Topic 3: Thermodynamics of non-ideal liquid mixtures
Derivations from ideality (Raoult's Law) are defined with reference to pressure-composition diagrams and the sign of ΔHmix
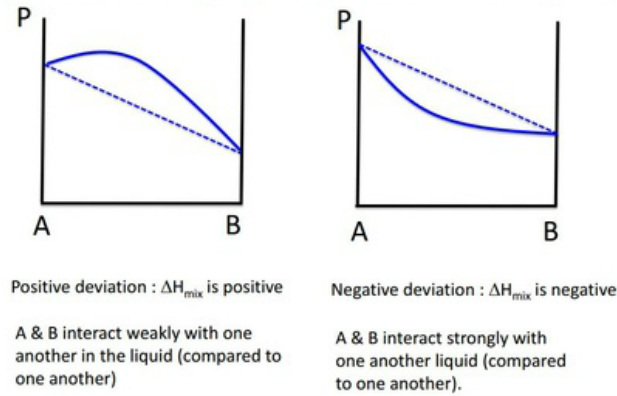
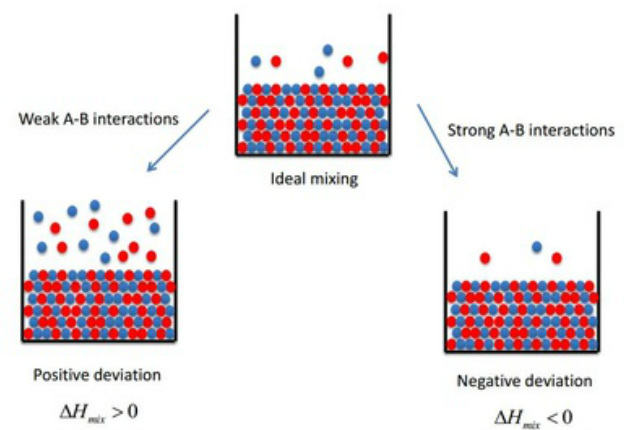
Mixtures with positive deviation from Raoult's Law exhibit a positive ΔHmix (e.g. water and dioxane) - they mix poorly with weak A-B (intermolecular) interactions compared to interactions with one another. This positive change in enthalpy is due to increased repulsion interactions making it less favourable for the liquid state to be present.
Mixtures with negative deviation from Raoult's Law exhibit a negative ΔHmix (e.g. water and ethanol) - they mix well and form strong A-B (intermolecular) interactions compared to interactions with one another. This is due to stabalising interactions between the molecules making a positive
Examples of non-ideal mixtures
Positive deviation, i.e ΔHmix > 0
e.g. water and dioxane, toluene and benzene...
Negative deviation, i.e ΔHmix < 0
e.g. water and ethanol (ethanol can bind to water), acetone and chloroform...
Non-ideal mixtures - departures from Raoult's Law
Mixtures with negative deviation from Raoult's Law exhibit a negative ΔHmix (e.g. water and ethanol) - they mix well and form strong A-B (intermolecular) interactions compared to interactions with one another. This is due to stabalising interactions between the molecules making a positive
Examples of non-ideal mixtures
Positive deviation, i.e ΔHmix > 0
e.g. water and dioxane, toluene and benzene...
Negative deviation, i.e ΔHmix < 0
e.g. water and ethanol (ethanol can bind to water), acetone and chloroform...
Non-ideal mixtures - departures from Raoult's Law
Non-ideal mixtures: graphically
Henry's Law
For dilute solutions: The solute molecules, B, are (on average) completely surrounded by solvent molecules. But solvent molecules are (on average) surrounded by other solvent molecules.
Therefore the vapour pressure of the solute is still proportional to its mole fraction, but not its vapour pressure.
For dilute solutions: The solute molecules, B, are (on average) completely surrounded by solvent molecules. But solvent molecules are (on average) surrounded by other solvent molecules.
Therefore the vapour pressure of the solute is still proportional to its mole fraction, but not its vapour pressure.
In this dilute state, the environment of B does not change with composition.
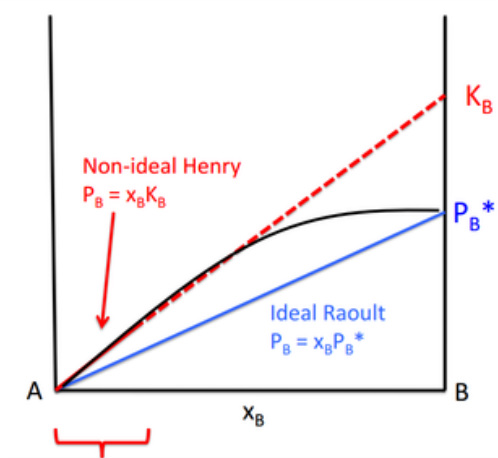
Therefore the vapour pressure of B will be proportional to XB even though mixing is not ideal.
PA = XBKB
KB : Henry Constant: with comparison to Raoult's Law can be thought of as the vapour pressure of B when surrounded completely by A.
KB depends on A, B, and T.
Henry's Law on the pressure - composition diagram
Therefore the vapour pressure of B will be proportional to XB even though mixing is not ideal.
PA = XBKB
KB : Henry Constant: with comparison to Raoult's Law can be thought of as the vapour pressure of B when surrounded completely by A.
KB depends on A, B, and T.
Henry's Law on the pressure - composition diagram
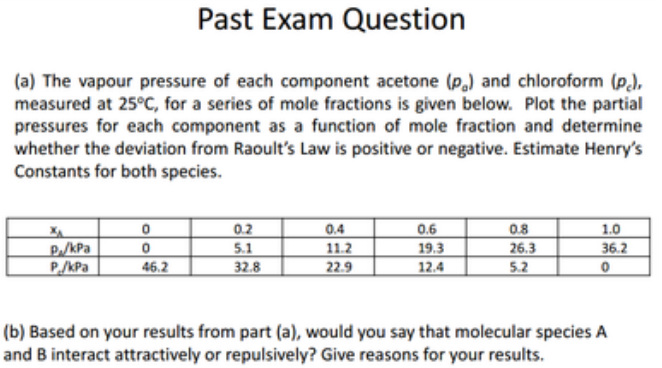
Past Exam Question
Temperature - composition (T - C) diagrams for non-ideal mixtures
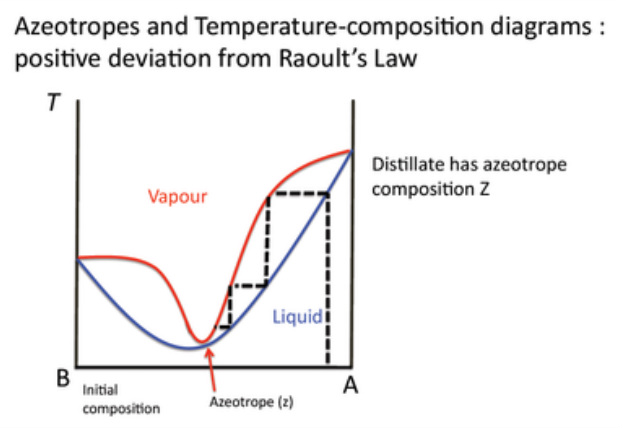
A maximum in the T-C diagram may occur when favourable interactions between A & B occur. This stabilizes the liquid, decreasing the vapour pressure of the mixture below the ideal value, hence increasing the boiling point.
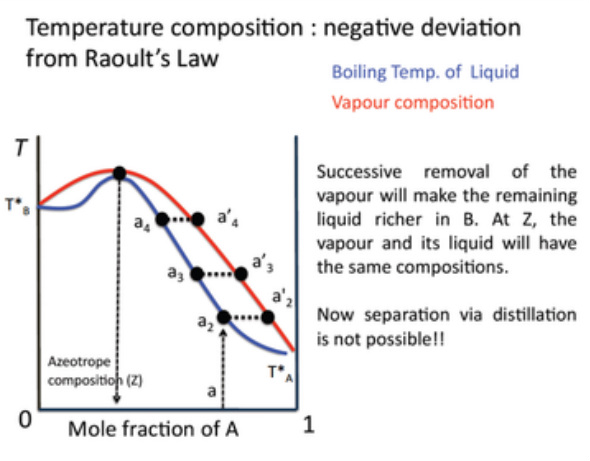
Those showing a minimum mean that the A - B interactions are unfavourable and hence destabilize the liquid, increasing the vapour pressure and therefore lowing the boiling point.
A maximum in the T-C diagram may occur when favourable interactions between A & B occur. This stabilizes the liquid, decreasing the vapour pressure of the mixture below the ideal value, hence increasing the boiling point.
Those showing a minimum mean that the A - B interactions are unfavourable and hence destabilize the liquid, increasing the vapour pressure and therefore lowing the boiling point.
Azeotropes
In non-deal liquid mixtures, such as examples given earlier, the composition of the liquid and vapour phases eventually become the same. At this point it is not possible to separate them through distillation. How else could you separate them?
In non-deal liquid mixtures, such as examples given earlier, the composition of the liquid and vapour phases eventually become the same. At this point it is not possible to separate them through distillation. How else could you separate them?
|
Topic One: Single Component Mixtures
Topic 2: Thermodynamics of Liquid Mixtures Topic 3: Thermodynamics of Non Ideal Mixtures Topic 4: Two and Three Component Mixtures Topic 5: Colligative Properties |