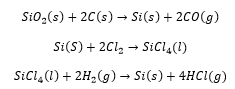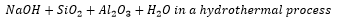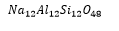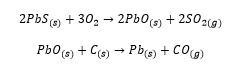|
|
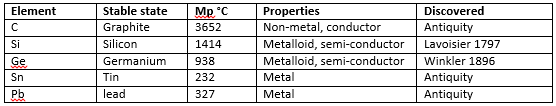
Group 14 is one of the most important groups in the periodic table; containing carbon, which has a whole chemistry topic after it (organic), silicon found wide spread and used in many chemical vessels and processes, tin and lead are found widespread in many chemical processes in the form of catalysts and both are found with wide uses in industry.
The properties of the elements changes quite drastically down the group, from the non-metals carbon and silicon to the metalloid germanium and then the metals tin and lead. Silicon and germanium are used as semiconductors. The reason that the heavier elements are metals are the fact that their outer shells are more diffuse and can form cations more easily this gives them more metal-character compared to carbon which has a less diffuse shell. This diffuse shell leads to less electronegativity, which further decreasing the melting points. The bond strength also decreases down the group, an important property caused by diffuse shells.
An important factor with the group 14 elements is that they have 4 valence electrons meaning that +4 is their most common oxidation state. An exception to this is lead which has an oxidation state of +2 more commonly, this is due to the inert pair effect which is a feature of all heavier p block elements.
Carbon, silicon and germanium form electron precise compounds, this is an important factor to note as they do not act as Lewis acids or Lewis bases. This can make some of their structures highly un-reactive such as graphite.
Carbon and silicon are quite similar in how they react and the structures that they form, it is important to note that carbon and silicon have very similar electronegativities to hydrogen meaning that their molecules do not form highly intermolecular bonding active molecules.
The properties of the elements changes quite drastically down the group, from the non-metals carbon and silicon to the metalloid germanium and then the metals tin and lead. Silicon and germanium are used as semiconductors. The reason that the heavier elements are metals are the fact that their outer shells are more diffuse and can form cations more easily this gives them more metal-character compared to carbon which has a less diffuse shell. This diffuse shell leads to less electronegativity, which further decreasing the melting points. The bond strength also decreases down the group, an important property caused by diffuse shells.
An important factor with the group 14 elements is that they have 4 valence electrons meaning that +4 is their most common oxidation state. An exception to this is lead which has an oxidation state of +2 more commonly, this is due to the inert pair effect which is a feature of all heavier p block elements.
Carbon, silicon and germanium form electron precise compounds, this is an important factor to note as they do not act as Lewis acids or Lewis bases. This can make some of their structures highly un-reactive such as graphite.
Carbon and silicon are quite similar in how they react and the structures that they form, it is important to note that carbon and silicon have very similar electronegativities to hydrogen meaning that their molecules do not form highly intermolecular bonding active molecules.
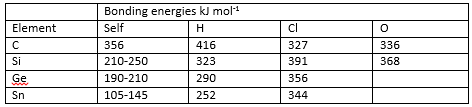
The difference in properties between the compounds that these elements form is due to the large difference in bonding energies between each element and hydrogen, oxygen and bonds with itself.
This shows how it is more favourable for carbon to bond with itself and hydrogen rather than oxygen. This is important as it allows the formation of hydrocarbon chains, the bonding energy of the silicon oxygen bond is very high. This means that it is very rare for there to be the formation of the silicon hydrogen bonds. Silicon is therefore mostly found as a silicate.
Carbon
Carbon has very different chemical properties to the rest of the group 14 elements due to the differences in electron density and radius; this is pronounced by the small atomic radius being able to form π bonds.
Carbon is well known for the major difference it shows in its allotropes. These are shown in the two allotropes of carbon: Graphite and diamond.
Graphite consists of stacks of planar graphene layers each bonded with 3 other carbons the remaining perpendicular p orbitals can overlap forming π bonds; this means the carbon is sp2 hybridised in graphite. The intermolecular forces bonding the layers together are relatively weak allowing the layers to slide over one another. This allows graphite to actually be used as a lubricant.
Diamond is bonded forms four bonds to carbons around it forming a regular tetrahedron; this forms a very rigid covalent structure in a strong three dimensional framework.
Nanotubes can be viewed as rolled layers of graphene, this is an exciting and new technology that is actually very effective at some functions that normal carbon is not capable of.
Carbon is the only group 14 elements that can form π bonds; this is due to the small atomic radius of carbon allowing it to form π-bonds. To efficiently form multiple bonds p orbitals need to join to form π bonds because p orbitals point straight up it is important that both atoms are actually very close together to allow bonding to occur, this doesn’t happen with the larger elements. This is typical across most of the early elements.
Carbon has very different chemical properties to the rest of the group 14 elements due to the differences in electron density and radius; this is pronounced by the small atomic radius being able to form π bonds.
Carbon is well known for the major difference it shows in its allotropes. These are shown in the two allotropes of carbon: Graphite and diamond.
Graphite consists of stacks of planar graphene layers each bonded with 3 other carbons the remaining perpendicular p orbitals can overlap forming π bonds; this means the carbon is sp2 hybridised in graphite. The intermolecular forces bonding the layers together are relatively weak allowing the layers to slide over one another. This allows graphite to actually be used as a lubricant.
Diamond is bonded forms four bonds to carbons around it forming a regular tetrahedron; this forms a very rigid covalent structure in a strong three dimensional framework.
Nanotubes can be viewed as rolled layers of graphene, this is an exciting and new technology that is actually very effective at some functions that normal carbon is not capable of.
Carbon is the only group 14 elements that can form π bonds; this is due to the small atomic radius of carbon allowing it to form π-bonds. To efficiently form multiple bonds p orbitals need to join to form π bonds because p orbitals point straight up it is important that both atoms are actually very close together to allow bonding to occur, this doesn’t happen with the larger elements. This is typical across most of the early elements.
Silicon
Silicon holds many of the similar properties to carbon although it does not form π bonds as readily as carbon due to the orbitals being quite large, this is why CO2 is a gas and SiO2 forms polymeric minerals.
Silicon chips are taken from a slice of “pure” silicon. Silicon is purified taking sand (SiO2) and carbon and reacting it together releasing carbon monoxide. The silicon can then be reacted with chlorine and this precipitated with hydrogen giving pure silicon:
Silicon holds many of the similar properties to carbon although it does not form π bonds as readily as carbon due to the orbitals being quite large, this is why CO2 is a gas and SiO2 forms polymeric minerals.
Silicon chips are taken from a slice of “pure” silicon. Silicon is purified taking sand (SiO2) and carbon and reacting it together releasing carbon monoxide. The silicon can then be reacted with chlorine and this precipitated with hydrogen giving pure silicon:
Silicon tetrachloride is highly volatile and can therefore be separated from the other compounds through fractional distillation. This allows very pure silicon to be formed in the final stage of the reaction.
Elemental silicon normally forms a tetrahedral structure very similar to diamond.
Organosilicon monomers and silicones
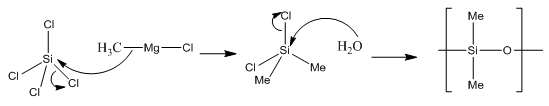
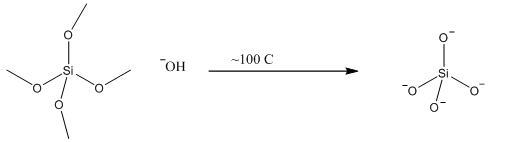
Organosilicon polymers can be produced from the reaction between silicon tetrachloride and a Grignard reagent. On addition of water a polymerisation reaction occurs forming a polymeric chain, these are known as silicones:
Elemental silicon normally forms a tetrahedral structure very similar to diamond.
Organosilicon monomers and silicones
Organosilicon polymers can be produced from the reaction between silicon tetrachloride and a Grignard reagent. On addition of water a polymerisation reaction occurs forming a polymeric chain, these are known as silicones:
These are high molecular weight polymers and have similar structural principles to the silicates above. The methyl groups attached cannot cross link and are hydrophobic to allow more cross linking less Grignard reagent should be used. This forms a much more plastic polymer rather than the usual oil.
Silicates
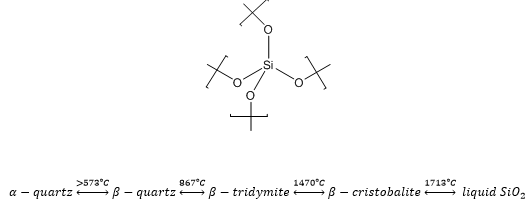
Silicon atoms are too large to form π bonds with oxygen molecules meaning a tetrahedral structure is preferred. This means that silicon is more likely to form large polymeric groups with oxygen being the central binding group. SiO2 has a high thermal stability based on the Si-O bond. It is based on corner-sharing tetrahedral structures. There is NO double bond. There are eight different crystal types that are found at different temperatures as less favourable energy states can be accessed.
Silicates
Silicon atoms are too large to form π bonds with oxygen molecules meaning a tetrahedral structure is preferred. This means that silicon is more likely to form large polymeric groups with oxygen being the central binding group. SiO2 has a high thermal stability based on the Si-O bond. It is based on corner-sharing tetrahedral structures. There is NO double bond. There are eight different crystal types that are found at different temperatures as less favourable energy states can be accessed.
There are a number of uses for silicon dioxide, α-quartz can be used as a piezoelectric crystal oscillator and a transducer. Vitreous silica has a very low thermal expansion coefficient and a high thermal shock resistance, this is normally used in glass ware. The low UV absorption also makes it beneficial for this to be used in optical instruments. Amorphous silica is used in silica gel, it is highly porus and has a large surgace area. It is used as a drying agent, catalyst support and a chromatography stationary phase.
These silca materials are stable to acids except from HF. These form SiF4 or SiF62-. When attacked by hot concentrated alkali they form a silicate anion, this is an important physical process. The silicate anion has the formula SiO42-.
These silca materials are stable to acids except from HF. These form SiF4 or SiF62-. When attacked by hot concentrated alkali they form a silicate anion, this is an important physical process. The silicate anion has the formula SiO42-.
Water can decrease the melting point of the silica due to interactions between the oxide bridging groups and the polar water. This is a hydrothermal process regularly seen in rock formation.
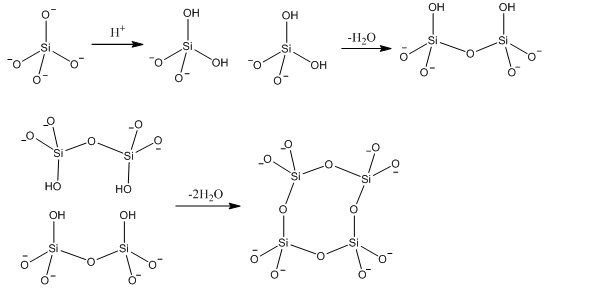
These silicates can be used to form polymeric materials with different compounds as well as having a large amount of control on their own formation. This can be carried out by adding small amounts of acid and allowing a condensation reaction to occur. This can form highly hydrophilic structures which due to a high polarity. There are often used as a stationary phase in normal phase HPLC. This condensation reaction shown below, can form sheets of silicates which can then be used to form cation storage compounds.
These silicates can be used to form polymeric materials with different compounds as well as having a large amount of control on their own formation. This can be carried out by adding small amounts of acid and allowing a condensation reaction to occur. This can form highly hydrophilic structures which due to a high polarity. There are often used as a stationary phase in normal phase HPLC. This condensation reaction shown below, can form sheets of silicates which can then be used to form cation storage compounds.
On the addition of acetate as a terminating group the addition of water can hydrolyse this group and form a further cross linked structure. These are commonly used in bathroom sealants.
Chain silicates
Chain silicates are large silicate chains that form a large negatively charged sheet. These sheets can be exposed to cations causing a trapping effect. The silicate sheets have distinct cation sizes which can be filled with different sized cations creating a different structure each time. When a cation is too big the silicate sheet can distort allowing the uptake of the larger cation.
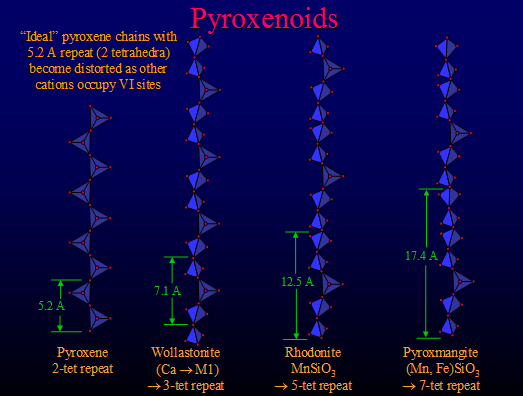
Pyroxenes
Pyroxenes:- (XYSi2O6)n where X and Y are two different cations. These cations can fit into one of two different octahedral holes labelled M1 and M2. M2 is a larger hole and can be distorted, this means that when there are two cations and the largest can still accesses the M2 hole the pyroxene structure can be produced. This is seen with MgCaSi2O6 having a pyroxene structure and CaSiO3 not.
Chain silicates
Chain silicates are large silicate chains that form a large negatively charged sheet. These sheets can be exposed to cations causing a trapping effect. The silicate sheets have distinct cation sizes which can be filled with different sized cations creating a different structure each time. When a cation is too big the silicate sheet can distort allowing the uptake of the larger cation.
Pyroxenes
Pyroxenes:- (XYSi2O6)n where X and Y are two different cations. These cations can fit into one of two different octahedral holes labelled M1 and M2. M2 is a larger hole and can be distorted, this means that when there are two cations and the largest can still accesses the M2 hole the pyroxene structure can be produced. This is seen with MgCaSi2O6 having a pyroxene structure and CaSiO3 not.
This diagram shows how the distortion of the structure can occur allowing a larger amount of cation variability.
Asbestos
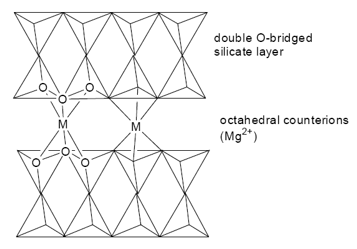
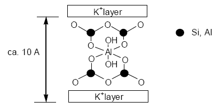
Asbestos is a chain/layed silicate. It is a fibrous material and has some incredible properties such as being fire resistant. The silicate layers in asbestos form so to allow octahedral cations to fit between the layers maintaining the structure. There is no bonding directly between the silicate layers.
Asbestos
Asbestos is a chain/layed silicate. It is a fibrous material and has some incredible properties such as being fire resistant. The silicate layers in asbestos form so to allow octahedral cations to fit between the layers maintaining the structure. There is no bonding directly between the silicate layers.
White asbestos is the most common. This has the formula: [Mg3(Si2O5)(OH)4], this is a layer silicate and constitutes 98% of world use. Blue asbestos is based on [Si4O11]6- double chains.
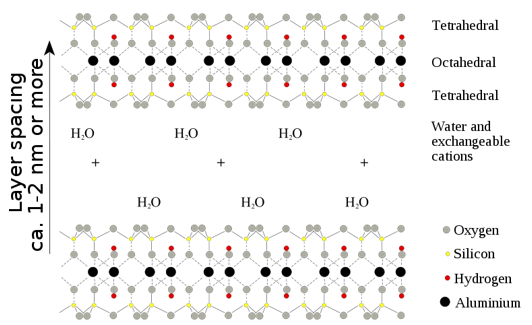
It is important to remember that many silicate materials are based on the principle that a (alumino)silicate clay layered structure can hold water and exchangeable cations inside of it. This can then allow the movement of different cations in and out of the structure and the replacement of cations. This allows these compounds to act as reducing agents.
It is important to remember that many silicate materials are based on the principle that a (alumino)silicate clay layered structure can hold water and exchangeable cations inside of it. This can then allow the movement of different cations in and out of the structure and the replacement of cations. This allows these compounds to act as reducing agents.
Mg2+ and Ca2+ can be exchanged for Na+ and H+, these act as excellent ion exchangers and acidic catalysts. These can all be stored in the central aqueous section. The layering of the compounds can also allow the substance to be used as lubricants for oil drilling. The capacity for swelling and absorption has been used since roman times for, degreasing and cleansing wool and pet litter.
The following structures are more complex and it is unlikely they will be questioned although it is important to note they have the same properties of cation exchange as well as being silicate sheets.
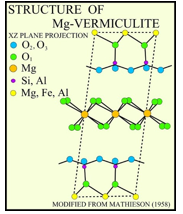
Vermiculite
Octahedral Mg2+ partially replaced by Al3+, this gives a porous layer material on heating: chemical inert packaging, insulation and agriculture.
The following structures are more complex and it is unlikely they will be questioned although it is important to note they have the same properties of cation exchange as well as being silicate sheets.
Vermiculite
Octahedral Mg2+ partially replaced by Al3+, this gives a porous layer material on heating: chemical inert packaging, insulation and agriculture.
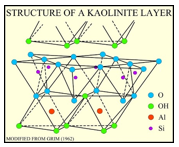
Kaolinite
Clay mainly used as paper filler.
Clay mainly used as paper filler.
Ball Clay
Ball clay is a mixture of kaolinite + clay-mica + quartz. It is best for porcelain and earthenware. It is highly plastic material when hydrated and can be expanded to give porous support for industrial catalysts.
Mica
“I can be green I can be mean I can be anything more.”
Ball clay is a mixture of kaolinite + clay-mica + quartz. It is best for porcelain and earthenware. It is highly plastic material when hydrated and can be expanded to give porous support for industrial catalysts.
Mica
“I can be green I can be mean I can be anything more.”
Mica cleaves across the layers into tough elastic transparent plates. They are early forms of electrical insulators and can be made thin enough to become a capacitor due to the presence of potassium ions and oxide ions. The sheet mica can also form a very strong window, used in many furnaces.
Clay hardening
Clay hardening happens in much the same way as the silica sheet formation occurs. Hydroxide goups bonded to the aluminium can react with one another in a condensation reaction forming larger structures.
Zeolites
Zeolites are an important part of many ion capture storage methods. They work by being a aluminosilicate mineral in a cage like structure. These have a very high surface area trapping many ions inside of the cages.
Clay hardening
Clay hardening happens in much the same way as the silica sheet formation occurs. Hydroxide goups bonded to the aluminium can react with one another in a condensation reaction forming larger structures.
Zeolites
Zeolites are an important part of many ion capture storage methods. They work by being a aluminosilicate mineral in a cage like structure. These have a very high surface area trapping many ions inside of the cages.
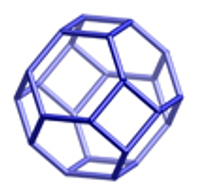
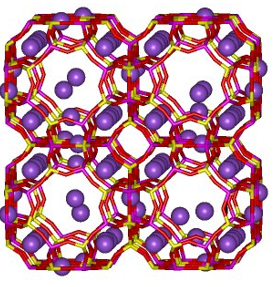
Above is a sodalite, this is a truncated cuboctahedron, this is the building unit for the entire zeolite structure. This uses a three dimensional interconnection via 4- and 6- membered rings. This are formed out of the following reactants.
Zeolite A
In this example it is important to note that the Aluminium and the silicon are in a 1:1 ratio. The negatively charged aluminium is counteracted by the positively charged sodium.
The purple atoms represent sodium ions trapped in the structure. These are able to move in and out of the channel network.
For zeolite A there are 8 sodalite cages which are linked by 4-rings to give a supercage with and 8 ring entrance. It has a pose size of 4-5 A which makes it highly suitable for water and also for ion exchange. It can be used as a non-phosphate water softener in washing powders. It can also absorb water so can be used as a drying agent.
Other zeolite structures
There are over 100 natural zeolite structures that form, plus 100s of synthetic phase zeolites that have been produced. Some of the more important are ZSM-5 which catalyses carbocationic reactions. This is able to catalyse these reactions as it can stabalise cationic species allowing there transition across its surface. This allows the reaction of benzyl groups to its surface.
Sodalite
Sodalite is a non-pourous arrangement of cuboctahedral, they are cages that contain anions. Soadalite cages contain trapped S3- and S2- radical ions which give a non-fading blue pigment.
Tin
Tin has two main allotropes α tin and β tin, α tin has the same structure to diamond although it is not that stable as tin has lager atomic orbitals this means it usually degrades into a 6 bonded structure above 13˚C.
When tin is warmed a shiny metal is given, when tin is cooled below 13˚C α-tin begins to form which is a powdery structure as compared to β-tin. In the medieval times organ pipes were made of tin, when the winter came in the northern areas of Britain β-tin which is shiny and brilliant, and forms to α-tin and was thought to be caused by the devil.
Tin occurs naturally mainly as cassiterite: SnO2 and can be retained by reduction with carbon at very high temperatures. SnCl4 is tetrahedral in structure and is a Lewis acid (This is due to an expanded valence shell! Not like CCl4!) This means that tin tetrachloride can form complexes with diethyl ether etc.
Tin alkyls are extremely toxic and used to be used to stop algae and crustaceans growing on ships. They can also work as fungicides so wood preserving agents can be used.
When group 14 chemistry is looked at most of the time these are tetravalent compounds. Tin is normally in oxidation state +4, although there are times when tin is only present as a +2. This happens with the halogens.
The purple atoms represent sodium ions trapped in the structure. These are able to move in and out of the channel network.
For zeolite A there are 8 sodalite cages which are linked by 4-rings to give a supercage with and 8 ring entrance. It has a pose size of 4-5 A which makes it highly suitable for water and also for ion exchange. It can be used as a non-phosphate water softener in washing powders. It can also absorb water so can be used as a drying agent.
Other zeolite structures
There are over 100 natural zeolite structures that form, plus 100s of synthetic phase zeolites that have been produced. Some of the more important are ZSM-5 which catalyses carbocationic reactions. This is able to catalyse these reactions as it can stabalise cationic species allowing there transition across its surface. This allows the reaction of benzyl groups to its surface.
Sodalite
Sodalite is a non-pourous arrangement of cuboctahedral, they are cages that contain anions. Soadalite cages contain trapped S3- and S2- radical ions which give a non-fading blue pigment.
Tin
Tin has two main allotropes α tin and β tin, α tin has the same structure to diamond although it is not that stable as tin has lager atomic orbitals this means it usually degrades into a 6 bonded structure above 13˚C.
When tin is warmed a shiny metal is given, when tin is cooled below 13˚C α-tin begins to form which is a powdery structure as compared to β-tin. In the medieval times organ pipes were made of tin, when the winter came in the northern areas of Britain β-tin which is shiny and brilliant, and forms to α-tin and was thought to be caused by the devil.
Tin occurs naturally mainly as cassiterite: SnO2 and can be retained by reduction with carbon at very high temperatures. SnCl4 is tetrahedral in structure and is a Lewis acid (This is due to an expanded valence shell! Not like CCl4!) This means that tin tetrachloride can form complexes with diethyl ether etc.
Tin alkyls are extremely toxic and used to be used to stop algae and crustaceans growing on ships. They can also work as fungicides so wood preserving agents can be used.
When group 14 chemistry is looked at most of the time these are tetravalent compounds. Tin is normally in oxidation state +4, although there are times when tin is only present as a +2. This happens with the halogens.
Lead
Lead does not have a diamond like allotrope. Lead is a metallic element and has well defined Pb2+ cations due to the inert pair effect it is a very strong oxidising agent and can be extracted by burning in air and then reducing with coke:
Lead does not have a diamond like allotrope. Lead is a metallic element and has well defined Pb2+ cations due to the inert pair effect it is a very strong oxidising agent and can be extracted by burning in air and then reducing with coke:
Because Lead has high affinity for sulphur compounds it can bind to proteins in the brain, making it a nerve toxin.
Although Lead (IV) does exist it is not very stable and can actually be used as an oxidising agent.
It is considered a “soft” element. A soft element has a low charge to radius ratio. Lead is a good example of it having a large deformable cation. This means it is normally seen with other “soft” elements. This is normally present as PbS.
Although Lead (IV) does exist it is not very stable and can actually be used as an oxidising agent.
It is considered a “soft” element. A soft element has a low charge to radius ratio. Lead is a good example of it having a large deformable cation. This means it is normally seen with other “soft” elements. This is normally present as PbS.