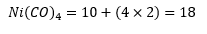Electron counting
Electron counting is one of the most important aspects of organometallic chemistry. The importance of electron counting is based on the 18 electron rule:
Electron counting is a very important part of the court that is purely based on finding the number of electrons that surround a metal centre.
For a long time there has been a large amount growth in the d-block chemistry. There are more new types of reaction unusual structures and many types of organic synthesis.
The idea that many bonds are very strong and hard to break is well known in chemistry. Organometallics are highly important as they can allow many different structures to form allowing a number of different reactions to be allowed to take place. The importance of allowing different metal structures and how to form these different structures has a large basis in how ligands bond to the surface of metals.
Tranisition mediated reactions used in organic synthesis is where the money lies.
Electron counting is a very important part of the court that is purely based on finding the number of electrons that surround a metal centre.
For a long time there has been a large amount growth in the d-block chemistry. There are more new types of reaction unusual structures and many types of organic synthesis.
The idea that many bonds are very strong and hard to break is well known in chemistry. Organometallics are highly important as they can allow many different structures to form allowing a number of different reactions to be allowed to take place. The importance of allowing different metal structures and how to form these different structures has a large basis in how ligands bond to the surface of metals.
Tranisition mediated reactions used in organic synthesis is where the money lies.
Theory
The d orbital
A transition metal is one which contains a partially filled d-orbital scandium and zinc are therefore no transition metals as they have partially filled d orbitals. These d-orbitals are highly directional either being occupied or un-occupied.
Transition metals are important due to the use of the d-orbital. This is a well-studied property and is of a large interest in inorganic chemist. These metal orbitals can hybridise leading to a variety of coordination numbers. Although the coordination numbers ranging from 4-6 are more regularly seen all coordination are possible up to 8. It is important to remember that most coordination’s have a typical geometry although other geometries can be seen.
This also leads to the ability to hybridise these d orbitals with other orbitals forming different shapes. This means that any coordination geometry can be accessible through transition metals.
These orbitals can accept electron density from the ligand making it less electron rich and therefore more likely to react with a nucleophile. A ligand can also coordinate with electron dense transition metal by adding a number of electron dense ligands to that metal a process called back bonding can occur. This is where electrons are forced into the ligand making it more likely to react as a nucleophile.
The d orbital can interact with the π and π* orbitals of unsaturated organic molecules. This is the basis of the movement of electrons. The changing of reactivity patterns caused by this is the basis of catalysis.
Octet rule
The octet is where there a sufficient number of electrons that allows the filling of the orbitals and therefore the stability of the atom. The stability of the molecules can be predicted by looking at the octet rule. It is important to remember the different ways that the octet rules can be allowed to form. The 18-electron rule
The 18-eletron rule is a stable complex with the valence electron count of the next highest noble gas is obtained when the sum of the metal d-electrons (already present in the atom) and the electrons donated from the ligands and of the overall charge on the complex equals 18.
So:
· Add electrons on metal
· With electrons from ligands
· With overall charge
· Equals 18.
This is the octet rule for transition metals. In a majority of cases this rule is found and allows the complex to be found by working backwards while using this rule. All carbonyl complex’s follow this rule allowing a very easy analysis of structure. It is true that most organometallic compounds do actually follow these rules. This is not true for hydrated species etc.
There are a few exceptions that need to be learnt.
The number of d-electrons in a chemical environment is the group number. Easy as that. Again:
· Group number.
· Electrons from ligands.
· With overall charge.
· Equals 18.
It is not necessary to form oxidation states in this method. It is purely homolysis bond breaking giving neutral radicals.
The zerovalent convention is usually used. So long as one convention is rigidly stuck to then the right answer will be found. This is important.
So why does the 18 electron rule work?
This draws a lot of information back from last years material on the filling of orbitals in a bonding structure. There are three different magnitudes of ligand field splitting possible which are described by three complex classes.
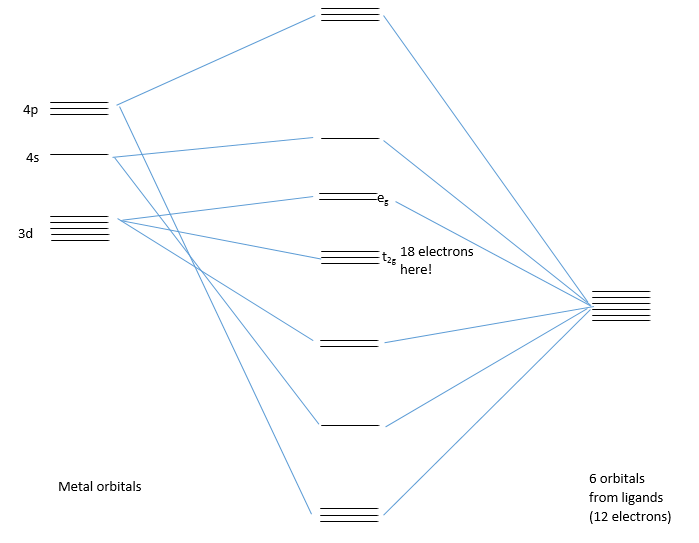
Firstly the ligand field splitting diagram needs to be understood:
A transition metal is one which contains a partially filled d-orbital scandium and zinc are therefore no transition metals as they have partially filled d orbitals. These d-orbitals are highly directional either being occupied or un-occupied.
Transition metals are important due to the use of the d-orbital. This is a well-studied property and is of a large interest in inorganic chemist. These metal orbitals can hybridise leading to a variety of coordination numbers. Although the coordination numbers ranging from 4-6 are more regularly seen all coordination are possible up to 8. It is important to remember that most coordination’s have a typical geometry although other geometries can be seen.
This also leads to the ability to hybridise these d orbitals with other orbitals forming different shapes. This means that any coordination geometry can be accessible through transition metals.
These orbitals can accept electron density from the ligand making it less electron rich and therefore more likely to react with a nucleophile. A ligand can also coordinate with electron dense transition metal by adding a number of electron dense ligands to that metal a process called back bonding can occur. This is where electrons are forced into the ligand making it more likely to react as a nucleophile.
The d orbital can interact with the π and π* orbitals of unsaturated organic molecules. This is the basis of the movement of electrons. The changing of reactivity patterns caused by this is the basis of catalysis.
Octet rule
The octet is where there a sufficient number of electrons that allows the filling of the orbitals and therefore the stability of the atom. The stability of the molecules can be predicted by looking at the octet rule. It is important to remember the different ways that the octet rules can be allowed to form. The 18-electron rule
The 18-eletron rule is a stable complex with the valence electron count of the next highest noble gas is obtained when the sum of the metal d-electrons (already present in the atom) and the electrons donated from the ligands and of the overall charge on the complex equals 18.
So:
· Add electrons on metal
· With electrons from ligands
· With overall charge
· Equals 18.
This is the octet rule for transition metals. In a majority of cases this rule is found and allows the complex to be found by working backwards while using this rule. All carbonyl complex’s follow this rule allowing a very easy analysis of structure. It is true that most organometallic compounds do actually follow these rules. This is not true for hydrated species etc.
There are a few exceptions that need to be learnt.
The number of d-electrons in a chemical environment is the group number. Easy as that. Again:
· Group number.
· Electrons from ligands.
· With overall charge.
· Equals 18.
It is not necessary to form oxidation states in this method. It is purely homolysis bond breaking giving neutral radicals.
The zerovalent convention is usually used. So long as one convention is rigidly stuck to then the right answer will be found. This is important.
So why does the 18 electron rule work?
This draws a lot of information back from last years material on the filling of orbitals in a bonding structure. There are three different magnitudes of ligand field splitting possible which are described by three complex classes.
Firstly the ligand field splitting diagram needs to be understood:
This shows how there are 18 electrons once the t2g orbitals have been filled. So why is this d orbital splitting important? And why doesn't the 18 electron rule matter for all transition metal structures.
Class I complexes
Class I complexes have low energy splitting ligands. This means that the eg orbital becomes only slightly antibonding and the t2g orbital is almost completely non-bonding still. This means there is little energy difference on whether these orbitals are filled or not. This means that it is common for these complexes do have much below or much above 18 electrons, meaning they don't obey the 18 electron rule.
Class II complexes
These usually have a large transition metal center with ligands high in the spectrochemical series. The eg tends to be fairly antibonding but the t2g remains non-bonding this means that some of these complexes will follow the 18 electron rule but there is variation.
Class III complexes
These complexes tend to be first row transition metals with ligands high on the spectrochemical series. This causes a large amount of splitting between the eg and the t2g orbital. This means that the eg if highly antibonding and the t2g is highly bonding causing the filling of the t2g to be favoured and the filling of the eg to be highly unfavoured.
Class I complexes
Class I complexes have low energy splitting ligands. This means that the eg orbital becomes only slightly antibonding and the t2g orbital is almost completely non-bonding still. This means there is little energy difference on whether these orbitals are filled or not. This means that it is common for these complexes do have much below or much above 18 electrons, meaning they don't obey the 18 electron rule.
Class II complexes
These usually have a large transition metal center with ligands high in the spectrochemical series. The eg tends to be fairly antibonding but the t2g remains non-bonding this means that some of these complexes will follow the 18 electron rule but there is variation.
Class III complexes
These complexes tend to be first row transition metals with ligands high on the spectrochemical series. This causes a large amount of splitting between the eg and the t2g orbital. This means that the eg if highly antibonding and the t2g is highly bonding causing the filling of the t2g to be favoured and the filling of the eg to be highly unfavoured.
Electron counting
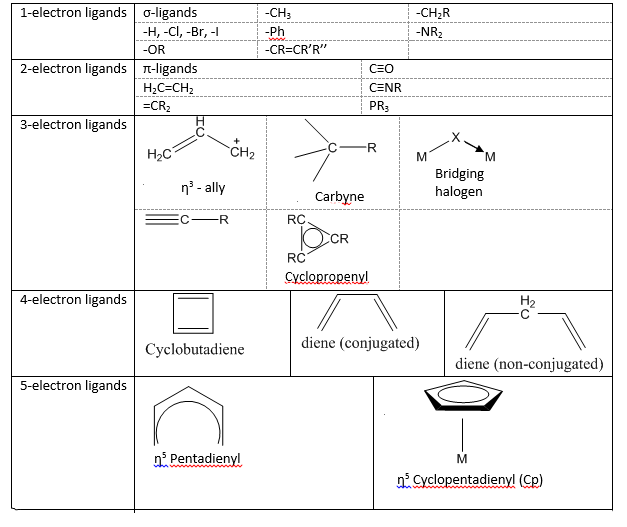
Each Ligand can "donate" a different number of electrons.
This shows the large variation in the possible ligands that can bond to a transition metal.
2-electron donators are seen in two different ways. A dative donor is seen as 2-electrons being given. As there are no electron coming from the metal. π ligands meaning any organic molecule with a π bond will donate in the same way.
Bridging halides are strange in which 3 electrons are given but to two different metal centres.
Ligands
For a long time it has been known that an electron donor can bind to a metal centre through shared electron density. The first ligands that were used were usually lone pair donors that could easily bond in a predictable manor with a metal centre. More commonly now is the use of high electron density species which have a large amount of electron density around them. Ligands such as Ferrocene and alkenes were important and were and still are used regularly in Ziegler Natta catalysts etc. Transition metal chemists draw the bond going from the metal to the middle of the ring.
It is important that in catalysis that the transition metals are produced with the correct stereochemistry. Carbon monoxide is a 2-electron donor and can be found to be bridging 3 different metals only two thirds of an electron can be donated to each. This can only arise with a number of different electron present.
Adding this to the electron counting principles above and this is what we get:
2-electron donators are seen in two different ways. A dative donor is seen as 2-electrons being given. As there are no electron coming from the metal. π ligands meaning any organic molecule with a π bond will donate in the same way.
Bridging halides are strange in which 3 electrons are given but to two different metal centres.
Ligands
For a long time it has been known that an electron donor can bind to a metal centre through shared electron density. The first ligands that were used were usually lone pair donors that could easily bond in a predictable manor with a metal centre. More commonly now is the use of high electron density species which have a large amount of electron density around them. Ligands such as Ferrocene and alkenes were important and were and still are used regularly in Ziegler Natta catalysts etc. Transition metal chemists draw the bond going from the metal to the middle of the ring.
It is important that in catalysis that the transition metals are produced with the correct stereochemistry. Carbon monoxide is a 2-electron donor and can be found to be bridging 3 different metals only two thirds of an electron can be donated to each. This can only arise with a number of different electron present.
Adding this to the electron counting principles above and this is what we get:
Prefixes
The prefix comes before the ligand which takes the structure ηn where n in the number of electrons coming from the ligand. When ever there is an absence of the prefix assume the maximum. Hapticity can also be used for this nomenculture. is used to indicate how many metal centres that are present and are bridging between a metal centre. K is the number of metals present.
Metal-metal bonds
This is where a radical left over can dimerise forming a metal metal bonds where unstable molecules can stabilise themselves. Although this is quite a short entry it should be noted that it is very important to look out for.
Bridiging ligands.
A good example of bridging ligands is the Mn(CO)5Br ligand. This involves the lone pairs being able to bridge between two metal centres. This can be seen when the manganese group is heated losing a CO ligand. This allows the formation of the bridging bromine ions with a 3 centre and 2 electron bond.
The 18 electron rule is not valuable in coordination chemistry only in organometallic chemistry. Classical coordination chemistry usually forms an octahedral complex. The 18 electron rule works, and shouldn’t be looked into too deeply.
Exceptions to the 18 electron rule
Exceptions to the 18 electron rule are explained below these normally form due to steric effects in the early transition metals. It is also worth watching out for Cp ligands as these can cause further stability than expected. This causes less electrons to be found in SOME complexes.
Ti(CH2SiMe3)4 is small tranision metal which here is completely saturated and still has no more space for further groups to donate electrons.
Cp2ZrCl2 follows the same principle.
It is important to look at the different possibilities of ligand filling but also important to remember the steric effects.
From chromium through to iron groups normally have 18 electrons.
When the late transition metals are seen there are possibilities of the formation of square planar complexes. These metals tend to follow a 16 electron rule as the increased stabilization of the d-shell as the stabilization increases. This causes the dz2 orbital to be filled due to the higher electron density which causes this orbital to no longer be acknowledged and a 16 electron rule form.
The prefix comes before the ligand which takes the structure ηn where n in the number of electrons coming from the ligand. When ever there is an absence of the prefix assume the maximum. Hapticity can also be used for this nomenculture. is used to indicate how many metal centres that are present and are bridging between a metal centre. K is the number of metals present.
Metal-metal bonds
This is where a radical left over can dimerise forming a metal metal bonds where unstable molecules can stabilise themselves. Although this is quite a short entry it should be noted that it is very important to look out for.
Bridiging ligands.
A good example of bridging ligands is the Mn(CO)5Br ligand. This involves the lone pairs being able to bridge between two metal centres. This can be seen when the manganese group is heated losing a CO ligand. This allows the formation of the bridging bromine ions with a 3 centre and 2 electron bond.
The 18 electron rule is not valuable in coordination chemistry only in organometallic chemistry. Classical coordination chemistry usually forms an octahedral complex. The 18 electron rule works, and shouldn’t be looked into too deeply.
Exceptions to the 18 electron rule
Exceptions to the 18 electron rule are explained below these normally form due to steric effects in the early transition metals. It is also worth watching out for Cp ligands as these can cause further stability than expected. This causes less electrons to be found in SOME complexes.
Ti(CH2SiMe3)4 is small tranision metal which here is completely saturated and still has no more space for further groups to donate electrons.
Cp2ZrCl2 follows the same principle.
It is important to look at the different possibilities of ligand filling but also important to remember the steric effects.
From chromium through to iron groups normally have 18 electrons.
When the late transition metals are seen there are possibilities of the formation of square planar complexes. These metals tend to follow a 16 electron rule as the increased stabilization of the d-shell as the stabilization increases. This causes the dz2 orbital to be filled due to the higher electron density which causes this orbital to no longer be acknowledged and a 16 electron rule form.