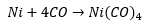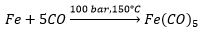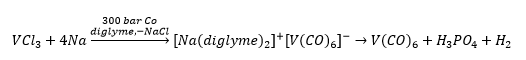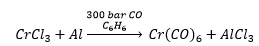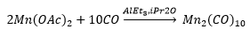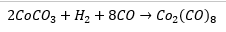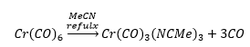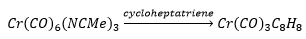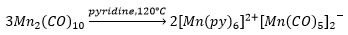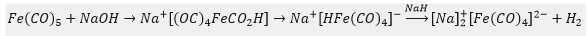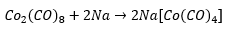Organometallic carbonyls
The carbonyl ligand is common ligand in organometallic chemistry, it has a number of interesting bonding characteristics and analysis of the carbon oxygen bond can yield information about the metal center.
Synthesis
The synthesis of metal carbonyl complexes has a long history. The first complex produced was nickel metal with carbon monoxide. This yellow liquid produced has a boiling point of 34°C which could be distilled to a pure form. This was important as nickel could be purified at a large scale with this process as high purities could be obtained. This was then burnt giving carbon dioxide and Nickel metal.
The synthesis of metal carbonyl complexes has a long history. The first complex produced was nickel metal with carbon monoxide. This yellow liquid produced has a boiling point of 34°C which could be distilled to a pure form. This was important as nickel could be purified at a large scale with this process as high purities could be obtained. This was then burnt giving carbon dioxide and Nickel metal.
This is an 18VE tetrahedral structure. Nickel tetracarbonyl is a potent carcinogen.
Iron pentacarbonyl is a yellow liquid also:
Iron pentacarbonyl is a yellow liquid also:
These are the only two metals that react with carbon monoxide directly. The difference in carbonyl ligands is purely based on the ability to form 18 electrons.
With other metals this is not as easy. Potassium napthalide is a potent reducing agent.
With other metals this is not as easy. Potassium napthalide is a potent reducing agent.
This gives the formation of titanium in oxidation state -2. This is rare for other ligands which makes carbonyl ligands very important. When working with carbonyl complexes it is sterically unfavourable to be able to get higher than seven carbonyl groups surrounding it. As this is impossible more electrons are added to the metal which gives the negative charge on the ligand to still make the 18VE.
This forms a compound with an 18 electron compound initially and then the 17 electron hexacarbonyl vanadium. It is not possible to form a vanadium bond in this situation.
This gives an 18 electron species which is highly favourable.
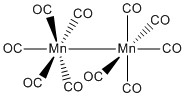
The reaction of manganese acetate with carbon monoxide produces a dimer. This is due to the monomer species having 17 electrons total. This is unfavourable so two manganese complexes bond together forming a dimer.
The reaction of manganese acetate with carbon monoxide produces a dimer. This is due to the monomer species having 17 electrons total. This is unfavourable so two manganese complexes bond together forming a dimer.
The dimer formed can be seen to have two octahedral maganese complexes bonded together. The fact that they are octahedral is very predictable now but becomes important a bit later. The species formed is a yellow crystal meaning that it can be separated and easily identified.
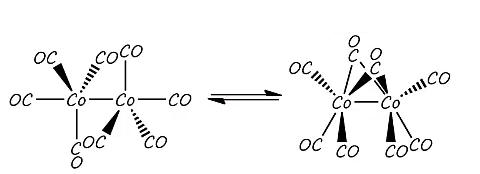
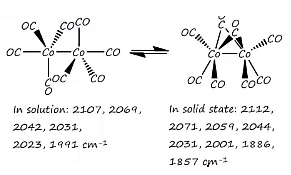
Cobalt carbonate uses some interesting features such as the reduction with hydrogen and the fact that a number of bridging ligands can occur. The formation of the bridging carbonyl ligands occurs due to the fact that a six bonded structure can be formed. The cobalt species donates nine electrons to the total count and each carbonyl directly bonded donates 2 electrons as normal. The bridged carbonyl donates one electron to each metal center.
The two bridging carbonyls give one electron to each metal centre. Each of these are present in different amounts in solution and solid state.
Metal-CO bonding has many different classical ligands such as NH3, which is a Lewis base and form donor bonds to Lewis acids such as Ni2+ via lone pairs. These ligands do not make complexes with zerovalent metals. CO is a very weak base so does not easily bond to metals on paper, although the molecular orbital theory can have a better explanation.
The HOMO of carbonyl ligand very easily overlaps with the orbitals of the metal. This is the s antibonding orbital which interacts easily with the dx2-y2 this donation allows strong bonding of the metal carbon bond. With the other orbitals the LUMO being the π* antibonding orbitals immediately interact with the orbitals of the metal producing a stronger M-C bond. This means that both of these compounds form stronger M-C bonds.
This process is known as back bonding.
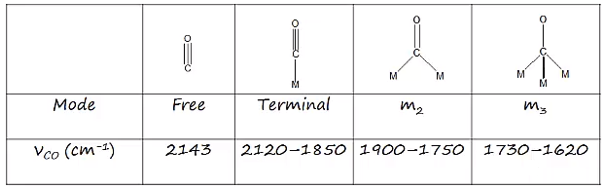
The bonding modes of carbon monoxide is a polar molecule. This has a carbon oxygen triple bond which has a distinctive stretching frequency, around 2000 cm-1. This means that the different bonds that form cause a difference in the carbon – oxygen bond strength causing differnet stretching frequencies.
Metal-CO bonding has many different classical ligands such as NH3, which is a Lewis base and form donor bonds to Lewis acids such as Ni2+ via lone pairs. These ligands do not make complexes with zerovalent metals. CO is a very weak base so does not easily bond to metals on paper, although the molecular orbital theory can have a better explanation.
The HOMO of carbonyl ligand very easily overlaps with the orbitals of the metal. This is the s antibonding orbital which interacts easily with the dx2-y2 this donation allows strong bonding of the metal carbon bond. With the other orbitals the LUMO being the π* antibonding orbitals immediately interact with the orbitals of the metal producing a stronger M-C bond. This means that both of these compounds form stronger M-C bonds.
This process is known as back bonding.
The bonding modes of carbon monoxide is a polar molecule. This has a carbon oxygen triple bond which has a distinctive stretching frequency, around 2000 cm-1. This means that the different bonds that form cause a difference in the carbon – oxygen bond strength causing differnet stretching frequencies.
Bridging carbonyl ligands are therefore seen at lower freqecncies due to donation of lone pair of electrons. This causes back donation which decreases the strength of the carbon oxygen bond.
In solution it can be seen that there are no bridging carbonyl ligands. There are low frequency carbonyl stretches at 1886 – 1857 cm-1.
This just takes some thinking but it really quite simple. Once understood back bonding needs to be considered.
So when back bonding isn’t present the triple bond becomes stronger this occurs with boron with carbonyl ligands.
Homoleptic compounds
Compounds are homoleptic when they have the same ligands surrounding one another.
Reactions of 18 electron species
An 18 electron complex, does not have the ability to accept a ligand as this would form a twenty electron complex. This means it moves through a coordinatively unsaturated intermediate. This is a dissociative substitution process.
The use of larger metals react faster in these reactions.
There are two ways that promote substitution to allow different complex to form. This is carried out either thermally, where the complex is heated releasing ligands, it the use of electronic excitation.
The reflux of hexacarbonyl chromium with acetonitrile forms a tricarbonyl species. This occurs because the acetonitrile is in excess which means it is kinetically favoured for this compound to form:
This just takes some thinking but it really quite simple. Once understood back bonding needs to be considered.
So when back bonding isn’t present the triple bond becomes stronger this occurs with boron with carbonyl ligands.
Homoleptic compounds
Compounds are homoleptic when they have the same ligands surrounding one another.
Reactions of 18 electron species
An 18 electron complex, does not have the ability to accept a ligand as this would form a twenty electron complex. This means it moves through a coordinatively unsaturated intermediate. This is a dissociative substitution process.
The use of larger metals react faster in these reactions.
There are two ways that promote substitution to allow different complex to form. This is carried out either thermally, where the complex is heated releasing ligands, it the use of electronic excitation.
The reflux of hexacarbonyl chromium with acetonitrile forms a tricarbonyl species. This occurs because the acetonitrile is in excess which means it is kinetically favoured for this compound to form:
This can be reacted with cycloheptatriene, this can then be reduced forming a cycloheptatriene molecule can be formed.
This is an 18 electron compound.
Carbonyl substitution reactions usually involve milder reaction conditions. This allows the synthesis of more labile products. This allows the formation of liabile complexes. When there is the excitation of a metal centre alkenes can be directly added to the group. The addition of cyclo-octene can form a bond to the iron tetracarbonyl centre. This can be carried out twice with excitation inbetween the additions. If everything else is remained neutral. By reducing the number of CO ligands is rapidly becoming much stronger.
When the octene molecules are reacted with the metal centre there is an increase in strength of the metal carbon bond increases meaning it is unlikely that the metal carbon bond will allow any removal of more carbonyl groups. The carbonyl ligand is being replaced by an alkene ligand which means there is a reduction of CO ligands.
Metal carbonyl anions
There are a number of metal carbonyl anions. These are present due to the need to form an 18 electron count. The stable anions complexes can be much more preferred. The reaction of dimaganese diccarbonyl in a single chemical reasons.
Carbonyl substitution reactions usually involve milder reaction conditions. This allows the synthesis of more labile products. This allows the formation of liabile complexes. When there is the excitation of a metal centre alkenes can be directly added to the group. The addition of cyclo-octene can form a bond to the iron tetracarbonyl centre. This can be carried out twice with excitation inbetween the additions. If everything else is remained neutral. By reducing the number of CO ligands is rapidly becoming much stronger.
When the octene molecules are reacted with the metal centre there is an increase in strength of the metal carbon bond increases meaning it is unlikely that the metal carbon bond will allow any removal of more carbonyl groups. The carbonyl ligand is being replaced by an alkene ligand which means there is a reduction of CO ligands.
Metal carbonyl anions
There are a number of metal carbonyl anions. These are present due to the need to form an 18 electron count. The stable anions complexes can be much more preferred. The reaction of dimaganese diccarbonyl in a single chemical reasons.
[Mn(Py)6]2+ does not use organometallic ligands and therefore only has 17 electrons present.
The stable electron donation of two electrons for the pyridine carbonyl ligands can stabalise low or negative oxidation states. Although it is very rare to see transition metals in different oxidation states.
Iron pentacarbonyl with sodium hydroxide forms a carboxylate intermediate which released carbon dioxide. This forms a hydrido species which can then be removed forming hydrogen and tetracarbonyl ferrate.
The stable electron donation of two electrons for the pyridine carbonyl ligands can stabalise low or negative oxidation states. Although it is very rare to see transition metals in different oxidation states.
Iron pentacarbonyl with sodium hydroxide forms a carboxylate intermediate which released carbon dioxide. This forms a hydrido species which can then be removed forming hydrogen and tetracarbonyl ferrate.
Whenever a dinuclear metal complex are present there is the possibility of very easy reduction. This allows the addition of an electron which stabalised in THF. This can be carried out by the following:
These carbonyl anions are thermally stable but they are sensitive to oxidation. The negative charge is centred on the metal. This can be removed by oxidation. A strong oxidant will react with these compounds. These are metal centred bases, or metal centre nucleophiles.
Carbonyl metallates are strong bases/ nucleophiles. The basicity increase down the group. Strong donor ligands increase basicity. Weak base implies strong conjugate acid this is where carbonyl hydrides can be used.
The electron density on the metal centre is based on how the ligands give or remove electron density from the metal centre. Carbonyls remove electrons from the metal centre making the compound less basic. Large cyclo-ene ligands allow large donations of electrons meaning there is a large basicity.
The relative basicity of these compounds varies by around 7 factors of 10. Weak conjugate basis: strong conjugate base. This means that any sort of hydride ligands attached to these metal carbonyls form very strong acids.
Metal carbonyl hydrides
There is a large amount of electron density on the proton meaning it is no longer a proton. It is a hydride ligand. This means that when the molecule dissociates the hydrogen forms a negatively charged ion with a positively charged metal cation. These hydride species can be produced by reacting metal carbonyl anions with strong acids such as sulphuric acid.
This is only stable under a carbon monoxide atmosphere otherwise these structures decompose through to the metal. This can only be stopped by reacting under carbonyl metal compounds. The thermal stability increases from first row to third row.
Neutral carbonyl complexes are protonated by strong acids to give cationic hydrides. The addition of a hydride should be seen as the addition of a negative charge and the oxidation state will change accordingly.
Carbonyl metallates are strong bases/ nucleophiles. The basicity increase down the group. Strong donor ligands increase basicity. Weak base implies strong conjugate acid this is where carbonyl hydrides can be used.
The electron density on the metal centre is based on how the ligands give or remove electron density from the metal centre. Carbonyls remove electrons from the metal centre making the compound less basic. Large cyclo-ene ligands allow large donations of electrons meaning there is a large basicity.
The relative basicity of these compounds varies by around 7 factors of 10. Weak conjugate basis: strong conjugate base. This means that any sort of hydride ligands attached to these metal carbonyls form very strong acids.
Metal carbonyl hydrides
There is a large amount of electron density on the proton meaning it is no longer a proton. It is a hydride ligand. This means that when the molecule dissociates the hydrogen forms a negatively charged ion with a positively charged metal cation. These hydride species can be produced by reacting metal carbonyl anions with strong acids such as sulphuric acid.
This is only stable under a carbon monoxide atmosphere otherwise these structures decompose through to the metal. This can only be stopped by reacting under carbonyl metal compounds. The thermal stability increases from first row to third row.
Neutral carbonyl complexes are protonated by strong acids to give cationic hydrides. The addition of a hydride should be seen as the addition of a negative charge and the oxidation state will change accordingly.