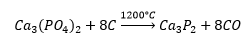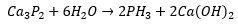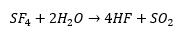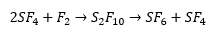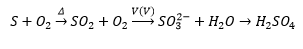Main group elements:- Summary, revision and questions.
Is not hard to realise there is a tonne of stuff discussed for the main groups elements. The other lecture note pages show quite how much there is. (<15000 words). This is a summary, if you have deeper questions refer to the other pages but those pages are too clunky and big to use for revision purposes. So here is a quick review. Also the lists are not exhaustive. Just some quick understanding points.
Group 13
|
Boron
Boron oxides
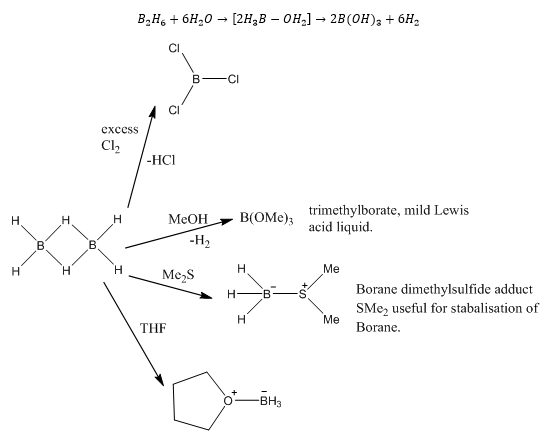
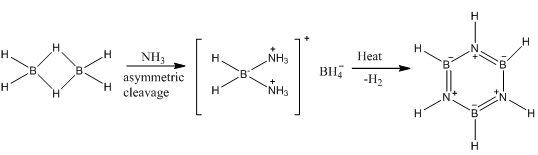
Boron hydrides
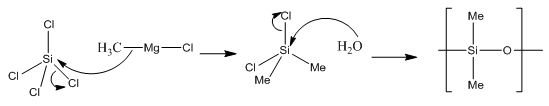
Boron halides
Boron nitrogen compounds
Alkyl boranes
Remember: Borane acts as a Lewis acid, it accepts electrons so in the precence of any Lewis bases it is very likely it will form an adduct. It is also easily oxidised forming hydrogen of HCl. |
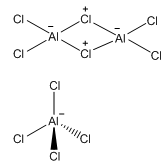
Aluminum
Aluminium complex
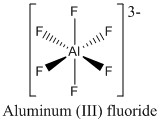
This is important to remember that aluminium fluoride can act as a Lewis acid further:
This occurs because aluminium is a big enough atom to allow an expanded octet allowing a hexavalent structure to form.
Remember: Aluminium holds many similar properties to boron but can utilize an expanded octet allowing larger ions to form. 6e- species of aluminium are strong Lewis acids.
|
Group 14
|
Carbon
Has strong formation of double bonds and does not have a very high affinity for oxides compared to silicon. Silicon
|
Pyroxenes and other chain silicate sheets
Tin
Lead
Remember: Carbon can form double bonds and does not favour formation or the oxide as much as silicon. Formation of silicates through hydrolysis of negative oxide groups is highly effective. Silicate structure can form many different sheets which can be used for cation capture and exchange.
|
Group 15
|
Nitrogen
Phosphorus
The formation of phosphines can occur through a metallic phosphine group:
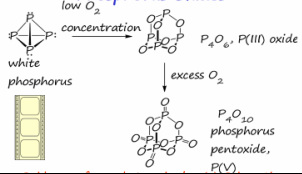
Phosphours oxides are produced by reacting different oxygen concentrations with the phosphorus group:
|
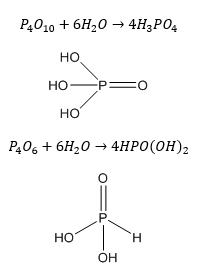
This can then be hydrolysed forming different phosphorus acids.
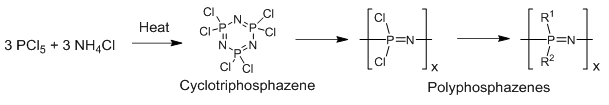
Phosphorus has the ability to react with nitrogen to form a phosphazenes.
Phosphorus halides
PIII
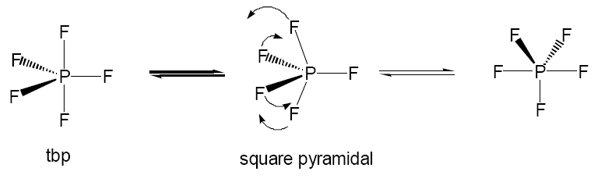
PV PF5 is a gas with boiling point -84.5°C, it maintains a trigonal bipyrimadal structure. Which can undergo Berry rotation. There is an importance of this rotation as the NMR shows only one electron environment due to the usual two. |
Group 16
Sulphur
Sulphur halides
Sulphur will react with most halogens, this can be hydrolysed form sulphide. Sulfur hexafluoride will will react with flourine and then disproportionate.
|
Oxides of sulphur
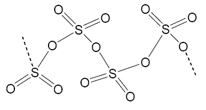
The reactions of sulphur with oxygen produces sulphur dioxide which can be reacted with oxygen with a vanadium catalyst and then hydrolysed forms sulphuric acid. Polymeric ice like formation can occur between SO4 molecules with the loss of water. This only occurs at low temperatures <17C
|
Group 17
|
Noble Gases
|