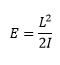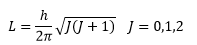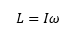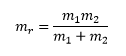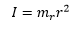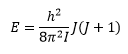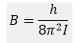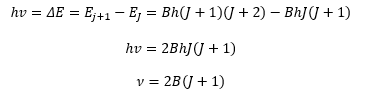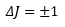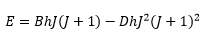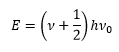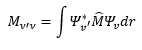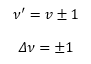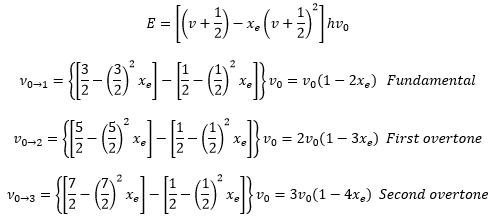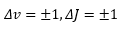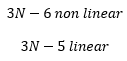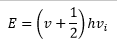Rotational spectroscopy
For a rotational spectrum to be taken microwave radiation is used. This measures the energy absorbed by the rotations of the molecule. The angular momentum depends on the mass of the atoms in the molecule and the rotational quantum number of the molecule. The energy differences in exciting a molecule from one rotational state to another can be mapped and analysed.
|
Rotational energy
The rotational energy can be represented by the quantum mechanical factor L and the inertia I. |
Rotational quantum number
The rotational quantum number is a discrete value that is achieved by solving the Schrodinger equation. This can give the value for L |
Inertia
The inertia of the molecule is given as the relationship between the reduced mass and the radius of the molecule. |
These values can be used to understand a rational spectrum and why the relations between these values and the energy levels.
|
Energy levels in a spectrum
Substituting L for the quantum mechanical values gives: The parts of this equation that remain constant can be changed into a single value B:
This means then a molecule absorbs radiation it moves from a lower energy state to a higher energy state:
|
This allows a value of B to be found as the spacing between lines is given as 2B. This means the inertia of the molecule can be found.
These transitions only occur if the following selectrion rule has been maintained: This then leaves the centrifugal distortion effect that might occur due to the molecule spinning. This is more likely to occur at high values of J as this is when the molecule is spinning fastest. This causes the molecule to be more drawn out meaning centrifugal forces take more of an effect. This can be represented by the equation:
Another important point is the use of isotopic substitution to look at one bond in particular. The slight differences in bond energy can allow a greater measure of the bond being taken into account.
|
Vibrational spectroscopy
|
The relationship between the molecule and the vibration frequency is related to the restoring force of the molecule, this is determined by the bond length and therefore strength.
|
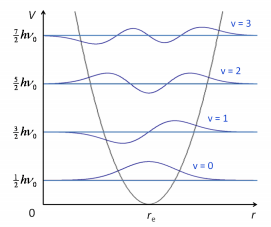
The emergy levels can be mapped by simple harmonic motion diagram as such:
|
This shows how the energy levels are discrete values which can be mapped by the Schrodinger equation:
Which gives the first selection rule:
|
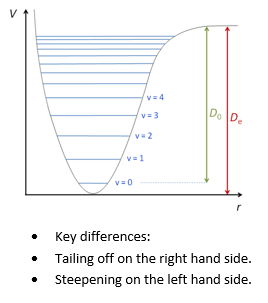
This does leave an issue though, the simple harmonic oscillator doesn't take into account that molecules can repulse when brought too close to one another and dissociate when at high energy an separation.
|
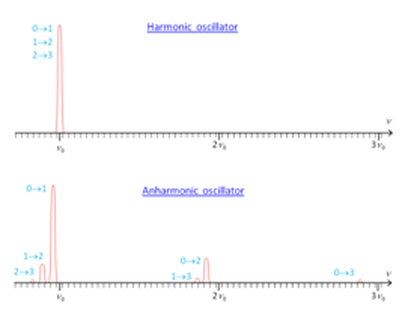
The levels in the anharmonic quantum oscillator are therefore not equally spaced which is seen in the comparison between the two:
|
This shows the presence of hot and overtone bands. Hot bands occur from an already excited state into another state and overtone are when there is a doubly excited movement,
|
These equations show that it is important to have both a measure of the fundamental and the overtone bands so the fundamental vibration constant can be calculated v0, this allows the the anharmonicity constant to be calculated.
|
Rotational and vibrational spectra
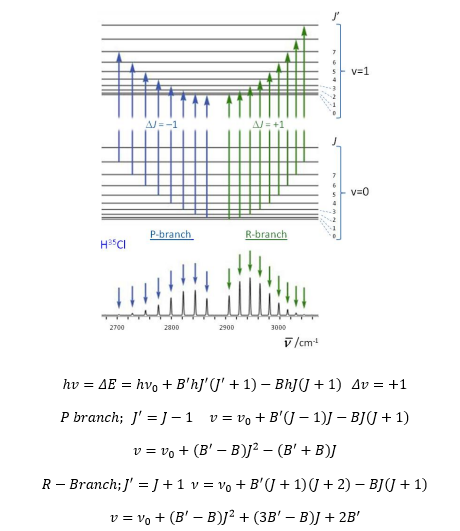
These spectra allow highly detailed analysis of a molecule. The following selection rule applies though:
These spectra allow highly detailed analysis of a molecule. The following selection rule applies though:
These can be represented by R and P branches:
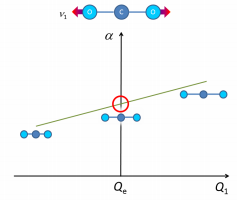
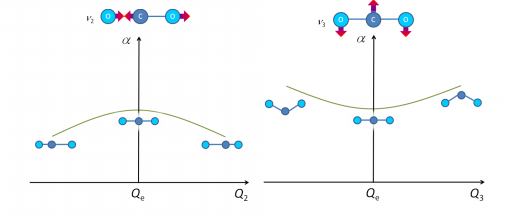
Vibrational specra of polyatomic molecules
Raman spectroscopy
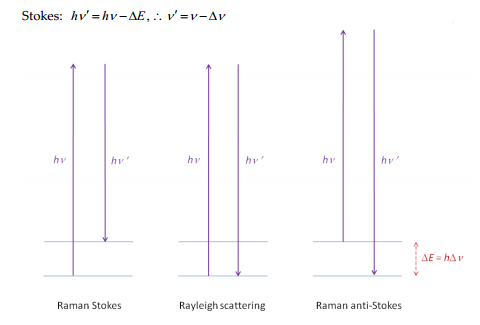
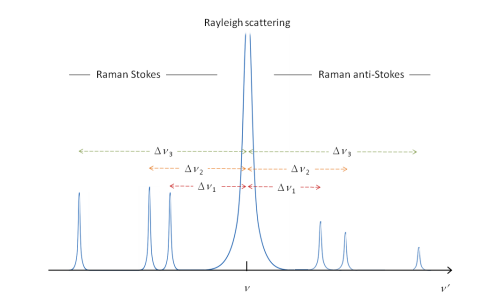
The basis of Raman spectroscopy is the difference in energy caused by the scattering of light by a molecule. This measure a change in polarisation, forming Stokes and anti-Stokes lines as well as the un-changed Rayleigh scattering.
The difference in starting energy means that Raman anti-Stoke lines decrease in intensity.
If the polarisability changes in the course of a given vibration, as the molecule passes through its equilibrium configuration that vibration will be Raman active.