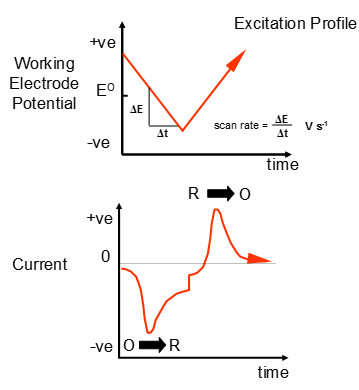
Voltammetry
Voltammetry works by using a defined but variable potential to the sensor electrode. Thie means that the redox reactions can occur directly on the electrodes surface at any point when the electrode potential has been set to a value that would allow this. To make best use of this ability the following need to be considered:
· The reduction potentials and electron stoichiometry of redox reactions.
· The reversibility of the redox reactions undergone by a moleulce.
· Diffusion coefficients of species in stable oxidation states.
This allows the production of an excitation profile:
Voltammetry works by using a defined but variable potential to the sensor electrode. Thie means that the redox reactions can occur directly on the electrodes surface at any point when the electrode potential has been set to a value that would allow this. To make best use of this ability the following need to be considered:
· The reduction potentials and electron stoichiometry of redox reactions.
· The reversibility of the redox reactions undergone by a moleulce.
· Diffusion coefficients of species in stable oxidation states.
This allows the production of an excitation profile:
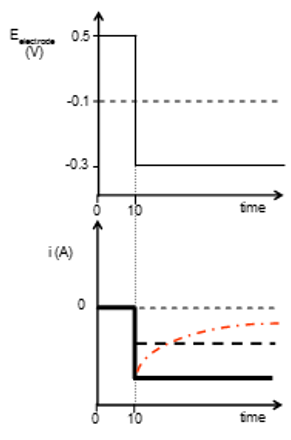
Step voltammetry
A 5 mM concentration of Oxidised material O, in an experiment is held with an electrode potential of 0.5 V this is instantaneously switched to -0.3 V. This causes the molecule to become reduced. The current measured becomes negative rapidly as electrons are leaving the potential surface. In a stirred material this current remains the same as there is rapid replacement of reduced material with oxidised material but unstirred this slowly approachs zero again. At ½ concentration there is ½ the current.
A 5 mM concentration of Oxidised material O, in an experiment is held with an electrode potential of 0.5 V this is instantaneously switched to -0.3 V. This causes the molecule to become reduced. The current measured becomes negative rapidly as electrons are leaving the potential surface. In a stirred material this current remains the same as there is rapid replacement of reduced material with oxidised material but unstirred this slowly approachs zero again. At ½ concentration there is ½ the current.
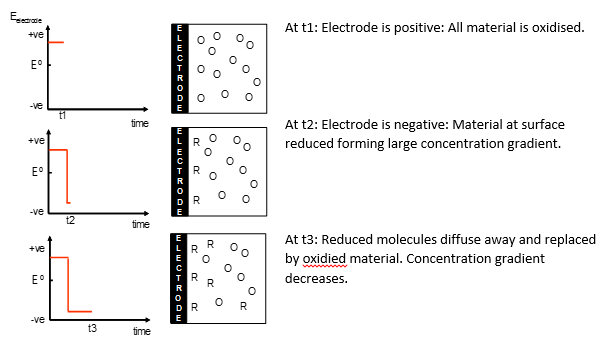
The elementary steps at the electrode
As the oxidised material at the electrode becomes reduced (or vice-versa) an diffusion gradient is produced drawing more oxidised material. This means that slowly the oxidised material is all reduced although the rate at which this occurs decreases over time.
As the oxidised material at the electrode becomes reduced (or vice-versa) an diffusion gradient is produced drawing more oxidised material. This means that slowly the oxidised material is all reduced although the rate at which this occurs decreases over time.
This can be better represented in a graph showing the concentration of the materials at different distances from the electrode.
This leads to the Cottrell equation showing how the current is related to the concentration. This is where n is the charge of the ion, and is the diffusion coefficient.
Accounting for the potential dependence of the current
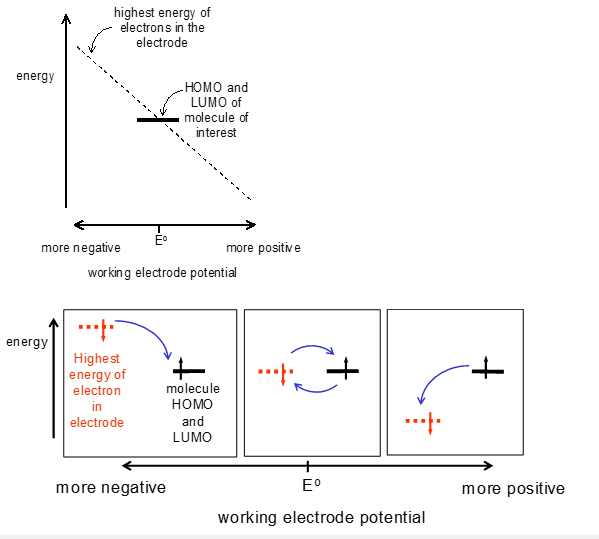
The potential dependence on the current can be explained using molecular orbital theory. By moving the electrode potential the HOMO and LUMO of the orbtials in the electrode are moved. If this is moved to a large positive value the electrons are brought very low in energy which causes the analyte to lose its electrons to the surface, at very negative values the electrons are high in energy and are lost to the analyte.
The potential dependence on the current can be explained using molecular orbital theory. By moving the electrode potential the HOMO and LUMO of the orbtials in the electrode are moved. If this is moved to a large positive value the electrons are brought very low in energy which causes the analyte to lose its electrons to the surface, at very negative values the electrons are high in energy and are lost to the analyte.
The Gibbs free energy of a material is shown as being related to the electrode potential as, like all reactions, there is an activation barrier for this reaction. This can be seen in the following equations and graphs:
Linear sweep (Cyclic) Voltammetry
Linear sweep voltammetry can give information on different oxidation states of the compound. The scan rate can be easily controlled and has the units V s-1.
The current-plot recorded can be difficult to understand although it follows all the same principles of the older current plots.
Linear sweep voltammetry can give information on different oxidation states of the compound. The scan rate can be easily controlled and has the units V s-1.
The current-plot recorded can be difficult to understand although it follows all the same principles of the older current plots.
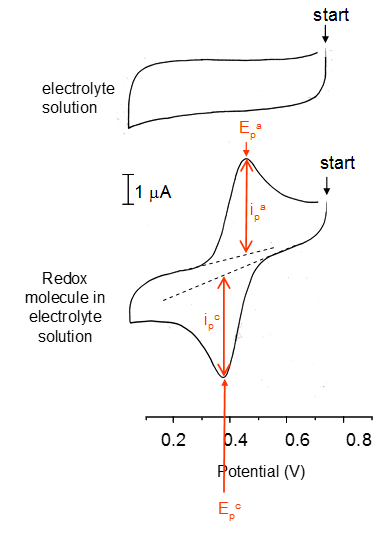
The current here can be seen as becoming more negative as the working electrode reduces the ions present. This then hits a maximum when the concentration gradient is highest meaning that the rate of electron donation is highest. This then begins to decrease as this now follows Cottrell behaviour as the majority of ions are now reduced. There is a step in current at the change in voltage. The exact process then occurs again but in the positive direction.
The peaks of a current potential plot will centre at the electrode potential of the molecule.
The peaks of a current potential plot will centre at the electrode potential of the molecule.
The reversibility is defined by the peaks i_p^a and i_c^p when the reaction is fully reversible everything that is oxidised is also reduced then:
The reduction potential is the average of the peak potentials
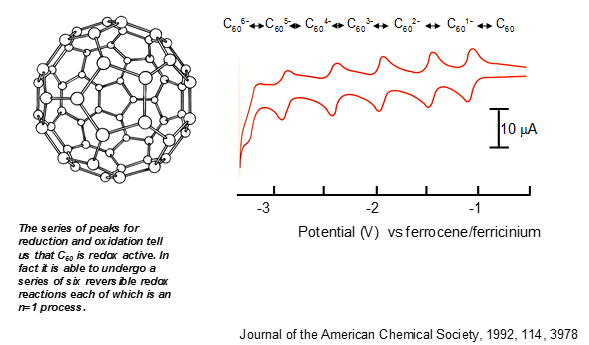
Buckminsterfullerene
There are a number of peaks present through the analysis of Buckminsterfullerene
There are a number of peaks present through the analysis of Buckminsterfullerene
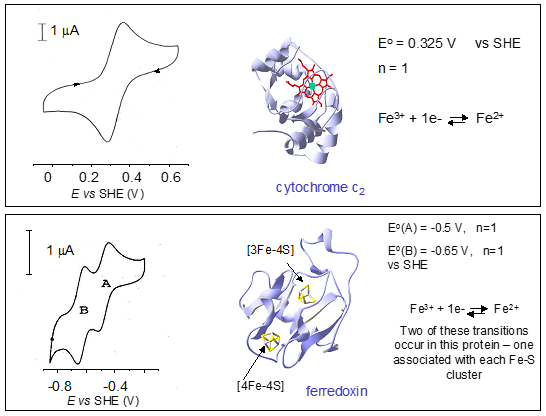
Redox chemistry in proteins with Fe Cofactors:
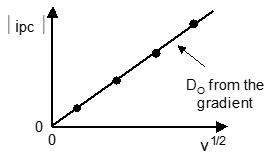
Variation of scan rate
Variation of the scan rate provides information of the diffusion of the reactants.
This is where ν is the scan rate this is useful for calculating the effect of diffusion on the reaction.
Variation of the scan rate provides information of the diffusion of the reactants.
This is where ν is the scan rate this is useful for calculating the effect of diffusion on the reaction.
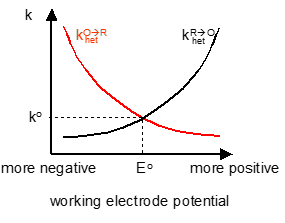
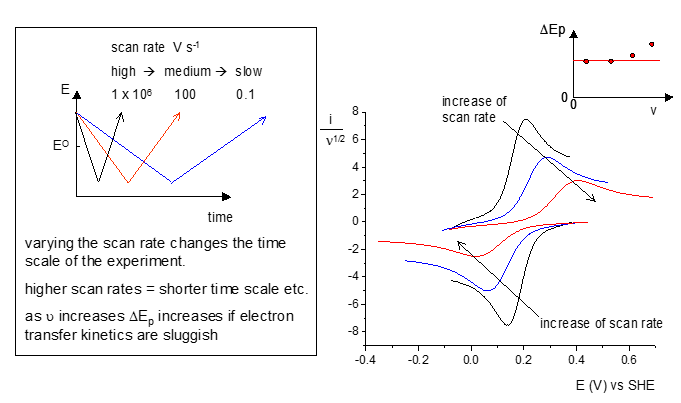
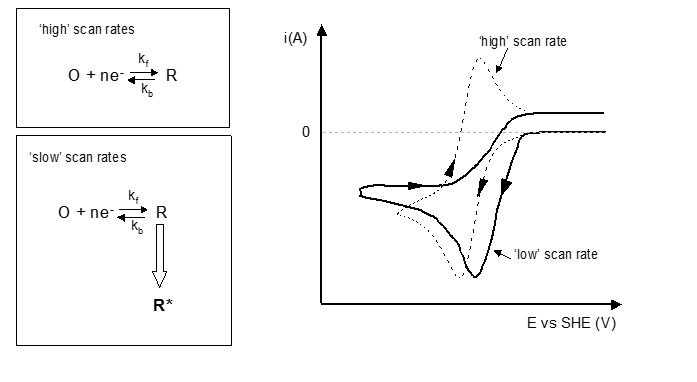
Electron transfer kinetics
There is also a link between this and the current against potential with the scan rate being varied. At increasing scan rates there is less resolution of the peaks produced. This means that there is an important equilibrium that needs to be considered when scan rates are considered where electron transfer is slow.
There is also a link between this and the current against potential with the scan rate being varied. At increasing scan rates there is less resolution of the peaks produced. This means that there is an important equilibrium that needs to be considered when scan rates are considered where electron transfer is slow.