The art of designing a synthesis route is tested heavily in the organic exam. There are many different ways to design a synthetic route, one of the most common being retrosynthesis.
Retrosynthesis
The art of retrosynthesis itself is carried out by looking at the final compound and working backwards from it. It obviously helps having a large library of reaction mechanisms to follow back on.
Synthons
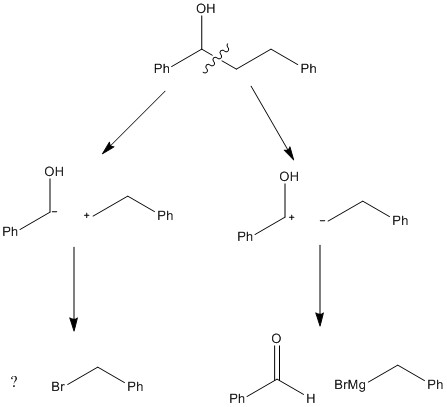
The idea of synthons is breaking the compound into varying ions and working backwards forming ionic "reactants". The splitting of the each bond forming a positive and a negative charge can give a very effective prediction of how each bond formed.
For an effective planning of the synthesis of a target molecule there needs to be:
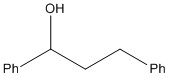
Taking into account the molecule below:
The art of retrosynthesis itself is carried out by looking at the final compound and working backwards from it. It obviously helps having a large library of reaction mechanisms to follow back on.
Synthons
The idea of synthons is breaking the compound into varying ions and working backwards forming ionic "reactants". The splitting of the each bond forming a positive and a negative charge can give a very effective prediction of how each bond formed.
For an effective planning of the synthesis of a target molecule there needs to be:
- Recognition of the functional groups in the target molecule.
- Awareness of how you could make these functional groups.
- Recognition of a potential carbon-carbon bond forming step.
Taking into account the molecule below:
The target molecule below can be seen to have one main functional group, the alcohol group. The phenyl groups do not need to be considered in this reaction as they cannot be "produced" easily, they have a further effect that is explained later on.
The ionic species here are synthons. These show that the bond can break forming a cation and an anion. The alcohol group can be produced through a nucleophilic attack on a carbonyl and an alkyl chain can behave as a nucleophile when it is present as a grignard reagent.