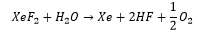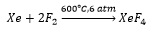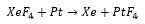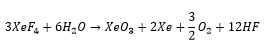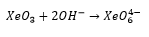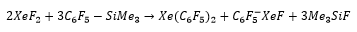Group 18: The noble gases
The chemistry of the noble gases is somewhat limited due to the inherent inactivity of the gases due to the full electron shells. These cause the molecules to be very un-reactive, but they still have trends going down the group and the final noble gases can still form compounds due to their polarisability. The high electron densities of these compounds allow the most electronegative elements to form bonds with the larger groups such as xenon.
Noble gases form clathrates with hydroquinone under high pressures these clathrates are just large cages of hydrogen bonds with the polarisaed molecule forming a structure that has the ability to store the noble gases. These can even store the radioactive isotopes of these compounds.
Helium
Helium is one of the most used noble gases. It is very unreactive and it is a valuable resource. It is a by-product of nuclear fission and natural gas production. It is needed for low temperature devices such as NMR spectroscopy and magnetic resonance imaging. Helium can diffuse through rubber and glass.
Argon
Argon is made through separation of air, which can be fractionally distilled away from it. It is a relatively heavy inert gas making it useful for blanket coverage of a reaction vessel.
Xenon
Xenon reacts with fluorine at 400°C and 1atm of pressure, this forms a crystalline solid showing how the high electron density has such a large effect the negativity of fluorine allows the formation of bonds. This is a strongly oxidising agent.
The chemistry of the noble gases is somewhat limited due to the inherent inactivity of the gases due to the full electron shells. These cause the molecules to be very un-reactive, but they still have trends going down the group and the final noble gases can still form compounds due to their polarisability. The high electron densities of these compounds allow the most electronegative elements to form bonds with the larger groups such as xenon.
Noble gases form clathrates with hydroquinone under high pressures these clathrates are just large cages of hydrogen bonds with the polarisaed molecule forming a structure that has the ability to store the noble gases. These can even store the radioactive isotopes of these compounds.
Helium
Helium is one of the most used noble gases. It is very unreactive and it is a valuable resource. It is a by-product of nuclear fission and natural gas production. It is needed for low temperature devices such as NMR spectroscopy and magnetic resonance imaging. Helium can diffuse through rubber and glass.
Argon
Argon is made through separation of air, which can be fractionally distilled away from it. It is a relatively heavy inert gas making it useful for blanket coverage of a reaction vessel.
Xenon
Xenon reacts with fluorine at 400°C and 1atm of pressure, this forms a crystalline solid showing how the high electron density has such a large effect the negativity of fluorine allows the formation of bonds. This is a strongly oxidising agent.
This can be further reacted forming a pseudo octahedral complex
This is an even stronger fluorinating agent than .
There is the possibility for the formation of Xenon oxide compounds. Which can be carried out using a flouronating agent with water. These form very unstable complexes mostly being explosive.
With increased addition of hydroxide ions this can form a perxenate anion:
Xenon can also bind to alkyl fluoride groups with a large electron density.
This leaves the highly unexpected (and apparently cool complex) gold (II) xenon ion.