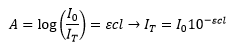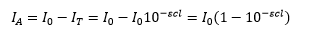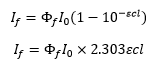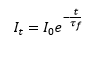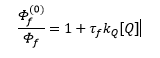Fluorometer
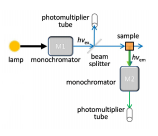
The main differences between a UV-Vis spectrometer and a fluorometer are the fact that there are two monochromators in a fluorometer and the detector is placed at a right angle to the sample.
The reason for there being two monochromators is that the wavelength of the excitation beam is measured by wavelength and the fluorescence is then measured. This has to pass through another monochromator as multiple wavelengths of light can be fluoresced.
Detectors for fluorescence need to be very sensitive so PMT is often used as this amplifies the result resulting in this being a very useful detector to use for the small changes in value of intensity.
The reason for there being two monochromators is that the wavelength of the excitation beam is measured by wavelength and the fluorescence is then measured. This has to pass through another monochromator as multiple wavelengths of light can be fluoresced.
Detectors for fluorescence need to be very sensitive so PMT is often used as this amplifies the result resulting in this being a very useful detector to use for the small changes in value of intensity.
Process
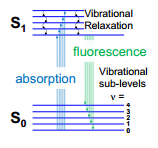
Fluroescence spectroscopy, processes can be explained on a Jablonski diagram. This shows how the electronic states are split into vibrational energy levels. When the sample is excited it move to a higher electronic energy state, this relaxes down the vibrational states releasing energy as temperature.
Once the excited electronic state has the minimum amount of vibrational energy the only way it can re-enter the ground state is to emit energy in the form of a photon. This is fluorescence.
The difference in wavelength between the light absorbed and the light emitted is caused by the difference in energy to the loss of energy as temperature. The excitation spectrum of a molecule is the energy absorbed by the molecule that leads to fluorescence, this is carried out by fixing the emission monochromator and scanning through with the excitation monochromator. This means that there is a measure of the absorption. This is identical to the absorption spectrum as the fluorescence intensity is proportional to the absorption.
The multiple transitions of excitation and emission contribute width to both the absorption and emission bands. This can be seen in the vibrational energy levels and the fact that there are multiple rotational and vibrational energy levels.
Fluorescence requires an initial absorption of light. This can be represented by the amount of light transmitted and the initial intensity of light.
The difference in wavelength between the light absorbed and the light emitted is caused by the difference in energy to the loss of energy as temperature. The excitation spectrum of a molecule is the energy absorbed by the molecule that leads to fluorescence, this is carried out by fixing the emission monochromator and scanning through with the excitation monochromator. This means that there is a measure of the absorption. This is identical to the absorption spectrum as the fluorescence intensity is proportional to the absorption.
The multiple transitions of excitation and emission contribute width to both the absorption and emission bands. This can be seen in the vibrational energy levels and the fact that there are multiple rotational and vibrational energy levels.
Fluorescence requires an initial absorption of light. This can be represented by the amount of light transmitted and the initial intensity of light.
This is simply the Beer-Lambert law. The absorbed light is the difference between these two so:
The proportion of this absorbed light that leads to fluorescence is defined as the fluorescence quantum yield.
The quantum yield shows that the fluorescence is proportional to the initial intensity I0 and the concentration. The issue with this is at high concentrations there can be self-absorption affects and the collisions between particles causing a loss in energy through means other than fluorescence.
This is important as it shows how the energetic state does not instantaneously cause fluorescence but rather has a lift time. If anything occurs in this life time to the molecule that can cause it to lose its energy it will not fluoresce.
This is important as it shows how the energetic state does not instantaneously cause fluorescence but rather has a lift time. If anything occurs in this life time to the molecule that can cause it to lose its energy it will not fluoresce.
Because of the importance that fluorescence quantum yield can be achieved at low concentrations it is surprising that fluorescence is still very sensitive. This is due to the fact that there are low light levels against a dark background.
Quenching
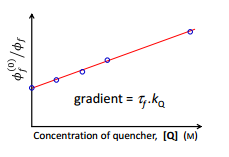
A quencher is a molecule that can collide or interact with the excited state of another molecule but does not fluoresce itself. This causes the amount of fluorescence to decrease. This related by the Stern-Volmer equation. This is where the initial quenching φ0f can be compared to the quantum yield with the addition of quencher.
This can be plotted on the Sterm Volmer plot:
So as the concentration of the quencher increases the quantum yield of the quenched solution decreases.
Forster resonance energy transfer
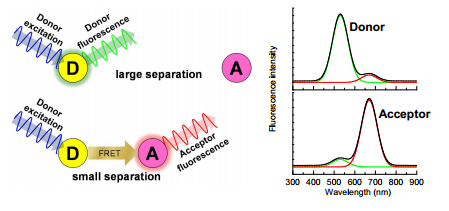
Forster resonance energy transfer is awhere a excited molecule releases its energy to another molecule through a dipole interaction (there is no emission/absorption occuring here!) this causes a low in energy again which forces the wavelength of the radiation down further. This occurs in chlorophyll and other natural systems.
Forster resonance energy transfer
Forster resonance energy transfer is awhere a excited molecule releases its energy to another molecule through a dipole interaction (there is no emission/absorption occuring here!) this causes a low in energy again which forces the wavelength of the radiation down further. This occurs in chlorophyll and other natural systems.
Cuvette types
Cuvettes need to have perfectly flat, parallel windows which must be positioned so they are perpendicular to the beam of radiation. Absorption cell windows only need to have two optically transparent sides. This is because a single straight beam of light passes through the cell cuvette. In fluorescence spectroscopy it is important that all sides are transparent to allow the radiation to be emitted in all directions.
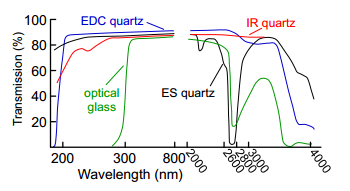
EDC quartz
is a very effective cuvette type, as it can be used in the UV range.