The stereochemistry of the carbonyl group
The stereochemistry of the carbonyl group, although not that in depth or long, is very important to recognise and understand.
Introduction:- stereochemisty of reactions
The geometry of the reactions of carbonyl groups are important and has both an effect for straight chain carbonyls and carbonyls in the ring formation.
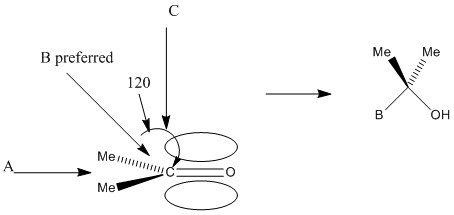
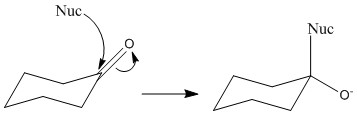
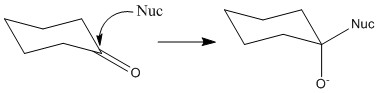
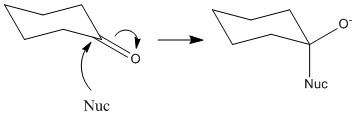
The carbonyl group is entirely planar with bond angles of 120°, this means that the ring is equally likely to be attacked from either face. The angle of attack normally taken is 109° to the carbonyl centre. This is important as it minimises rearrangement. Only 3 of the groups will move to 109° from 120° rather than all 4.
This can be shown by the preference of B. Here although C is not sterically hindered it is attacking an area of high electron density. This means that in most cases it bounces off and doesn’t achieve any bond formation. A has the obvious steric hindrance of the two groups and so is therefore not favoured.
The stereochemistry of the carbonyl group, although not that in depth or long, is very important to recognise and understand.
Introduction:- stereochemisty of reactions
The geometry of the reactions of carbonyl groups are important and has both an effect for straight chain carbonyls and carbonyls in the ring formation.
The carbonyl group is entirely planar with bond angles of 120°, this means that the ring is equally likely to be attacked from either face. The angle of attack normally taken is 109° to the carbonyl centre. This is important as it minimises rearrangement. Only 3 of the groups will move to 109° from 120° rather than all 4.
This can be shown by the preference of B. Here although C is not sterically hindered it is attacking an area of high electron density. This means that in most cases it bounces off and doesn’t achieve any bond formation. A has the obvious steric hindrance of the two groups and so is therefore not favoured.
When this occurs on a carbonyl that is attached to a ring the reaction direction makes a large difference to the product formed.
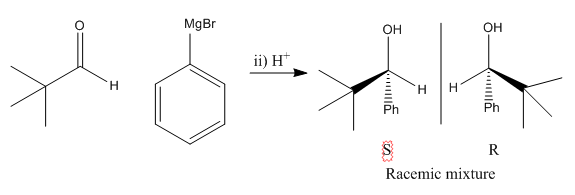
When a reaction occurs there is an equal possibility for top face or bottom face attack to occur. When an un-symettrical ketone reacts there is a formation of a racemic mixture by this fact. It is important to remember that this reaction only occurs through top or bottom reaction. The substituents on the ketone do not make one of these more favourable over another.
When a reaction occurs there is an equal possibility for top face or bottom face attack to occur. When an un-symettrical ketone reacts there is a formation of a racemic mixture by this fact. It is important to remember that this reaction only occurs through top or bottom reaction. The substituents on the ketone do not make one of these more favourable over another.
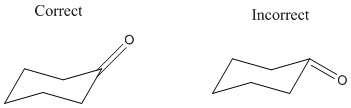
The carbonyl group attached to the ring is kept in a neither axial nor equatorial state but rather between the two, it is important that is kept in the same plane as shown below:
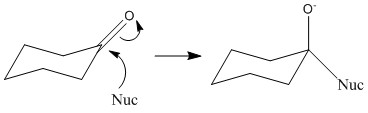
When nucleophilic addition occurs the carbonyl group is pushed into a different position depending on the angle of attack. An attack from below will push the group up but an attack from the top will push the group down. It is important to remember that the oxygen can end in either the equatorial or the axial position.
A series of example are shown below:
A series of example are shown below:
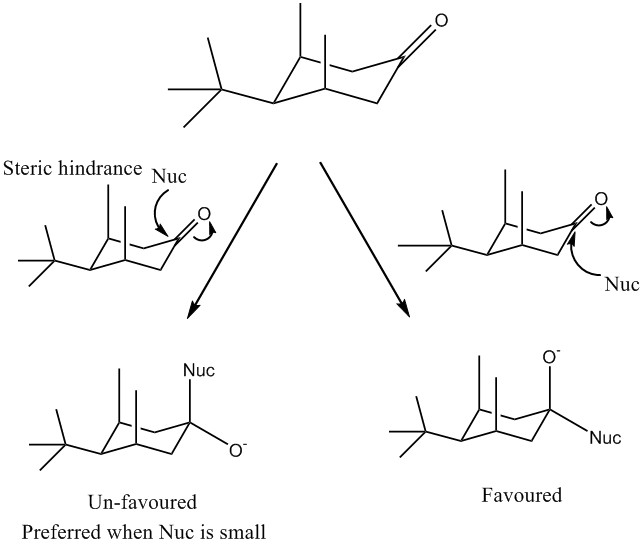
The compound below has a tert-butyl group attached. This means that it is locked in this configuration and therefore can’t undergo any form of ring flip to a potentially more favourable formation. In this reaction the nucleophile can attack from either above or below the ring. The favoured product is where the nucleophile attacks from the equatorial position. This is due to the lack of steric hindrance from the methyl groups which occur when attacked from the axial position. The axial addition is only possible for small nucleophiles. In which case axial attack is preferred as it pushes the oxygen group into the equatorial position.
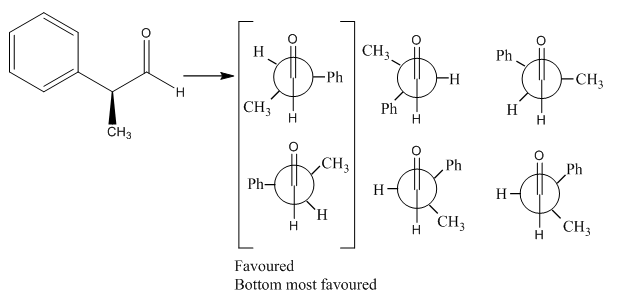
Crams rule
Crams rule is an important aspect of understanding how the configuration of carbonyl group affects how the reaction occurs and how the stereochemistry of the product is affected. This is carried out by ranking the subsitutent groups in size in comparison to one another. The torsional interactions that are present due to the presence of the carbonyl group forces the most stable configuration where the large group is perpendicular to the carbonyl group.
The positions of the groups depending on the how the most favoured configuration can occur. This can be shown in the following diagrams:
Crams rule is an important aspect of understanding how the configuration of carbonyl group affects how the reaction occurs and how the stereochemistry of the product is affected. This is carried out by ranking the subsitutent groups in size in comparison to one another. The torsional interactions that are present due to the presence of the carbonyl group forces the most stable configuration where the large group is perpendicular to the carbonyl group.
The positions of the groups depending on the how the most favoured configuration can occur. This can be shown in the following diagrams:
This means that the two more favoured conformations will be present in a higher amount than the other conformations. This means that the nucleophile will tend to attack from the least sterically hindered direction while still maintaining the necessary 109.5° angle.
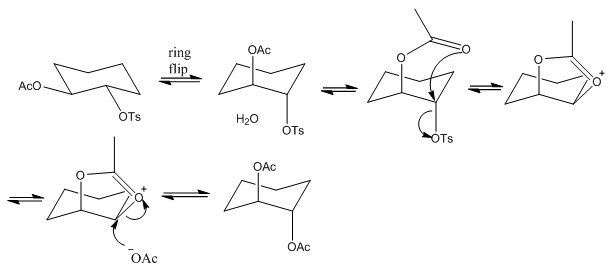
Neighbouring Group Participation or NGP
Neighbouring group participation or NGP is known as anchimetric assistance. This is where a reaction occurs at a much faster rate due to the neighbouring groups close to the functional group. When looking for these reactions homonuclear molecules tend to have the greatest effect.
Neighbouring group participation or NGP is known as anchimetric assistance. This is where a reaction occurs at a much faster rate due to the neighbouring groups close to the functional group. When looking for these reactions homonuclear molecules tend to have the greatest effect.
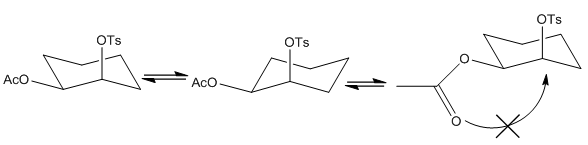
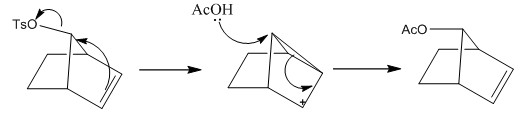
This shows the importance that both attached molecules have the same stereochemistry and therefore allow the interaction between the acetone group and the tosile group.
When the groups are in different positions (equatorial or axial) to one another then the reaction cannot occur as the electron donation is too far away to be effective. This means that there is no acceleration in the reaction although the substitution may still take place slowly.
NGP chemistry and its effect on stereochemistry
When an SN2 reaction occurs there is usually stereochemical inversion at the reacting group. This occurs due to the attacking group attacking at a 180° angle to the position. This doesn’t occur in NGP chemistry as there are the equivalent of two SN2 reactions occurring even if the first one if uni-molecular it is with two different sections of the same molecule. With all of these reactions there is an electron dense species next to a very strong leaving group. Tosulate is one of the most used examples. This is a very stable leaving group and can easily be taken off by the electron density of an alkene group.
When an SN2 reaction occurs there is usually stereochemical inversion at the reacting group. This occurs due to the attacking group attacking at a 180° angle to the position. This doesn’t occur in NGP chemistry as there are the equivalent of two SN2 reactions occurring even if the first one if uni-molecular it is with two different sections of the same molecule. With all of these reactions there is an electron dense species next to a very strong leaving group. Tosulate is one of the most used examples. This is a very stable leaving group and can easily be taken off by the electron density of an alkene group.
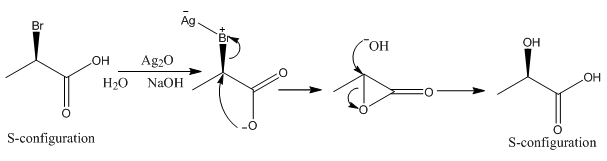
In the reaction above the sodium hydroxide is only added in enough concentration to cause the loss of the proton from the carboxylic acid group. This can then donate its electron density causing the loss of the bromide group which is now a stable leaving group. This forms an unstable three membered ring which can quickly react with the hydroxide ions left. This is a much faster reaction than the addition of hydroxide in the first instance.
The alkene group in this example only forms in the same place according to Bredt’s rule it cannot form on either of the two bridging carbons as this has too much angle strain.
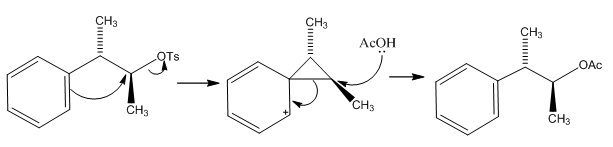
This example shows how a ring can be formed and then attacked with an acetone group. This is quite usual and doesn’t come with any stereochemical changes although it is worth noting that when there is a more stable carbo cation to be formed the attack may occur at a different part of the ring causing a different configuration:
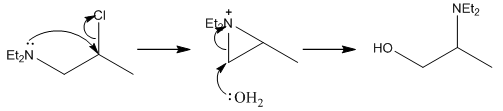
Here the water molecule has attacked the more exposed carbon causing the amine group to shift into the secondary position. This shows that when the substitution is slow at the secondary position that a different mechanism can occur so the substitution actually occurs in the primary position but to a better leaving group.
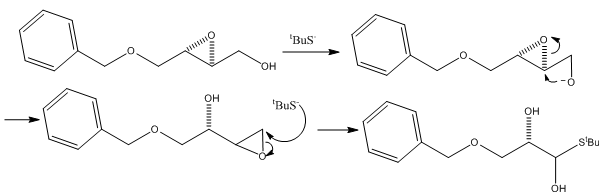
Payne rearrangement
Here the substitution of an epoxide ring occurs. This can be produced when there are a number of hydroxide groups are present. This can then be treated with base following formation of the epoxide group which can then allow the formation of a second epoxide group. This forces the movement of the movement of groups.
Payne rearrangement
Here the substitution of an epoxide ring occurs. This can be produced when there are a number of hydroxide groups are present. This can then be treated with base following formation of the epoxide group which can then allow the formation of a second epoxide group. This forces the movement of the movement of groups.
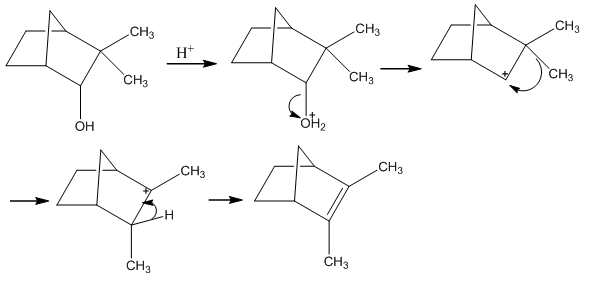
Wagner-Meewein Rearrangements
The Wagner-Meerwein rearrangements are a series of rearrangements that are based on producing the most stable carbocation possible.
The Wagner-Meerwein rearrangements are a series of rearrangements that are based on producing the most stable carbocation possible.
In the first example the hydride shift doesn’t occur as it is very unfavourable to produce a carbocation on a bridging carbon group. When tackling these problems it is important to look for any quaternary carbons as these are able to produce a tertiary carbon with increased stability.
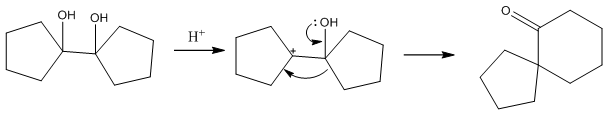
Pinacol rearrangements
The pinacol rearrangement is a very common rearrangement that occurs when there are two hydroxide groups adjacent to one another. This can then be acidified causing the loss of a water molecule leading to a methyl shift. This is an important reaction in ring forming.
The pinacol rearrangement is a very common rearrangement that occurs when there are two hydroxide groups adjacent to one another. This can then be acidified causing the loss of a water molecule leading to a methyl shift. This is an important reaction in ring forming.
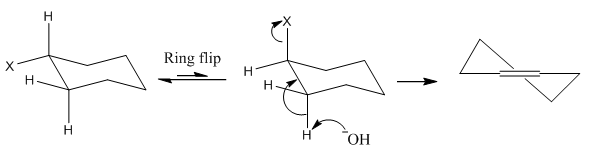
E2 reaction
E2 reactions involve the elimination of two groups form a molecule forming an carbon double bond. This is where there is an attack on the hydrogen group where a leaving group is antiperiplanar to the hydrogen group. As noted the leaving groups need to be antiperiplanar to one another which is rarely the favourable formation for a group.
E2 reactions involve the elimination of two groups form a molecule forming an carbon double bond. This is where there is an attack on the hydrogen group where a leaving group is antiperiplanar to the hydrogen group. As noted the leaving groups need to be antiperiplanar to one another which is rarely the favourable formation for a group.
It is worth remembering the differences in stabilities of the products formed in these reactions. It is much more favourable for there to be the formation of a tertiary alkene over a primary or secondary alkene.
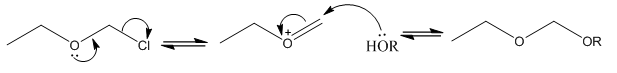
Solvolysis reactions
The SN1 ionisation and SN2 direct displacement reactions are two extremes that are met, the overlap of these mechanisms are the ion-pair reactions. These involve the intermediate being seen as a positively charged and a negatively charged ion. These can be stabilised in different ways, the solvent used in the reaction is a big example of this.
The solvent used in the reaction can actually produce a different mechanism for the reaction. This is where for the SN1 reactions there is an increase in the solvent polarity there is an increase in reaction rate this is explained by stabilizing the charged transition state. For SN2 reactions there is little effect. The increase in solvent polarity and ion solvating ability may change the reaction from SN1 to SN2.
For solvolysis reactions the structure of the halide and the stability of the carbocation formed are closely linked. It is also important to take into account how stable the carbocation formed in the reaction is. The more stable the carbocation the more racemation as the reaction can equilibrate more successfully when the solvent is more polar.
Solvolysis reactions
The SN1 ionisation and SN2 direct displacement reactions are two extremes that are met, the overlap of these mechanisms are the ion-pair reactions. These involve the intermediate being seen as a positively charged and a negatively charged ion. These can be stabilised in different ways, the solvent used in the reaction is a big example of this.
The solvent used in the reaction can actually produce a different mechanism for the reaction. This is where for the SN1 reactions there is an increase in the solvent polarity there is an increase in reaction rate this is explained by stabilizing the charged transition state. For SN2 reactions there is little effect. The increase in solvent polarity and ion solvating ability may change the reaction from SN1 to SN2.
For solvolysis reactions the structure of the halide and the stability of the carbocation formed are closely linked. It is also important to take into account how stable the carbocation formed in the reaction is. The more stable the carbocation the more racemation as the reaction can equilibrate more successfully when the solvent is more polar.