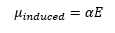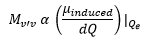Raman spectroscopy is another method which uses a molecules interaction with radiation to quantify certain species present. Raman does not use the absorption of light but rather light's scattering once it interacts with the molecule.
Raman spectroscopy
Raman spectroscopy uses the scattering of light instead of the absorption of light. This is termed Rayleigh scattering. The molecular polarizability is an important part of Rayleigh scattering. This is simple as a fluctuating electric field of an electromagnetic radiation impinging on a molecule is given by E. This forms an induced dipole given by:
Raman spectroscopy uses the scattering of light instead of the absorption of light. This is termed Rayleigh scattering. The molecular polarizability is an important part of Rayleigh scattering. This is simple as a fluctuating electric field of an electromagnetic radiation impinging on a molecule is given by E. This forms an induced dipole given by:
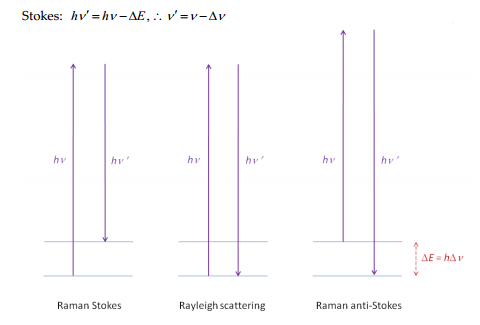
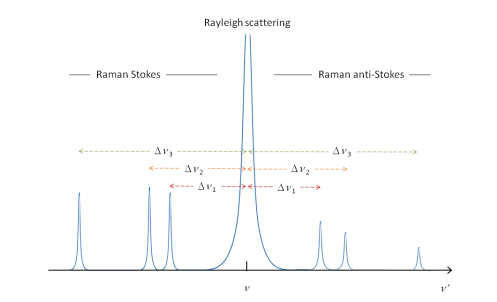
The Raman scattering is measured by changes in the frequency of the light that were characteristic to the sample but not due to fluorescence. The Rayleigh scattering is equal to the induced radiation across the molecule although there are two further groups of lines that are seen in the spectrum. These are termed the Raman Stokes and the Raman anti-Stokes lines.
The Stokes lines do not decrease in intensity as there is a very similar chance of the ground state being at many different points. Whereas the anti-Stokes involves excitation from a high energy state to a different energy state. The high energy state is rarer to occur meaning it decreases across the spectrum.
Raman scattering is a very weak process. The use of high powered lasers and increasingly sensitive detectors has made Raman spectroscopy a much better technique.
In principle Raman transitions can occur by all of the different energy processes, electronic, vibronic and rotational. Although vibronic is the most common. Evaluation of the transition dipole moment connecting states with quantum numbers v and v’ reveals that:
In principle Raman transitions can occur by all of the different energy processes, electronic, vibronic and rotational. Although vibronic is the most common. Evaluation of the transition dipole moment connecting states with quantum numbers v and v’ reveals that:
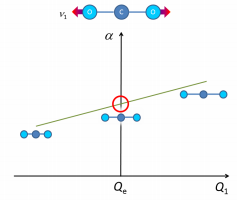
The importance on whether the change in polarizability changes uniformly during the symmetric stretch and the slope is non-zero at the equilibrium configuration.
If the polarizability changes in the course of a given vibration, as the molecule passes through its equilibrium configuration that vibration will be Raman active.
This means that the magnitude of the polarizability needs to be looked at during the transition. If the magnitude is equal on either sides of the vibration then the molecule is Raman inactive. If they are non-equal then the molecule is Raman active. A Raman active stretch is shown below:
If the polarizability changes in the course of a given vibration, as the molecule passes through its equilibrium configuration that vibration will be Raman active.
This means that the magnitude of the polarizability needs to be looked at during the transition. If the magnitude is equal on either sides of the vibration then the molecule is Raman inactive. If they are non-equal then the molecule is Raman active. A Raman active stretch is shown below:
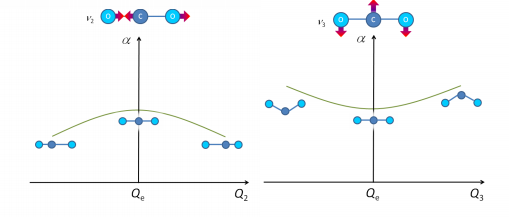
Below shows that the magnitude of the polarisability is the same on either side of the vibration. This means that this transition is Raman inactive, note it is IR active.
This shows further the mutual exculsion rule for centrosymmetric molecules. It can be seen that the IR active stretches are Raman forbidden. This is better seen by saying that Raman active modes must be of gerade (g) character and IR active must be of ungerade (u) character.