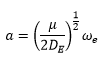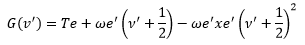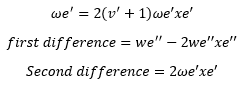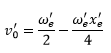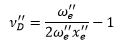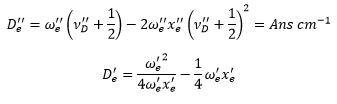These are the more common equations that regularly come up in exams. It is important to have a good grasp of these because when they are given they are usually asked to be explained.
The bond length α is given by:
The energy of the upper state is given by:
This is where Te is the electronic energy, ω is the angular momentum and xe is the anharmonicity. v is the vibrational energy level.
In a selection of excitations at different vibrational levels the following relationships are found between them:
In a selection of excitations at different vibrational levels the following relationships are found between them:
The zero point energy (bottom of the energy graph) can be found by:
And the dissociation energy level is found by:
This is always rounded to the nearest integer.
This can be converted into the dissociation energy by:
This can be converted into the dissociation energy by: