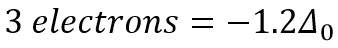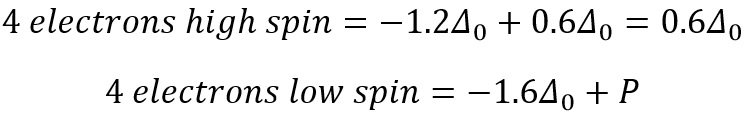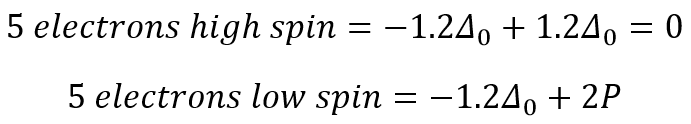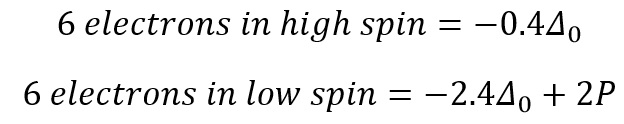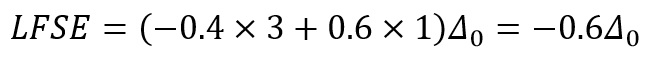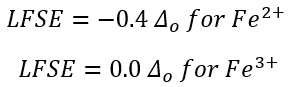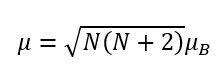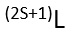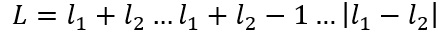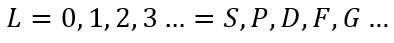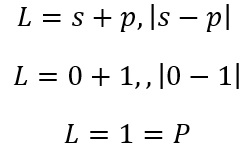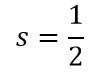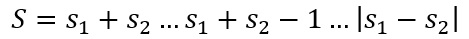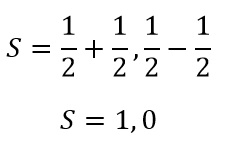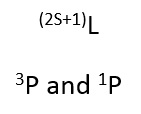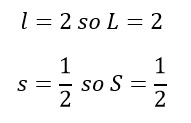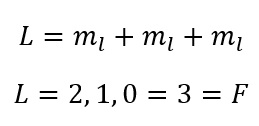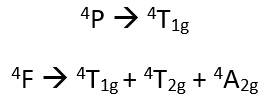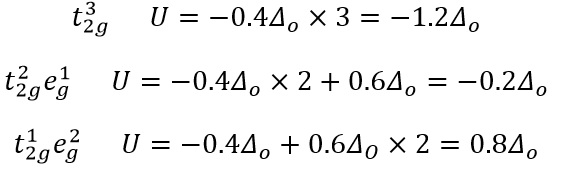Ligand field splitting
We have already discussed crystal field splitting. This section will focus a lot more on ligands themselves and the spectrochemical series.
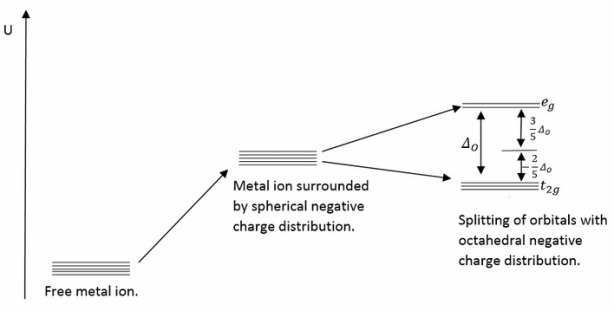
Octahedral splitting of the d-shell.
We have already discussed crystal field splitting. This section will focus a lot more on ligands themselves and the spectrochemical series.
Octahedral splitting of the d-shell.
The octahedral splitting of the d-shell shown above shows how the different energy levels vary in energy compared to the initial excited energy level. As the first three electrons enter the base shells the stabilisation is -0.6Δ0.
The next electrons to be added can now enter the high spin orbital or they can pair with the low energy t2g orbital. These two different configurations correspond to two different energies.
Where P is the pairing energy.
This can then be compared to the two different five electron states.
This can then be compared to the two different five electron states.
This explains why the five electron configuration has a similar hydration enthalpy due to the symmetry of the orbitals present. The symmetry is caused by each orbital being filled equally and there being no overall stabilisation present compared to the ground state.
For the d6 states the behaviour starts to change as now pairing HAS to occur. This can either occur in high spin or low spin.
For the d6 states the behaviour starts to change as now pairing HAS to occur. This can either occur in high spin or low spin.
This value is 2P as there needs to be pairing occurring now, so therefore does not stabilise or add more energy than the ground state. This means that finding the stablilisation energy of each field is actually a very simple process that is based on the stabilisation of the orbitals and the pairing that is present for each.
Once d8 and d9 are reached there can be no high or low spin states meaning the stabilisation is purely on the overall electrons in the ground state there are no pairing energies here.
Molecular orbital description of ligand field theory
The use of crystal field theory still leaves many holes in the acknowledgment of how the ligands effect the properties of the complex. If there is one thing to remember in this it is the fact that the d orbitals are antibonding orbitals, of ligands that bond to the metal centre. It is also important to remember all of the rules that surround MO and that the orbitals that contribute to bonding must do so with geometric overlap and similar energy levels.
σ donor
The σ donor ligands such as a chloride ligand have electrons that are present in the p orbital. These can be donated by the overlap of a p bond to the dz2 or the d(x2-y2) of the orbital. In these examples the eg groups are therefore involved in the bonding and are therefore antibonding orbitals the t2g groups are not involved in the bonding as there is no orbital overlap present so no interactions occur.
As the σ donoation gets better there is more energy involved in the interactions this causes the splitting between the bonding and antibonding orbitals to get larger pushing the σ donoation gets better there is more energy involved in the interactions this causes the splitting between the bonding and antibonding orbitals to get larger pushing the eg orbitals even higher in energy. This can then cause low spin states to form more readily.
π donor
The p orbitals that are present can overlap with some the t2g orbitals of the metal. This can cause electron donation to occur which increases the energy of the t2g orbitals. As the π donation gets better and better the splitting becomes larger and the field splitting becomes smaller and smaller.
π acceptor
A π acceptior ligand is one that donates a σ bond and also has an empty or partially filled p orbital this can cause back donation which causes a larger Δ0 to form.
Effects of ligand field splitting
Thermochemical data
Hydration energies, redox potentials and lattice enthalpies. Can be used and can then explain the difference in ligand field stabilisation in the d-orbitals. The ionic radii and the coordination geometries are also related.
Hydration energy
Hydration energies are related to the formation and filling of the d-orbitals with a big basis on high spin low spin complexes. The symmetric spherical electron distributions are found to have a linear relation to one another the humps in the orbitals form around this.
The factors that affect hydration energies are the effective charge on the transition metal and the ionic radius that is therefore formed. It is also possible that there are ligand field effects on this. In this case we compare the hexaaquo ion of each of the metal ions is looked at. This leaves the ligand field symmetry to be taken into account, the haxaquo ion is octahedral. The water ligand is a weak field ligand, this means that a high spin complex is likely to form.
Using Chromium hexahydrate as an example:
The LFSE for this is therefore:
Once d8 and d9 are reached there can be no high or low spin states meaning the stabilisation is purely on the overall electrons in the ground state there are no pairing energies here.
Molecular orbital description of ligand field theory
The use of crystal field theory still leaves many holes in the acknowledgment of how the ligands effect the properties of the complex. If there is one thing to remember in this it is the fact that the d orbitals are antibonding orbitals, of ligands that bond to the metal centre. It is also important to remember all of the rules that surround MO and that the orbitals that contribute to bonding must do so with geometric overlap and similar energy levels.
σ donor
The σ donor ligands such as a chloride ligand have electrons that are present in the p orbital. These can be donated by the overlap of a p bond to the dz2 or the d(x2-y2) of the orbital. In these examples the eg groups are therefore involved in the bonding and are therefore antibonding orbitals the t2g groups are not involved in the bonding as there is no orbital overlap present so no interactions occur.
As the σ donoation gets better there is more energy involved in the interactions this causes the splitting between the bonding and antibonding orbitals to get larger pushing the σ donoation gets better there is more energy involved in the interactions this causes the splitting between the bonding and antibonding orbitals to get larger pushing the eg orbitals even higher in energy. This can then cause low spin states to form more readily.
π donor
The p orbitals that are present can overlap with some the t2g orbitals of the metal. This can cause electron donation to occur which increases the energy of the t2g orbitals. As the π donation gets better and better the splitting becomes larger and the field splitting becomes smaller and smaller.
π acceptor
A π acceptior ligand is one that donates a σ bond and also has an empty or partially filled p orbital this can cause back donation which causes a larger Δ0 to form.
Effects of ligand field splitting
Thermochemical data
Hydration energies, redox potentials and lattice enthalpies. Can be used and can then explain the difference in ligand field stabilisation in the d-orbitals. The ionic radii and the coordination geometries are also related.
Hydration energy
Hydration energies are related to the formation and filling of the d-orbitals with a big basis on high spin low spin complexes. The symmetric spherical electron distributions are found to have a linear relation to one another the humps in the orbitals form around this.
The factors that affect hydration energies are the effective charge on the transition metal and the ionic radius that is therefore formed. It is also possible that there are ligand field effects on this. In this case we compare the hexaaquo ion of each of the metal ions is looked at. This leaves the ligand field symmetry to be taken into account, the haxaquo ion is octahedral. The water ligand is a weak field ligand, this means that a high spin complex is likely to form.
Using Chromium hexahydrate as an example:
The LFSE for this is therefore:
The hydration enthalpy can be seen to closely realte to the LFSE this gives the impression that they can directly explain one another.
Electrode potentials
The ligand that has been added can alter the redox potential. The Fe3+/Fe2+ couple varies from ligand to ligand. With one ligand such as (o-phenanthroline)3 the oxidation potential is higher meaning the lower oxidation state is preferred. This means that Fe2+ is preffered in this half cell:
Electrode potentials
The ligand that has been added can alter the redox potential. The Fe3+/Fe2+ couple varies from ligand to ligand. With one ligand such as (o-phenanthroline)3 the oxidation potential is higher meaning the lower oxidation state is preferred. This means that Fe2+ is preffered in this half cell:
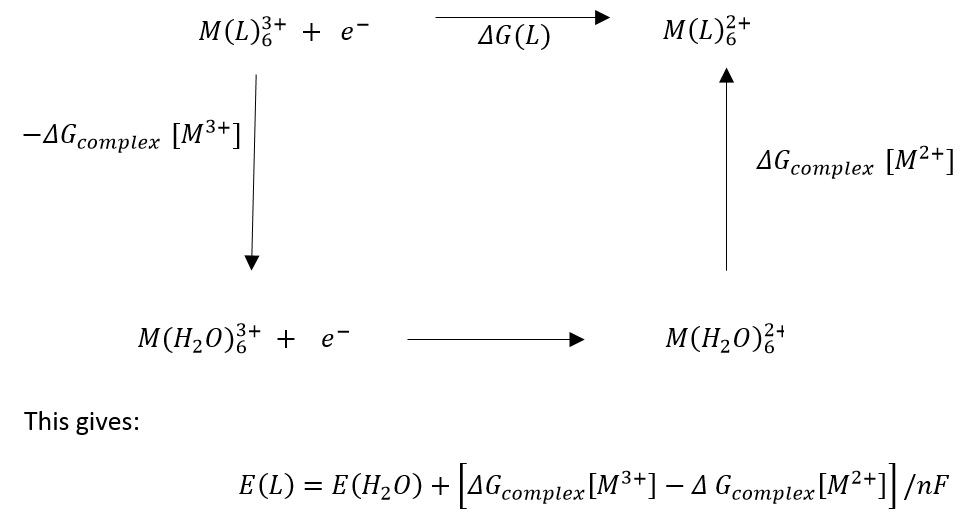
The electrode potentials for M3+/M2+ can be seen to vary from ligand to ligand but is this a direct effect of ligand field effects? Ligand formation enthalpies are large in the region of 5000kJ mol-1 whereas the LFSE are only 140kJ mol-1. This shows that the ligand field effects are greatly overcome compared to the formation enthalpy.
This means that the energy cycle can be produced. Giving the change in energy caused by changes in oxidation states. This means that the total energy of a ligand can be given.
This means that the energy cycle can be produced. Giving the change in energy caused by changes in oxidation states. This means that the total energy of a ligand can be given.
Electrode potentials
The ligand field alter the redox potential by preferentially stabilising one oxidation state over another. The LFSE is just one small component in This does show how the LFSE tends to be more stabalised at the lower oxidation states with the lower potential complex although the more likely explanation is that the M-ligand bond favours M2+ energy. The same is seen in cobalt but the opposite oxidation state is favoured so why could this be? The ionic radii could have the largest effect.
The filling of the d-orbitals needs to be remembered that these are antibonding in character. This means that the jump from chromium to manganese across the first row is due to the high spin state Cr2+ moving to the Manganese with an electron in the orbital.
Lattice site preferences
The Spinel structure has cubic close packed O2- ions. The A2+ is in 1/8 of the tetrahedral sites. The B3+ ion is in the more stabalised octahedral sites and is present in ½ of them. The electrostatic energy favours higher charge in the higher co-ordination.
In the structure which can be better written as: the LFSE favours in the tetrahedral sites. This can be seen by the different stabalisation:
The ligand field alter the redox potential by preferentially stabilising one oxidation state over another. The LFSE is just one small component in This does show how the LFSE tends to be more stabalised at the lower oxidation states with the lower potential complex although the more likely explanation is that the M-ligand bond favours M2+ energy. The same is seen in cobalt but the opposite oxidation state is favoured so why could this be? The ionic radii could have the largest effect.
The filling of the d-orbitals needs to be remembered that these are antibonding in character. This means that the jump from chromium to manganese across the first row is due to the high spin state Cr2+ moving to the Manganese with an electron in the orbital.
Lattice site preferences
The Spinel structure has cubic close packed O2- ions. The A2+ is in 1/8 of the tetrahedral sites. The B3+ ion is in the more stabalised octahedral sites and is present in ½ of them. The electrostatic energy favours higher charge in the higher co-ordination.
In the structure which can be better written as: the LFSE favours in the tetrahedral sites. This can be seen by the different stabalisation:
This shows that it is more benifical to have the Fe2+ to be in the octahedral sites and the Fe3+ to fill the remaineder of these sites and then the tetrahedral sites. This leaves an intresting situation for the ion:
CoFe2O4
The inverse spinel structure forms here with cubic close packed O2- ions here the Cobalt ion is even more favoured and fills the1 ¼ of the octahedral sites and the Fe3+ is found in the 1/8 of the tetrahedral sites and ¼ octahedral sites.
Magnetism
The magnetism of a metal complex can be based on the Jahn-Teller distortions and the co-ordination symmetries. The Jahn-Teller distortions are if the ground state of a non-linear complex is orbitally degenerate the complex will distort to minimise the possibility of one electron in a degenerate energy shell. This can be seen in a 4 electron high spin octahedral structure. A single electron is found in the orbital. This causes there to be tetragonal distortion. This works by allowing there to be a larger effect on the orbital.
The axis becomes distorted in Jahn-Teller distortion to allow the energies of the level to drop which causes the energy of the other orbital to increase. Weak field and strong field ligands causes a difference in stabalisation energies. Jahn-Teller distortions forming square planar is the most favoured of all of these. Although tetrahedral structure forms when steric effects take hold.
Spin-only formula
The spin only formula is based on the total spin quantum number of the complex and the Bohr magneton behaviour. This is represented by the spin and how many unpaired electrons are present.
CoFe2O4
The inverse spinel structure forms here with cubic close packed O2- ions here the Cobalt ion is even more favoured and fills the1 ¼ of the octahedral sites and the Fe3+ is found in the 1/8 of the tetrahedral sites and ¼ octahedral sites.
Magnetism
The magnetism of a metal complex can be based on the Jahn-Teller distortions and the co-ordination symmetries. The Jahn-Teller distortions are if the ground state of a non-linear complex is orbitally degenerate the complex will distort to minimise the possibility of one electron in a degenerate energy shell. This can be seen in a 4 electron high spin octahedral structure. A single electron is found in the orbital. This causes there to be tetragonal distortion. This works by allowing there to be a larger effect on the orbital.
The axis becomes distorted in Jahn-Teller distortion to allow the energies of the level to drop which causes the energy of the other orbital to increase. Weak field and strong field ligands causes a difference in stabalisation energies. Jahn-Teller distortions forming square planar is the most favoured of all of these. Although tetrahedral structure forms when steric effects take hold.
Spin-only formula
The spin only formula is based on the total spin quantum number of the complex and the Bohr magneton behaviour. This is represented by the spin and how many unpaired electrons are present.
The spin only formula is based on the magnetic moment of the d-block metal complex. It is important to base on whether or not the complex is high spin or low spin and how deviations from the spin only formula occur.
Deviaitons form the spin only formula
When the presence of angylar momentum cuases the movement of electrons the spin only formula is not as an effective prediction. This can be represented by the presence of un-paired electrons in un-filled orbitals. This causes the magnetic moment to change rapidly depending on the spin orbit coupling.
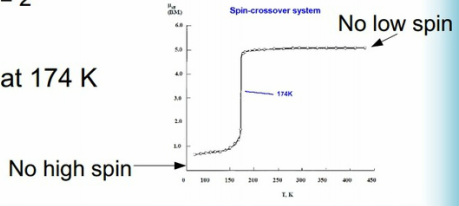
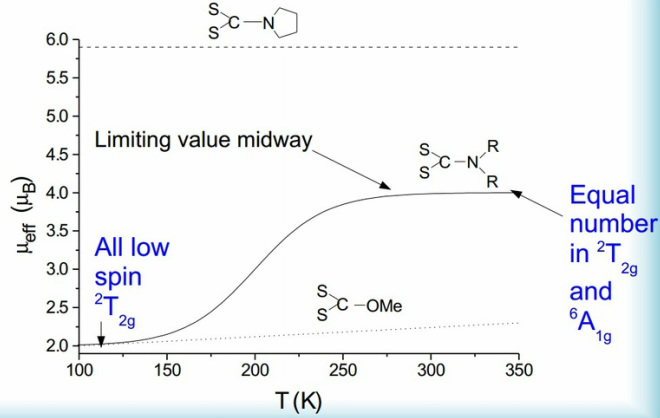
High spin / low spin crossover.
This occurs when there is a change in the ground state during the analysis. This can occur in thermal changes. It is important to keep in mind the possibility of Jahn-Teller effects etc. during these.
At high temperatures there can be a rapid movement of all the complexes from a low spin to a high spin state in some of these there is a 100% change so no atoms can be found in a low spin state.
Deviaitons form the spin only formula
When the presence of angylar momentum cuases the movement of electrons the spin only formula is not as an effective prediction. This can be represented by the presence of un-paired electrons in un-filled orbitals. This causes the magnetic moment to change rapidly depending on the spin orbit coupling.
High spin / low spin crossover.
This occurs when there is a change in the ground state during the analysis. This can occur in thermal changes. It is important to keep in mind the possibility of Jahn-Teller effects etc. during these.
At high temperatures there can be a rapid movement of all the complexes from a low spin to a high spin state in some of these there is a 100% change so no atoms can be found in a low spin state.
UV-Vis spectra
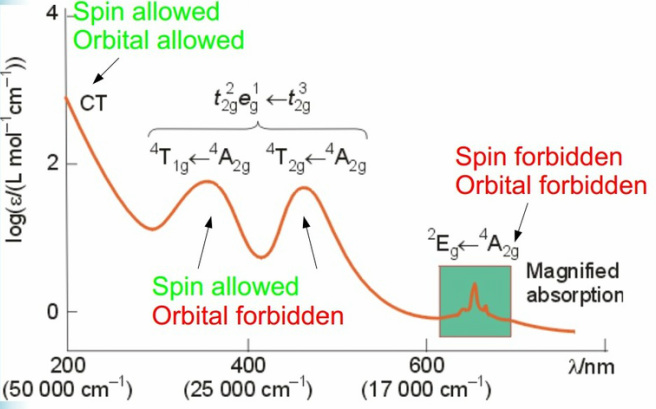
The analysis of transition metals using UV/Vis can be affective of predicting where the formation of bands will occur. This is represented by the d-d electron transitions and what sort of transitions are occurring.
The bands have varying energies depending on how “allowed” the movement is. If the nature of the transition is forbidden then the intensity of the band will be much reduced.
The selection rules that apply to this are:
The analysis of transition metals using UV/Vis can be affective of predicting where the formation of bands will occur. This is represented by the d-d electron transitions and what sort of transitions are occurring.
The bands have varying energies depending on how “allowed” the movement is. If the nature of the transition is forbidden then the intensity of the band will be much reduced.
The selection rules that apply to this are:
Spin
There must not be a change in spin. This can be easily seen from the term symbol if the singlet doublet triplet value changes.
Angular momentum
This transition still occurs due to slight mixing of the p orbital in these orbitals. This allows the transition to occur although it is highly forbidden.
Laporte selection rule
The transitions that do not cause a change in the symmetry are not allowed.
This transition is allowed when there are tetrahedral complexes this is where the reduction in symmetry removes centre of symmetry meaning there are no u/g labels. This means that the ΔL is relaxed as better p and d orbital mixing. This has a higher molar absorptivity.
Spectroscopic term symbols
It can be seen by the diagram below that there are two peaks close to one another, this gives the impression that one of the transitions is similar in value to another but are not fully degenerate.
There must not be a change in spin. This can be easily seen from the term symbol if the singlet doublet triplet value changes.
Angular momentum
This transition still occurs due to slight mixing of the p orbital in these orbitals. This allows the transition to occur although it is highly forbidden.
Laporte selection rule
The transitions that do not cause a change in the symmetry are not allowed.
This transition is allowed when there are tetrahedral complexes this is where the reduction in symmetry removes centre of symmetry meaning there are no u/g labels. This means that the ΔL is relaxed as better p and d orbital mixing. This has a higher molar absorptivity.
Spectroscopic term symbols
It can be seen by the diagram below that there are two peaks close to one another, this gives the impression that one of the transitions is similar in value to another but are not fully degenerate.
Up until now the electron configurations of atoms have been described using the designation of the number of electrons in each shell and which orbital (Li = 1s2,2s1). This is satisfactory but does not give the full picture. The use of these to describe atoms does not give any detail about what dimension the occupied orbital is in, whether it is x, y or z or the relative spin that the electron is in.
Russell-Saunders coupling
DON’T PANIC! We will get through this. Take it slow and don’t let yourself get too bogged down.
As discussed, the idea of using the electron configuration to describe an electron is ineffective. The complete description of an electron is termed a microstate, this gives a highly useful information but can only be used when electron-electron repulsions are not accounted for. This is not useful when electronic spectra are being determined so the microstates are grouped forming term symbols.
Summary: A microstate describes everything about an electron but does not take into account repulsion. This is useless so term symbols are used.
The use of term symbols groups microstates into easy to use data, this is done by summing the angular momentum and the spin of the electrons present and combining the two. This gives a simple approach to the presence of electrons represented by the term symbol equation below:
Russell-Saunders coupling
DON’T PANIC! We will get through this. Take it slow and don’t let yourself get too bogged down.
As discussed, the idea of using the electron configuration to describe an electron is ineffective. The complete description of an electron is termed a microstate, this gives a highly useful information but can only be used when electron-electron repulsions are not accounted for. This is not useful when electronic spectra are being determined so the microstates are grouped forming term symbols.
Summary: A microstate describes everything about an electron but does not take into account repulsion. This is useless so term symbols are used.
The use of term symbols groups microstates into easy to use data, this is done by summing the angular momentum and the spin of the electrons present and combining the two. This gives a simple approach to the presence of electrons represented by the term symbol equation below:
Okay, this looks all mean and scary but it’s not. Trust.
In these examples I will use an excited Beryllium atom with electron configuration: [He] 2s1 2p1
The L value
The L value is represented as the total angular momentum this is simply the addition of the sub-shells that the electrons are in.
In these examples I will use an excited Beryllium atom with electron configuration: [He] 2s1 2p1
The L value
The L value is represented as the total angular momentum this is simply the addition of the sub-shells that the electrons are in.
So for this L value find the maximum using and then the minimum: and fill the integers in-between.
Okay so, for the excited helium atom: [He] 2s1 2p1
We have:
We have:
So we have our L value. As both the minimum and the maximum value are equal to 1.
The S value
This leaves the S value, this is uses the same formula as the L value but purely based on the spin or electrons.
We know the electron spin is:
The S value
This leaves the S value, this is uses the same formula as the L value but purely based on the spin or electrons.
We know the electron spin is:
And:
This means that when there are two electron present the equation is now:
This means that when these two values are substituted into the term symbol equation:
Energy of the states
Once the values for L and S are found it is possible to identify the lowest energy term using Hunds rule.
1. For a given configuration the term with the greatest multiplicity lies lowest in energy.
The multiplicity is the 2S+1 value. Which means out of:
3P and 1P
The 3P lies lowest in energy.
1. For a given multiplicity the greatest angular momentum (L) lies lowest in energy.
This is justified physically by the ability for electrons to take up more space when L is high and therefore be able to minimize electron repulsion forces.
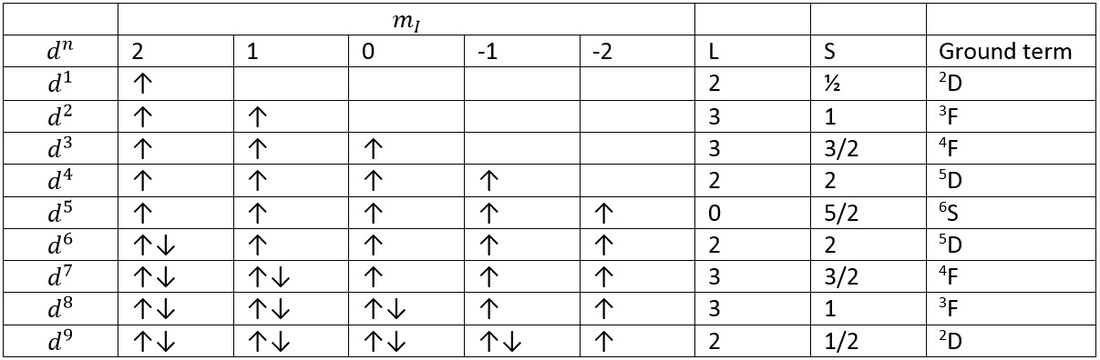
Use of Russell-Saunders in transition metals
One d electron can be represented by Russell-Saunders by the following d1:
Once the values for L and S are found it is possible to identify the lowest energy term using Hunds rule.
1. For a given configuration the term with the greatest multiplicity lies lowest in energy.
The multiplicity is the 2S+1 value. Which means out of:
3P and 1P
The 3P lies lowest in energy.
1. For a given multiplicity the greatest angular momentum (L) lies lowest in energy.
This is justified physically by the ability for electrons to take up more space when L is high and therefore be able to minimize electron repulsion forces.
Use of Russell-Saunders in transition metals
One d electron can be represented by Russell-Saunders by the following d1:
This should make sense as the orbital angular momentum is purely a d-orbital and the addition of a single electron means the spin value is 1/2.
This means the term symbol is:
2D
This means the term symbol is:
2D
This is where each arrow represents a microstate of an electron. For the d1 electron this microstate is 2+ because it is in the second value for ml and the + value for spin.
d3 electron configuration
Lowest energy terms for d3 are:
d3 electron configuration
Lowest energy terms for d3 are:
P is also found here which is higher in energy.
This means the two term symbol states are 4F and 4P.
In the effect of the ligand field the term symbols split.
This means the two term symbol states are 4F and 4P.
In the effect of the ligand field the term symbols split.
The 4P and the 4F have orbital splitting energy of 15B, this is explained later on.
So there are possible electron configurations in a strong ligand field case. These are:
So there are possible electron configurations in a strong ligand field case. These are:
Group theory is then used to relate each of these relationships to their term symbols.
Finished. TM.