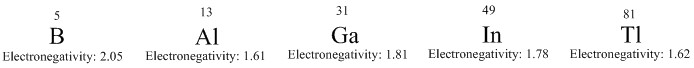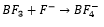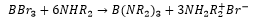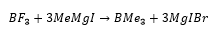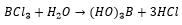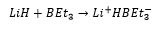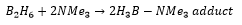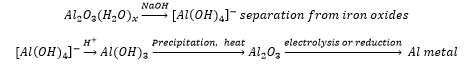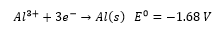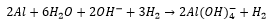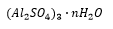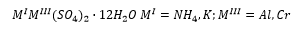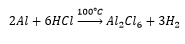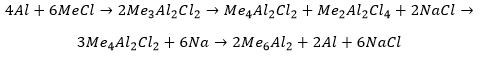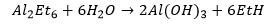Group 13 Elements
The elements of group 13 vary in abundance on the planet; aluminium is abundant whereas boron has a very small abundance in the atmosphere, from here the concentrations of the elements decreases down the group.
The physical properties of group 13, being a p block group, vary greatly moving down the group. This can be seen by boron being considered a non-metal, aluminium being a metalloid and the last three elements being metals.
This also means the bonding changes down the group, from boron bonding predominantly covalently and the bonding becoming progressively more ionic as you move down the group. The valence shell configuration of the group 13 elements is ns2np1 this means that the first elements tend to form +3 ions whereas the later molecules in the group tend to form +1 elements, this is where inert pair effect tends to take effect on the molecule causing it to be more stable.
Most group 13 chemistry is dominated by the fact that the elements can only form 3 bonds with their 3 valence electrons this means their reactions with Lewis bases is important chemistry. Due to their valence shell configuration these molecules are well documented for their strange products that they produce with themselves or other elements. One of the most documented being the 2 electron 3 center bonding present in Diborane.
Amphoteric
Amphoteric is a word that comes up a lot here, a molecule that is amphoteric is a molecule that can react with both Lewis acids and Lewis bases. The fact that aluminium is amphoteric means it is classed as a metalloid even though most of its other properties are metallic.
Boron - "Boron but not boring!" The state of these lectures determine that was a lie.
The physical properties of group 13, being a p block group, vary greatly moving down the group. This can be seen by boron being considered a non-metal, aluminium being a metalloid and the last three elements being metals.
This also means the bonding changes down the group, from boron bonding predominantly covalently and the bonding becoming progressively more ionic as you move down the group. The valence shell configuration of the group 13 elements is ns2np1 this means that the first elements tend to form +3 ions whereas the later molecules in the group tend to form +1 elements, this is where inert pair effect tends to take effect on the molecule causing it to be more stable.
Most group 13 chemistry is dominated by the fact that the elements can only form 3 bonds with their 3 valence electrons this means their reactions with Lewis bases is important chemistry. Due to their valence shell configuration these molecules are well documented for their strange products that they produce with themselves or other elements. One of the most documented being the 2 electron 3 center bonding present in Diborane.
Amphoteric
Amphoteric is a word that comes up a lot here, a molecule that is amphoteric is a molecule that can react with both Lewis acids and Lewis bases. The fact that aluminium is amphoteric means it is classed as a metalloid even though most of its other properties are metallic.
Boron - "Boron but not boring!" The state of these lectures determine that was a lie.
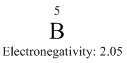
Boron's properties are based that it is an electron deficient atom. The electronegativity is central at 2.05. This means that it forms covalent bonds with other atoms.
Boron has many similar properties to silicon in that it forms acidic oxides whereas aluminium forms an amphoteric oxide.
Boron is well known for the 3 centre 2 electron bonds (3c,2e bonds). This is one of the best ways to stabilise borane forming a dimer. This can be thought of as the highly electronegative centre pulling electron density away from a neighbouring borane atom. It can also be noted here that the terminal boron-hydrogen bonds are much shorter than the 3c, 2e bonds.
Boron is found in several allotropes, amorphous boron is in the form of a brown powder but crystalline boron is hard and forms shiny black crystals. Boron usually forms large –hedron structures, using the B12 icosahedron as the bases for most of its structures. These structure when crystalline makes boron the hardest element after diamond. The boron element is very hard and brittle. This causes boron to have properties that are between the metals and non-metals. The natural occurrence “borax” which is a hydrated oxide of the boron compound. This can be extracted with acid to form a diborate group which can then be reduced by magnesium.
Boron has many similar properties to silicon in that it forms acidic oxides whereas aluminium forms an amphoteric oxide.
Boron is well known for the 3 centre 2 electron bonds (3c,2e bonds). This is one of the best ways to stabilise borane forming a dimer. This can be thought of as the highly electronegative centre pulling electron density away from a neighbouring borane atom. It can also be noted here that the terminal boron-hydrogen bonds are much shorter than the 3c, 2e bonds.
Boron is found in several allotropes, amorphous boron is in the form of a brown powder but crystalline boron is hard and forms shiny black crystals. Boron usually forms large –hedron structures, using the B12 icosahedron as the bases for most of its structures. These structure when crystalline makes boron the hardest element after diamond. The boron element is very hard and brittle. This causes boron to have properties that are between the metals and non-metals. The natural occurrence “borax” which is a hydrated oxide of the boron compound. This can be extracted with acid to form a diborate group which can then be reduced by magnesium.
The small size of the boron 3+ means it has a very large charge to radius ratio meaning it is not stable. This forms covalent bonds with elements as the bonding is highly polarizing pulling the electron density away from the element. There are a number of boron-oxygen compounds as well:
Boron oxygen compounds:
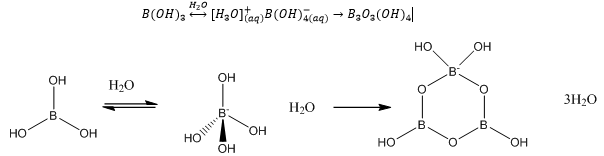
The most simple of the boron oxygen compounds is boric acid.
The most simple of the boron oxygen compounds is boric acid.
The charge here is spread over all of the bonds present as there is effective conjugation between the B-O and the π-conjugation. These complexes have a very low expansion coefficient which means they are much more resistant to thermal shock. They are often used as additives to glass.
Boron is also essential as a plate nutrient as it can inhibit certain enzyme complexation.
Boron halides
The boron halides have boiling points that form a distinct pattern:
Boron is also essential as a plate nutrient as it can inhibit certain enzyme complexation.
Boron halides
The boron halides have boiling points that form a distinct pattern:
This pattern is seen due to the changes in molecular weight, as the weight of the molecule increases so does its boiling point, the larger halogens are also easier to polarize due to more diffuse shells and more electrons being present. This means the Van der Vaals forces that form between the molecules occur more frequently and are of greater strength.
Boron tetraflouride
The formation of boron tetraflouride is useful for many chemical reactions. Boron tetraflouride can be present as a stabilising, non-coordinating anion. Usually coordinating with carbocations.
Boron tetraflouride
The formation of boron tetraflouride is useful for many chemical reactions. Boron tetraflouride can be present as a stabilising, non-coordinating anion. Usually coordinating with carbocations.
The boron halides themselves can be reacted with Lewis bases to allow the formation of boron compounds.
They can also be used as alkylating agents allowing for the formation of further compounds. This is carried out with a boron halide and a Grignard reagent.
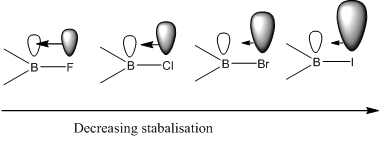
Lewis acidity (accepting electrons) increases from fluoride to iodide, this goes against common thinking. It would be expected for fluorine to pull electrons away from the boron making it more electropositive to accept electrons, this does not happen.
What happens is the lone pairs in the fluorine p orbitals are around the same energy level as the empty p orbital on the boron, this means that the p orbital on these fluorine’s can overlap with the empty p orbital on the boron making the overall molecule less capable of accepting electrons. This means that these molecules tend to be less reactive than BI3 for example.
What happens is the lone pairs in the fluorine p orbitals are around the same energy level as the empty p orbital on the boron, this means that the p orbital on these fluorine’s can overlap with the empty p orbital on the boron making the overall molecule less capable of accepting electrons. This means that these molecules tend to be less reactive than BI3 for example.
Reactions of the boron halides: The boron halides react to form adducts with Lewis bases this usually makes the boron halides very dangerous to handle as they are highly acidic:
This shows how when the electrons move from the water to the boron one of the hydrogen atoms on the water molecule becomes much more protic.
Boron nitrogen compounds
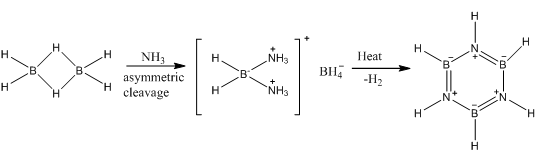
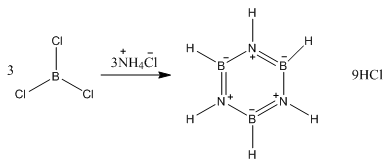
The reaction of a borane molecule with ammonia gives borazine, this is isoelectronic to benzene. Sharing similar properties of intermediate bond lengths.
Boron nitrogen compounds
The reaction of a borane molecule with ammonia gives borazine, this is isoelectronic to benzene. Sharing similar properties of intermediate bond lengths.
This can also be produced with ammonium and boron trichloride
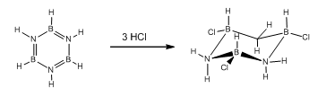
Borazine has a limited reactivity due to the fact that the bonds are polar, this means it is more reactive than the isoelectronic benzene. This polarity needs to be explisity stated. This allows the bonds to be even represented as localised charges on the atoms as the nitrogen is more likely to be a negative charge.
This can be reacted with hydrochloric acid, something that benzene cannot achieve.
This can be reacted with hydrochloric acid, something that benzene cannot achieve.
Boron nitride
Hexagonal boron nitrides: Similar to graphene in structure can be used as solid state lubricant. Dissociates at 3000°C
Cubic boron nitride: Second only to diamond in hardness.
Boron hydrides
The formation of boron hydrides is important as tetrahydroborate compounds are regually used as a reducing agent. This can be further reacted to produce a “superhydride” solution which is an even stronger reducing agent.
Hexagonal boron nitrides: Similar to graphene in structure can be used as solid state lubricant. Dissociates at 3000°C
Cubic boron nitride: Second only to diamond in hardness.
Boron hydrides
The formation of boron hydrides is important as tetrahydroborate compounds are regually used as a reducing agent. This can be further reacted to produce a “superhydride” solution which is an even stronger reducing agent.
The reactions of boron groups with bases is an example of an adduct forming reaction. The boron and the base could be seen as having a negative and positive charge respectively although these cancel out in the overall molecular formula.
This adduct can then be hydrolysed to boric acid.
Organoboranes
These are found as square planar monomers which are Lewis acids. These can form anionic complexes, it is usual for the organoboron species to be reacted with a nitrogen group forming borazines. These are the same as discussed above in that the electrons are highly localized.
These are found as square planar monomers which are Lewis acids. These can form anionic complexes, it is usual for the organoboron species to be reacted with a nitrogen group forming borazines. These are the same as discussed above in that the electrons are highly localized.

Aluminium
Aluminium is found in a large abundance in the earth’s crust. It is commonly found in its oxide form. This is due to aluminium being a very hard Lewis acid, meaning it bonds to oxygen very strongly. The high energy aluminium oxygen bond dominates the chemistry of aluminium.
The separation of aluminium from Bauxite is a very high energy process due to the breaking of the aluminium oxide bonds. This involves reduction with carbon as a sacrificial electrode. Electrolysis uses cryolite as part of the Hall-Heroult process, this lowers the melting point of the aluminium from ~2000°C to less than 1000°C. This allows a current to be based through the molten material forming aluminium.
Aluminium is found in a large abundance in the earth’s crust. It is commonly found in its oxide form. This is due to aluminium being a very hard Lewis acid, meaning it bonds to oxygen very strongly. The high energy aluminium oxygen bond dominates the chemistry of aluminium.
The separation of aluminium from Bauxite is a very high energy process due to the breaking of the aluminium oxide bonds. This involves reduction with carbon as a sacrificial electrode. Electrolysis uses cryolite as part of the Hall-Heroult process, this lowers the melting point of the aluminium from ~2000°C to less than 1000°C. This allows a current to be based through the molten material forming aluminium.
Aluminium can be very easily oxidised.
The reason why Aluminium is not easily dissolved by water is due to surface pacification. This involves the use of a protective oxide film that forms rapidly but is very hard to break down. As a result of this any oxidising acids will not dissolve aluminium metal. Although dilute non-oxidising acids such will not form this protective layer and will dissolve the aluminium.
Aluminium readily dissolves in strong base, in this case hydroxide ions:
Aluminium readily dissolves in strong base, in this case hydroxide ions:
Aluminium forms many different ligand molecules, these are affected by a series of properties that are held by aluminium and the ligand coordinating to the molecule.
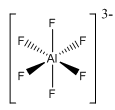
Some of the factors are the metal ion size: this explains why aluminium can form a six coordinate molecule whereas boron can only form a 4 coordinate molecule. Coordination number is also affected by the size of the ligand, in this many cases the ligands can be very large or small affecting the total number of charges able to fit round the metal cation. In relationship to the last point the charge of the ligand also affects the coordination number, highly charged ligands will repel each other meaning that other molecules will not be able to fit around the molecule.
At lower pH levels the water doesn’t dissociate.
Aluminium structures
AlO4
This structure dominates aluminium chemistry due to the strength of the bonds. Aluminium has more freedom due to the use of an expanded octet five and six- coordinate compounds can form.
Crystalline Al2O3 ceramics are used, α-Al2O3 is very hard and abrasive and γ-Al2O3 is capable of absorbing water, it is often used as a stationary phase for chromatography or as a drying agent.
Aluminosilicates
Aluminosilicates are an important compound which allow most plants to grow, they are commonly found in clays and rocks. The diagram below shows how the aluminosilicate is formed and how the tetrahedral aluminium compound and the silicon compound are isoelectronic. It can be seen though that the aluminium has a negative charge meaning that any cations can easily bond with this group. This means that water retention can occur through this charge or important cation minerals for growth.
Common Al salts
Some of the factors are the metal ion size: this explains why aluminium can form a six coordinate molecule whereas boron can only form a 4 coordinate molecule. Coordination number is also affected by the size of the ligand, in this many cases the ligands can be very large or small affecting the total number of charges able to fit round the metal cation. In relationship to the last point the charge of the ligand also affects the coordination number, highly charged ligands will repel each other meaning that other molecules will not be able to fit around the molecule.
At lower pH levels the water doesn’t dissociate.
Aluminium structures
AlO4
This structure dominates aluminium chemistry due to the strength of the bonds. Aluminium has more freedom due to the use of an expanded octet five and six- coordinate compounds can form.
Crystalline Al2O3 ceramics are used, α-Al2O3 is very hard and abrasive and γ-Al2O3 is capable of absorbing water, it is often used as a stationary phase for chromatography or as a drying agent.
Aluminosilicates
Aluminosilicates are an important compound which allow most plants to grow, they are commonly found in clays and rocks. The diagram below shows how the aluminosilicate is formed and how the tetrahedral aluminium compound and the silicon compound are isoelectronic. It can be seen though that the aluminium has a negative charge meaning that any cations can easily bond with this group. This means that water retention can occur through this charge or important cation minerals for growth.
Common Al salts
“Alum” is an aluminium sulphate, this is used in paper as a coagulate agent. There is also the possibility of forming mixed sulfates which have multiple metal ions at different oxidation states. An example shown below is:
Aluminium halides
Fluorides are regally seen bonded to aluminium these form large infitie crystal structures with octahedral Al3+. This is an important mineral cryolite (). This is very different from the Boron structures.
Fluorides are regally seen bonded to aluminium these form large infitie crystal structures with octahedral Al3+. This is an important mineral cryolite (). This is very different from the Boron structures.
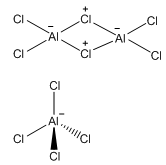
The larger chlorine atoms cannot form an octahedral complex with aluminium meaning only a tetrahedral structure can form. This single monomer is present in the gas phase, but it is often seen as the Al2Cl6 dimer.
This can be produced by the reaction of aluminium with hydrochloric acid in a tube reactor.
Aluminium alkyls
These can be synthesised by reacting with methyl chloride then reducing with sodium.
These can be synthesised by reacting with methyl chloride then reducing with sodium.
These form 3 electron 2 centre bonding species unlike the alkyl boranes. This is because the bigger aluminium atom can allow the fitting of more alkyl groups around it. If a hydride is present in the aluminium complex this will preferentially form the bridging atom over any alkyl groups.
This compounds react violently in oxygen forming aluminium oxides. These compounds form aluminium (III) hydroxide and an alkane on reaction with water.
This compounds react violently in oxygen forming aluminium oxides. These compounds form aluminium (III) hydroxide and an alkane on reaction with water.