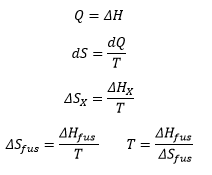Part 1: Single component mixtures
- Phase stability and phase changes
- Pressure - Temperature diagrams (PT - diagrams)
- Phase transitions and special points/regions on the PT-diagram
- Clapeyron Equation - gives the slope of the phase equilibrium lines (dP / dT) in terms of the entropy and volume changes accompanying the transition
- Consider two examples, water and plastic crystals, which each have interesting properties
- Consider Clausius - Clapeyron equation for case of vaporisation
Definitions of Phases
Phase: A form of matter that is uniform throughout in chemical composition and physical state.
Single Phase: A pure gas, a mixture of gases, pure liquid, two fully miscible liquids, sodium chloride solution, metal alloys...
Two-Phase systems: Ice/water slurry, dry ice, two immiscible metals, oil/water mixture...
Phase Transition: Spontaneous conversion from one phase to another at a characteristic temperature and pressure.
Latent Heat and Phase Transitions
Phase: A form of matter that is uniform throughout in chemical composition and physical state.
Single Phase: A pure gas, a mixture of gases, pure liquid, two fully miscible liquids, sodium chloride solution, metal alloys...
Two-Phase systems: Ice/water slurry, dry ice, two immiscible metals, oil/water mixture...
Phase Transition: Spontaneous conversion from one phase to another at a characteristic temperature and pressure.
Latent Heat and Phase Transitions
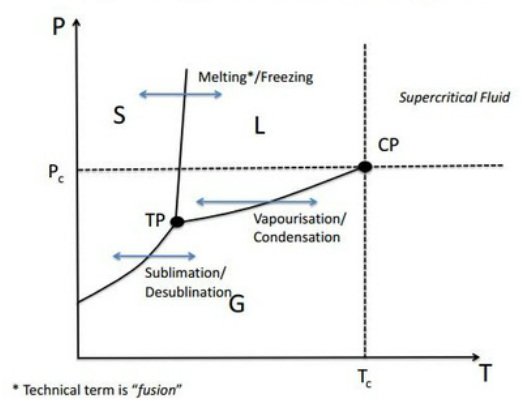
Single Component P-T Phase Diagrams
Definitions relating to Phase Diagrams
Phase Coexistence Line: Two different phases are in thermodynamic equilibrium with one another.
Critical Point: The temperature at which thermal energy overcomes cohesive energy.
Triple Point: A single point on the phase diagram where the solid, liquid and gas phase can coexist in equilibrium with one another.
Supercritical Fluid: Occurs above Tc. No distinction between a liquid and a gas. The fluid fills up all available volume and there is no phase boundary.
Phase Coexistence Line: Two different phases are in thermodynamic equilibrium with one another.
Critical Point: The temperature at which thermal energy overcomes cohesive energy.
Triple Point: A single point on the phase diagram where the solid, liquid and gas phase can coexist in equilibrium with one another.
Supercritical Fluid: Occurs above Tc. No distinction between a liquid and a gas. The fluid fills up all available volume and there is no phase boundary.
Phase transitions
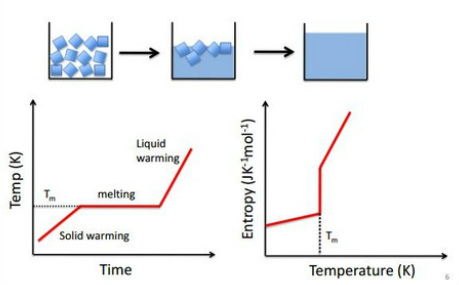
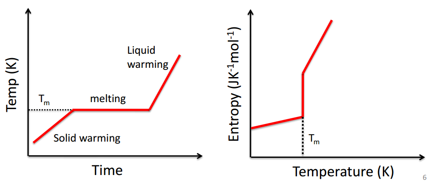
The graph on the left shows that a change in temperature plateaus when the sample is melting this causes a massive increase in entropy as seen on the right. It can also be seen that there is a large increase in the rate that entropy is gained in the liquid form. This is due to the more freedom that the liquid has to move around and therefore become more dispersed with a smaller energy increase. The transition between phases is important as there is a large change in properties with each phase transition.
The graph on the left shows that a change in temperature plateaus when the sample is melting this causes a massive increase in entropy as seen on the right. It can also be seen that there is a large increase in the rate that entropy is gained in the liquid form. This is due to the more freedom that the liquid has to move around and therefore become more dispersed with a smaller energy increase. The transition between phases is important as there is a large change in properties with each phase transition.
Chemical Potential
Is the potential that a substance has to produce a change in a system.
For a one-component system, chemical potential is equivalent to the 'molar Gibbs energy'
It can most easily though of as:
The increase in energy of a system when a small amount of a substance ( a particle) is added with the mass remaining homogeneous and the volume and entropy remaining unchanged.
Is the potential that a substance has to produce a change in a system.
For a one-component system, chemical potential is equivalent to the 'molar Gibbs energy'
It can most easily though of as:
The increase in energy of a system when a small amount of a substance ( a particle) is added with the mass remaining homogeneous and the volume and entropy remaining unchanged.
Thermodynamic Criteria for Phase Stability
Thermodynamic aspects of phase transitions
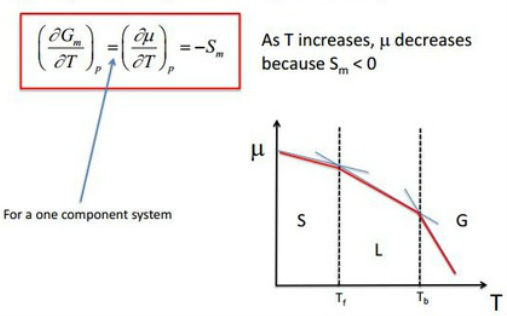
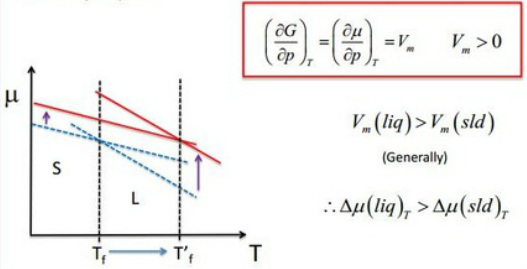
(a) Temperature dependence on phase stability
(a) Temperature dependence on phase stability
(b) Vapour pressure of a liquid subjected pressure
When a pressure is applied to a condensed phase, its vapour pressure increases.
This is because the applied pressure squeexes the molecules out of the condensed phase and into the gas phase.
This can occur through applying a mechanical pressure or by the presence of an inert gas (via increased vapour pressure)
When a pressure is applied to a condensed phase, its vapour pressure increases.
This is because the applied pressure squeexes the molecules out of the condensed phase and into the gas phase.
This can occur through applying a mechanical pressure or by the presence of an inert gas (via increased vapour pressure)
(c) The response of melting to applied pressure
Most substances melt at higher temperature when subjected to pressure.
Increased pressure prevents the formation of the less dense liquid phase.
Most substances melt at higher temperature when subjected to pressure.
Increased pressure prevents the formation of the less dense liquid phase.
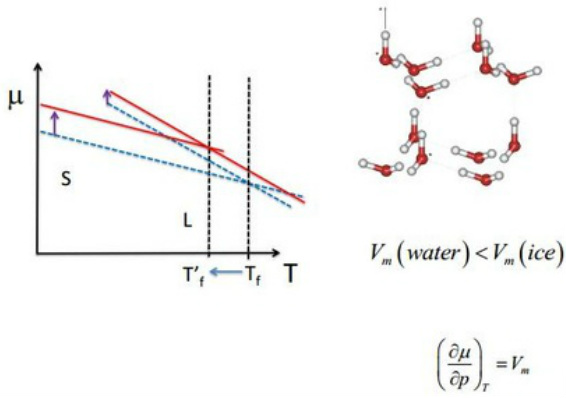
The exception - Water
Water is unique in that the opposite happens.
This is because the structure of ice has an extended H - Bonding network of hexagonal symmetry.
Its Solid phase is less dense than its Liquid phase!
Vm water < Vm ice
Applying pressure to ice, gives us back water!
Graphically
Water is unique in that the opposite happens.
This is because the structure of ice has an extended H - Bonding network of hexagonal symmetry.
Its Solid phase is less dense than its Liquid phase!
Vm water < Vm ice
Applying pressure to ice, gives us back water!
Graphically
Gradient of Phase Coexistence Lines
Clapeyron Equation
Relates the gradient of a phase transition line to fundamental thermodynamic properties. It is important to note that this equation only stands at the phase transition. The easiest way of calculating this is calculating the difference in between the densities of a substance in two different states and then calculating the difference in volume from there.
Clapeyron Equation
Relates the gradient of a phase transition line to fundamental thermodynamic properties. It is important to note that this equation only stands at the phase transition. The easiest way of calculating this is calculating the difference in between the densities of a substance in two different states and then calculating the difference in volume from there.
Reminder
Calculating Differences: It is always final minus initial!
ΔV fus/melt = V liquid - V solid
ΔV freeze = V solid - V liquid
ΔV freeze = - ΔV fus/melt
Calculating Differences: It is always final minus initial!
ΔV fus/melt = V liquid - V solid
ΔV freeze = V solid - V liquid
ΔV freeze = - ΔV fus/melt
Solid Liquid Boundary (fusion)
What happens when ice melts?
V Solid > V Liquid
ΔV fus = V Liquid - V Solid = negative number
ΔS fusion = positive (i.e. the liquid is more disordered)
What happens when ice melts?
V Solid > V Liquid
ΔV fus = V Liquid - V Solid = negative number
ΔS fusion = positive (i.e. the liquid is more disordered)
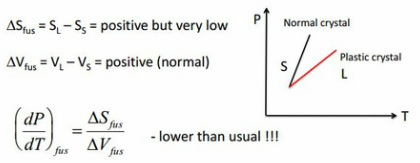
Plastic Crystals
These are solids formed by globular molecules (e.g. cyclohexane)
Relatively weak interaction between molecules (e.g. Van der Waal forces).
They have a high degree of rotational motion in the solid phase (i.e. disorder)
True crystal --> Plastic Crystal --> Liquid
ΔS fusion = S Liquid - S Solid = positive but very low
Molecule ΔS fusion ( J / K / mol)
n - hexane 73
cyclohexane (plastic crystal) 9
These are solids formed by globular molecules (e.g. cyclohexane)
Relatively weak interaction between molecules (e.g. Van der Waal forces).
They have a high degree of rotational motion in the solid phase (i.e. disorder)
True crystal --> Plastic Crystal --> Liquid
ΔS fusion = S Liquid - S Solid = positive but very low
Molecule ΔS fusion ( J / K / mol)
n - hexane 73
cyclohexane (plastic crystal) 9
Gradient of Phase Coexistence Lines - Plastic Crystals
The change in energy given to the material Q is equal to the change in enthalpy of the crystal. The small change in entropy due to the reasons discussed above means the constant enthalpy change needs to be negated by a higher temperature. This high temperature is the reason why plastic crystals react the way that they do.
What about the phase transition temperature (melting point)?
ΔS fus = ΔH fus / T
T = ΔH fus / ΔS fus
Where ΔS fus is abnormally low
Therefore the phase transition temperature (melting point) is abnormally high for plastic crystals
Molecule Melting Point ( 'C / K )
n - hexane - 95 / 168
cyclohexane (plastic crystal) 7 / 280
ΔS fus = ΔH fus / T
T = ΔH fus / ΔS fus
Where ΔS fus is abnormally low
Therefore the phase transition temperature (melting point) is abnormally high for plastic crystals
Molecule Melting Point ( 'C / K )
n - hexane - 95 / 168
cyclohexane (plastic crystal) 7 / 280
Liquid - Vapour boundary (vapourisation)
If V Gas is treated as an ideal gas (for 1 mole)
V Gas = RT / P
ΔV vap = V Gas - V Liquid assuming V Liquid << V Gas
ΔV vap = RT / P and ΔS vap = ΔH vap / T
ΔV vap dP = ΔS vap dT (Derived from Maxwell's relations)
(RT / P) dP = (ΔH vap / T) dT
If V Gas is treated as an ideal gas (for 1 mole)
V Gas = RT / P
ΔV vap = V Gas - V Liquid assuming V Liquid << V Gas
ΔV vap = RT / P and ΔS vap = ΔH vap / T
ΔV vap dP = ΔS vap dT (Derived from Maxwell's relations)
(RT / P) dP = (ΔH vap / T) dT
Clausius Clapeyron Equation
The Clausius Clapeyron equation is used for finding the different pressures between a difference in temperatures or vice versa. This only applies for vapourisation as this is the only point where the gas is present.
The Clausius Clapeyron equation is used for finding the different pressures between a difference in temperatures or vice versa. This only applies for vapourisation as this is the only point where the gas is present.
Summary of Important Concepts: Single component mixtures
- Phase behavior of single component substances can be described by PT phase diagrams. The lines represent phase transitions where two phases are in equilibrium and the chemical potential is the same for both phases.
- The Clapeyron Equation is
- The Clausius - Clapeyron Equation is an approximation that applies only to vapourisation and relates the vapour pressure with the temperature:
|
Topic One: Single Component Mixtures
Topic 2: Thermodynamics of Liquid Mixtures Topic 3: Thermodynamics of Non Ideal Mixtures Topic 4: Two and Three Component Mixtures Topic 5: Colligative Properties |