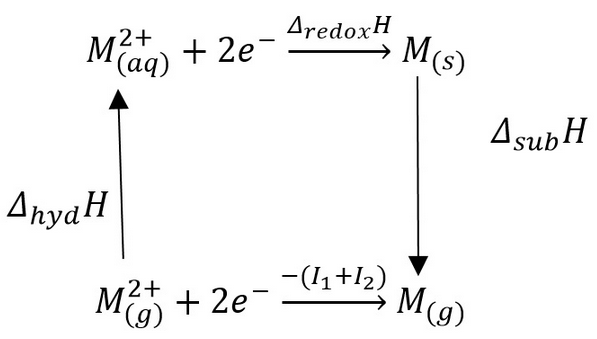A quick revision of all things transition metal: properties, oxidation states and energies.
|
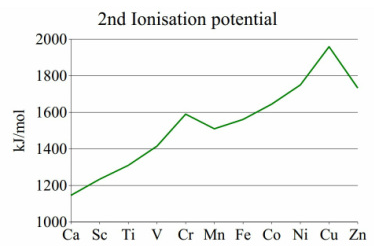
The ionisation potential of transition metals increases across the group. There is a spike found at the atom that has a d5 configuration at that oxidation state, it is easily seen for chromium in the second potential.
This is due to higher effective nuclear charge caused by orbital contraction. |
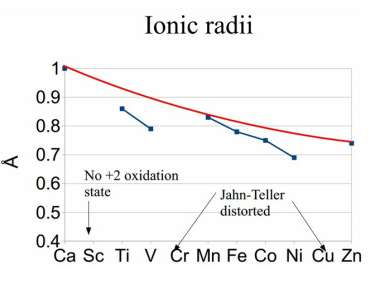
The ionic radii decreases across the period due to increased orbital contraction. The lower orbitals are seen due to partially filled orbitals having a lack of symmetry.
|
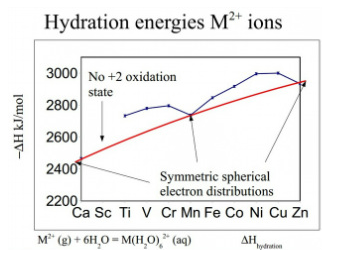
The hydration energies move through a series of humps due to unsymetric orbitals allowing water molecules to move in closer at a higher charge reaching a maximum when water molecules can get close at the higher nuclear charge around nickel but reduces again when the orbital fills further.
|
Oxidation states and energy cycles
|
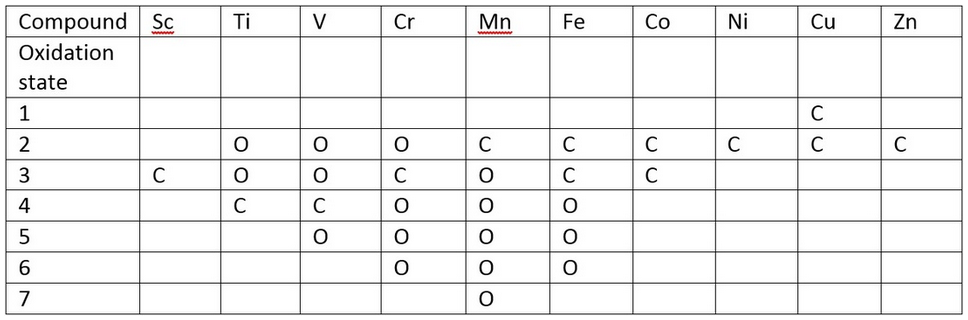
Some oxidation states are more common than others. This affects how hard or soft the ion is.
|
The energy cycle can be used to find other energies involved in the transition metal. This is important as ionisation enthalpies are only found in gas phase atoms. This means that the variation caused by the hydration of the molecule needs to be found to find redox enthalpies.
|
Key points:
As one moves along the d-block:
· Adding to the nuclear charge.
· Poor shielding of d-electrons.
· Increase in the effective nuclear charge.
· Contraction of the d-orbitals into the core.
As one moves along the d-block:
· Adding to the nuclear charge.
· Poor shielding of d-electrons.
· Increase in the effective nuclear charge.
· Contraction of the d-orbitals into the core.