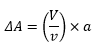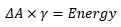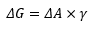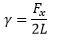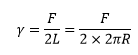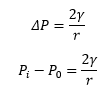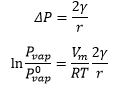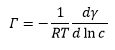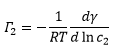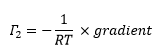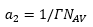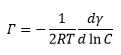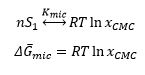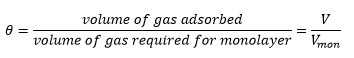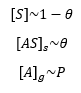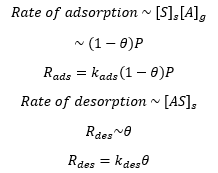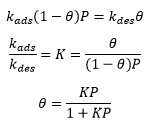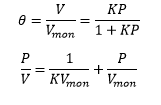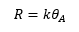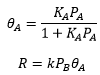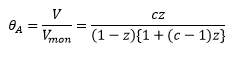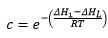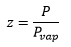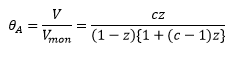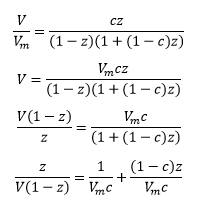Surface chemistry is a highly important aspect of the chemical sciences. It governs a large number of interactions that are seen every day as well as important lab processes such as catalysis. Many systems present are heterogeneous, multiphase and highly dispersed.
Introduction
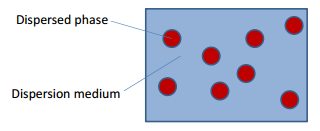
When a system is heterogeneous:- meaning more than one state of matter is found within it, and when the dimensions of the phase of interest are in the range of 1-1000 nm and have a high surface area they are described as colloids.
It is important to understand these systems as there structure can have a large influence on many different reactions.
Classification
Determined by the amount of dispersion of a system:
The dispersed phase in a dispersion medium.
When a system is heterogeneous:- meaning more than one state of matter is found within it, and when the dimensions of the phase of interest are in the range of 1-1000 nm and have a high surface area they are described as colloids.
It is important to understand these systems as there structure can have a large influence on many different reactions.
Classification
Determined by the amount of dispersion of a system:
The dispersed phase in a dispersion medium.
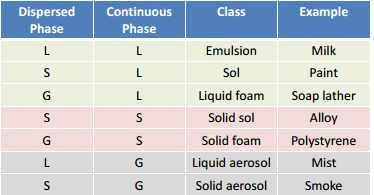
As both the dispersed phase and the dipersion medium can be in any number of states there are distinct classes for each of these.
This shows how the dispersed phase can be in the same phase as the dispersion medium. The examples shown should be understood and give a number of everyday systems that represent a colloid system.
|
Emulsions and microemulsions
Emulsions and microemulsions are two very important aspects of the surface chemistry. The basis of setting up an emulsion involves the stable suspension of an immiscible dispersed phase in a dispersion medium. This can be stabilised by a surfactant. These are usually lipid like materials that have a hydrophobic and hydrophilic section to allow interactions with both the medium and the phase. A nanoemulsion is a type of emulsion in which the sizes of the particles in the dispersed phase are defined as less than 10 nm. This is important as this takes the sample into the quantum dot range. Quantum dots can have a number of different uses, a common one is the ability to produce significant changes in the energy levels to allow different absorption and even fluorescence patterns to be produced. Microemulsions are mixtures of oil, water and surfactant which are usually less than 100 nm in size. They tend to be clear, isotropic (meaning it is evenly spread,) and thermodynamically stable (meaning it can be left for a long time without separating). Miniemulsions are obtained by shearing a miture comprising of two immiscible liquid phases and a surfactant, these tend to be thermodynamically unstable. They have a large droplet size from 100-1000 nm which makes the isotropic factor un-clear. They tend to scatter light and are often quite turbid. |
Sols
Sols are passed on rapid nucleation and slow particle growth. There is a great importance on the speed of particle growth and what temperature this occurs at. At high temperatures it is possible for nucleation to be rapid and growth to be rapid forming a low molecular mass as there is a large amount of nucleation. In a reaction vessel there can be monodisperse meaning they are all the same size and polydisperse meaning they are at a range of sizes. These can be stabilised with ions or with long chain molecules. Long chain molecules can not only stabilise the colloid but also allow stabilisation against oxidation effects etc. This occurs through forming a kinetic barrier between the solvent and the molecule being suspended in the solution. When it comes to the formation of a sol, the drop in temperature needs to be slow as a decrease in temperature causes a decrease in volume. A rapid decrease in temperature causes a too rapid decrease in volume. This causes the sol to collapse. When the temperature is reduced slowly the volume is kept high and density kept low. |
Fragmentation of a material
There is great importance on the size of particles in colloid chemistry and how the dispersion of a particle and the change in surface area due to this change.
There is great importance on the size of particles in colloid chemistry and how the dispersion of a particle and the change in surface area due to this change.
This is where V is the volume of the larger particle and v is the volume of the dispersed particle, A is the change in surface area and a is the surface area of the dispersed particle. This is closely related to the surface energy of the solid. Represented by ΔG.
The energy required to create this dispersed medium is based on the surface energy and the change in surface area:
The energy required to create this dispersed medium is based on the surface energy and the change in surface area:
The liquid-gas surface
The surface tension of the liquids is affected by the surface pressure and the pressure of the materials around the surface. The difference in pressure across a curved surface causes a change in the vapour pressure of the liquid. These effects bring about a number of unusual properties arising where highly curved surfaces are present, like those in bubbles.
Laplace equation goes onto explain how a capillary rise forms and how the stability in a capillary can be seen. Initially a better understanding of surface tension needs to be had first.
The energy required to expand surface
The surface tension of the liquids is affected by the surface pressure and the pressure of the materials around the surface. The difference in pressure across a curved surface causes a change in the vapour pressure of the liquid. These effects bring about a number of unusual properties arising where highly curved surfaces are present, like those in bubbles.
Laplace equation goes onto explain how a capillary rise forms and how the stability in a capillary can be seen. Initially a better understanding of surface tension needs to be had first.
The energy required to expand surface
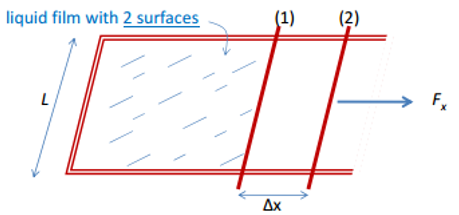
It is important that the surface formed is of minimum surface area, this is why droplets are formed. The liquids surface expansion term can be considered as: Energy per unit area or the force per unit length. This can be measured in the liquid film with two surfaces.
This surface energy is calculated by the force and twice the length due to the face that there are two surfaces formed in this test.
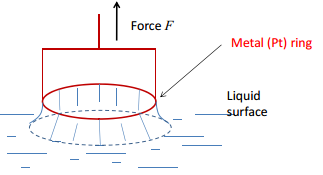
This can also be carried out by a measurement with a platinum ring. This is a highly effective measurement to carry out as this can give the force constant of many different materials.
The radius and the force involved has the following relationship:
Laplace equation
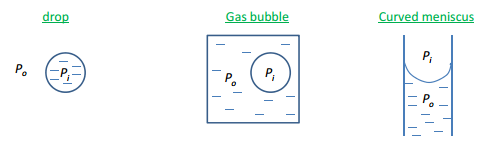
The difference across the curved surface needs to be calculated this involves taking into count the difference in pressure of the inside and outside of the curved surfaces. This can be seen on what direction the meniscus is curved into.
The difference across the curved surface needs to be calculated this involves taking into count the difference in pressure of the inside and outside of the curved surfaces. This can be seen on what direction the meniscus is curved into.
In all these situations pi is greater than p0. The pi value is always on the “inside” of the curved surface, this means that the larger pressure is causing the deviation which makes a big difference to the structure.
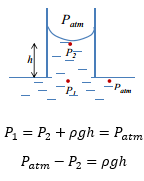
The difference in the pressures causes a capillary rise on either side, this causes the rise of the liquid with more pressure meaning this moves into the lower density space.
At non-equilibrium the liquid moves up the column with a pressure related to the surface tension of the liquid.
At non-equilibrium the liquid moves up the column with a pressure related to the surface tension of the liquid.
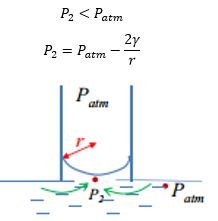
It is important to remember that the flow of materials is towards the low pressure area. This means that the pressure difference then takes effect on the different pressures present. This means that the flow of materials stops.
This means that at equilibrium in this movement.
This means that at equilibrium in this movement.
This shows how the formation of these equations are made from knowing that Patm is bigger than P2 and as the example above is in equailibrium the pressure at P1 is equal to that of P2.
|
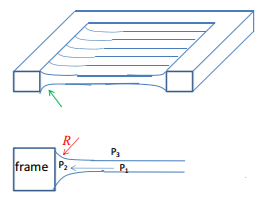
Capillary pressure
Where there is a thin film there will be a number of different pressures that need to be taken into account, and example is given below: This shows how at the flat surface P3=P1 as there is no meniscus so no difference in pressure. Where there is a curvature is can be seen that P3>P2 due to the fact that the liquid is curved. And P3 is in the centre.
|
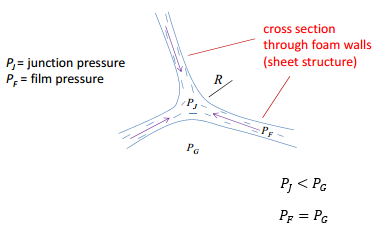
Foam stability
When a foam is produced. It is rare that a flat surface is formed there are almost always curved areas where there is a difference in pressure this means that the liquid flows from the sections of equal pressure to the sections of un-equal pressure. This causes the foam to retract over time. This causes liquid flow into the area designated PJ.
|
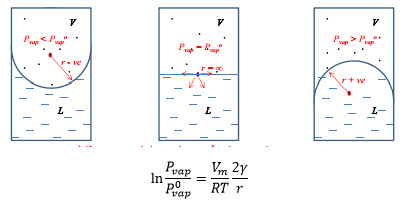
Kelvin equation
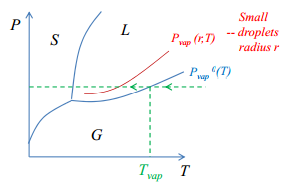
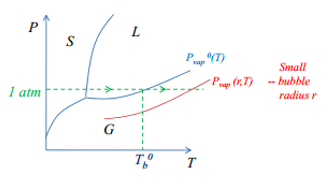
Depending on where the vapour pressure of the liquid is in comparison to the vapour pressure at a flat surface is shown in the following diagrams.
Depending on where the vapour pressure of the liquid is in comparison to the vapour pressure at a flat surface is shown in the following diagrams.
|
Super cooling
The consequence of the kelvin equation is that the supercoiling vapour state of a material can be produced. This is possible by increasing the vapour pressure which means that the material leaves the liquid phase quicker becoming a gas. This means that as soon as a flat surface is formed spontaneous condensation occurs. · Small droplets higher vapour pressure. · Cooling enters normal liquid region below Tvap- super cooling. · On formation of a flat surface spontaneous condensation occurs. |
Super heating
The super heating of a liquid causes bumping, this is where a bubble or a cavity which have Pvap<P0vap the liquid can be heated well above its normal boiling point. This system is stable but if a flat surface forms such as that of a large bubble the vaporisation is spontaneous. This forms a large cavity which can erupt. · Because small bubble cavities have Pvap < Pvap0 liquid can be heated above its normal boiling point. · System is then metastable if “flat” liquid surface forms vaporisation is spontaneous. Bumping
Bumping is the process where the super heating liquid vaporises. When liquid is super heated it can form a gas, this gas forms a bubble which is usually too small to support itself, which means the atmospheric pressure causes the fluid to crush the bubble. When there is a large cavity the compression causes rapid expansion of the gas forming a bumping effect. |
|
Evaporation/ condensation – liquid transfer
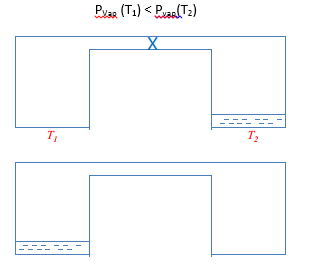
When there is a favourable environment for a liquid to move into it shall do so through evaporation and then condensation into the other environment. This makes a lot of sense when there are two different environments that are linked together. It is seen that the liquid will evaporate from one and then condense in the cooler section. This is due to the differences in vapour pressures. |
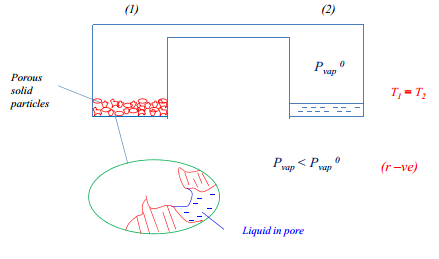
Another action is the use of capillary condensation. This is where there are small pores which have a varying vapour pressure due to the presence of surface tension. This creates a more favourable surface as the radius is negative and therefore the vapour pressure here will be less than the vapour pressure in the non-porous area.
|
Ostwald Ripening of emulsions
This is based on the vapour pressure of small droplets being higher than larger droplets. This means that there is an eventual evaporation of these smaller droplets which condense onto the larger droplets, this means that the larger droplets slowly gain mass while the smaller droplets disappear. This happens without contact being made between the two surfaces.
This is based on the vapour pressure of small droplets being higher than larger droplets. This means that there is an eventual evaporation of these smaller droplets which condense onto the larger droplets, this means that the larger droplets slowly gain mass while the smaller droplets disappear. This happens without contact being made between the two surfaces.
Liquid-Gas surface
The Laplace equation and the Kelvin equation are very important here:
The Laplace equation and the Kelvin equation are very important here:
The adsorption at a liquid interface is related to the surface tension of the liquid and facilitates the formation of high surface systems. Where a molecule is a surfactant this will have a hydrophobic tail and a hydrophilic head. If the surfactant is present in the water the tail will stick out wards into the air and the head will remain in the water. This collection of surfactant on the surface effects the surface tension.
This is where Γ is the concentration of molecules on the surface interface and c is the concentration of the surfactant on the surface. This is closely related to micelle formation.
Liquid gas surface
Molecules that selectively adsorb are called surface active. This is where an equilibrium exists between molecule in bulk liquid and at a surface. The adsorbed solute surface has a surface concentration of Γ2 (mol m-2) which is in equilibrium with solute that has been dissolved entirely at a concentration of C2 (mol m-3).
The surface (excess) concentration is the number of moles of the surfactant adsorbed at surface per unit area of surface. Gibbs adsorption equation is related to the difference in the surface tension caused by the concentration of the material at the surface and in the solution.
Liquid gas surface
Molecules that selectively adsorb are called surface active. This is where an equilibrium exists between molecule in bulk liquid and at a surface. The adsorbed solute surface has a surface concentration of Γ2 (mol m-2) which is in equilibrium with solute that has been dissolved entirely at a concentration of C2 (mol m-3).
The surface (excess) concentration is the number of moles of the surfactant adsorbed at surface per unit area of surface. Gibbs adsorption equation is related to the difference in the surface tension caused by the concentration of the material at the surface and in the solution.
This can then allow the two measureable values in this equation, the concentration and the surface tension to be plotted against one another. This gives the concentration at the surface to be calculated which is equal to:
This means that the surface concentration can be calculated for a liquid gas interface through a calculation of the γ whereas Γ can be calculated from the solid gas interface straight from the direct measurement of Γ.
The area per molecule adsorbed onto an interface is given by:
The area per molecule adsorbed onto an interface is given by:
This makes sense as the surface concentration given in (mol m-2) can be multiplied by avregados number (molecules mole-1) to get the total surface area of each molecule (m2 molecule). This only takes into account a single surfactant present. Although when the solute is an ion both ions need to be taken into account. Both the positive and the negative charge will reside on the surface:
The ½ value takes into account the presence of two ions.
Micelles
Micelles are surfactants that engage in a self assembly mechanism to form large aggregated structures in a solution. These structures are fundamental to detergency and are also important in a wide range of systems. This free energy for transfer of a surfactant molecule into a micelle can be obtained from the CMC “critical micelle concentration” this is where micelle formation begins to form inside of the liquid because the surface has become “full”.
Above the CMC there is the slow formation of micelles in the solution. The concentration on the surface does not change as no more of the micelle monomers can be fitted onto the surface. does not change due to the surface being saturated. This is where the monomers begin to form micelles in the solution.
The free energy can be shown by the equilibrium below with the free energy of my cell formation related to the mole fraction of surfactant at the CMC.
Micelles
Micelles are surfactants that engage in a self assembly mechanism to form large aggregated structures in a solution. These structures are fundamental to detergency and are also important in a wide range of systems. This free energy for transfer of a surfactant molecule into a micelle can be obtained from the CMC “critical micelle concentration” this is where micelle formation begins to form inside of the liquid because the surface has become “full”.
Above the CMC there is the slow formation of micelles in the solution. The concentration on the surface does not change as no more of the micelle monomers can be fitted onto the surface. does not change due to the surface being saturated. This is where the monomers begin to form micelles in the solution.
The free energy can be shown by the equilibrium below with the free energy of my cell formation related to the mole fraction of surfactant at the CMC.
The typical value for the CMC is around 10-5 – 10-2 mol dm-3. The aggregation number is normally between 80-100 although this depends on the molecule. This can be seen to follow the same sort of starburst stability rules as that of a dendrimer.
Entropy effects
For a spontaneous process the entropy must increase, making ΔG negative. This seems to go against what is happening in the micelle reaction where there seems to be monomers (high disorder) forming a micelle (low disorder). So how does disorder increase?
The monomer in the solution has a drag effect, the long hydrophopic tale still has some interaction with water causing a water shell to form around it, this creates a larger layer of low entropy. When the micelle is formed that water is released from this layer and surrounds the micelle. The overall surface area for this is lower and is therefore a less ordered system. This is the hydrophobic effect.
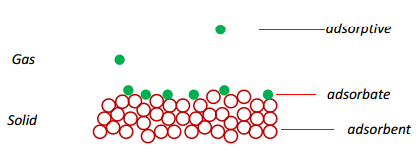
Gas Solid interface
A diagram showing a gas-solid interface is shown below with the different terms between the molecules:
Entropy effects
For a spontaneous process the entropy must increase, making ΔG negative. This seems to go against what is happening in the micelle reaction where there seems to be monomers (high disorder) forming a micelle (low disorder). So how does disorder increase?
The monomer in the solution has a drag effect, the long hydrophopic tale still has some interaction with water causing a water shell to form around it, this creates a larger layer of low entropy. When the micelle is formed that water is released from this layer and surrounds the micelle. The overall surface area for this is lower and is therefore a less ordered system. This is the hydrophobic effect.
Gas Solid interface
A diagram showing a gas-solid interface is shown below with the different terms between the molecules:
This is normally carried out with a high surface area material to be able to carry out an accurate measure of the change in gas concentration, measured by pressure.
There are two main types of adsorption. These are: Physisorption and chemisorption.
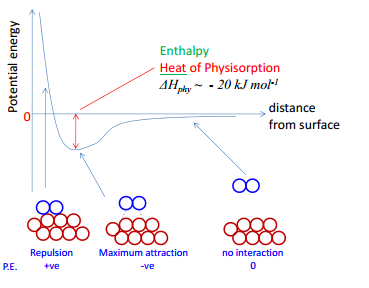
physisorption
This is a weak interaction where there are Van der Waals interactions between the molecule and the solid surface. This start with a small attraction until they become too close and form a repulsion. This is left with an area of maximum attraction and therefore minimum potential energy. This is measured as the distance from the surface.
There are two main types of adsorption. These are: Physisorption and chemisorption.
physisorption
This is a weak interaction where there are Van der Waals interactions between the molecule and the solid surface. This start with a small attraction until they become too close and form a repulsion. This is left with an area of maximum attraction and therefore minimum potential energy. This is measured as the distance from the surface.
This process occurs in a large number of molecules and the strength of the bond depends on the relative polarisabilities of the two surfaces.
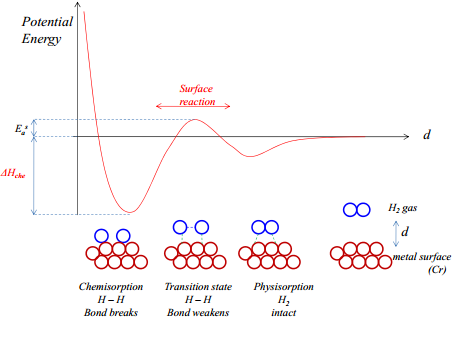
Chemisorption
The molecules are initially physisorbed onto the surface. They undergo a strong adsorption process where they form a physical bond with the surface. This enters a high energy state while the bonds are being broken and formed.
It is possible for dissociation to occur and a new bond formation to occur. Such as that seen in the hydrogen catalysts. The activation energy for surface dissociation is less than dissociation energy in gas phase allowing these to be used as catalysts.
Chemisorption
The molecules are initially physisorbed onto the surface. They undergo a strong adsorption process where they form a physical bond with the surface. This enters a high energy state while the bonds are being broken and formed.
It is possible for dissociation to occur and a new bond formation to occur. Such as that seen in the hydrogen catalysts. The activation energy for surface dissociation is less than dissociation energy in gas phase allowing these to be used as catalysts.
The solid gas interface can be introduced by releasing gas into a closed environement containing a solid. The change in pressure of this system from mixing to equilibrium is designated ΔP.
Coverage
The coverage is a measure of the amount of adsorption that has occurred on a surface. In this case it is at Standard temperature and pressure where 1mol ~ 22.4 dm3.
Coverage
The coverage is a measure of the amount of adsorption that has occurred on a surface. In this case it is at Standard temperature and pressure where 1mol ~ 22.4 dm3.
Adsorption isotherms
The adsorption isotherms are also represented by an increase in pressure at the same temperature and how there is an increase in the total coverage of the surface.
To characterise important parameters in adsorption approach is to measure and fir adsoption isotherms. θ has a dependence on P, and the specific area of the adsorbents begins to take effect at high coverage amounts.
When it comes to a solid gas interface the surface concentration of empty solid sites [S]s and the surface concentration of occupied sites [AS]s is related to the concentration of gas [A]g. This can be related to the coverage.
The adsorption isotherms are also represented by an increase in pressure at the same temperature and how there is an increase in the total coverage of the surface.
To characterise important parameters in adsorption approach is to measure and fir adsoption isotherms. θ has a dependence on P, and the specific area of the adsorbents begins to take effect at high coverage amounts.
When it comes to a solid gas interface the surface concentration of empty solid sites [S]s and the surface concentration of occupied sites [AS]s is related to the concentration of gas [A]g. This can be related to the coverage.
The rate of adsoption is related to the number of empty surface sites as well as the pressure of the gas, and the rate of desorption. This is a forward and reverse reaction and can therefore be seen as an equilibrium.
This can then be represented as being equal to one another at equilibrium as there is no overall change in the concentration:
The Langmuir isotherm is a graph showing the coverage of the surface against the pressure. The linear representation can be represented as:
Heterogeneous catalysts
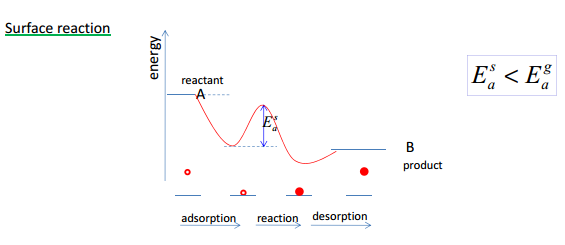
A catalyst is a species that reduces the energy needed for a reaction but does not get used up during the reaction.
The lowering of the activation energy from adsorption to the reaction and then to the release in desorption causes a large difference in the enthalpy of the reaction.
A catalyst is a species that reduces the energy needed for a reaction but does not get used up during the reaction.
The lowering of the activation energy from adsorption to the reaction and then to the release in desorption causes a large difference in the enthalpy of the reaction.
|
Unimolecular reactions
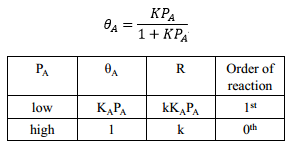
The catalyst at the gas/solid interface can be studied extensively to find the rate of reaction for the process. The unimolecular reaction involves the rate constant of the reaction on the surface multiplied by the total coverage on the surface of the catalyst. This can be represented by the equation: If the products readily desorb then a Langmuir isotherm is formed. This allows the rate of reaction dependent on the pressure of the reactant and the rate of reaction to be found relative to the rate constant and the pressure.
|
Bimolecular reactions
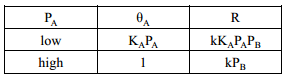
A bimolecular reaction between a molecular that needs to adsorb onto the surface for the reaction to occur and a molecular that will then react with this molecule. Can be mapped by: The rate of reaction is therefore related to the coverage of A, equivalent to the concentration of A that can actually react and the pressure of B which is equivalent to the concentration of B that can actually react. This then leads to the rate of reaction to be found at varying pressures of A relating to the amount of coverage.
|
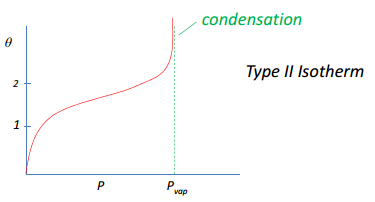
Type II Physisorption
Type II physisorption has the same bonding principles as normal physisorption but the weak interactions take place when P, T are close to condensation condition where PàPvap. As the pressure increases past Pvap there is an increase in multilayer formation until condensation occurs. This is where the coverage hits an asymptote to ∞ as there is no longer a gas present.
Type II physisorption has the same bonding principles as normal physisorption but the weak interactions take place when P, T are close to condensation condition where PàPvap. As the pressure increases past Pvap there is an increase in multilayer formation until condensation occurs. This is where the coverage hits an asymptote to ∞ as there is no longer a gas present.
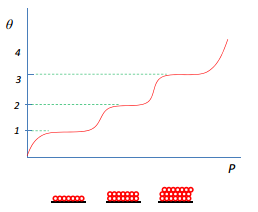
The slow increase in layers can be mapped by the sequential multiplayer adsorption diagram, this is where the coverage occurs in simple steps that show the formation of another multilayer only after complete coverage of the previous layer.
Although this occurs in some species it is not commonly observed in actual processes. This means other growth models need to be thought of.
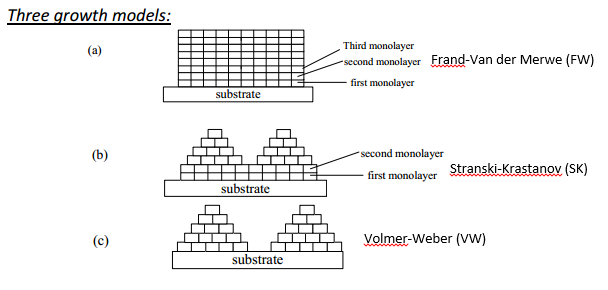
These three growth models can be used to explain almost all of the adsorption growth patterns observed. The Frand-Van der Merwe (FW) model explains the build-up of monolayer after monolayer in a discrete fashion.
The Stranski-Kratanov model shows how the first layers form as descrete monolayers once the adsorption has continued for a certain amount of time small defects and differences in energy binding sites cause the structure to start forming three dimensional groups. This can also be seen in the Volmer-Weber growth model although there is no formation of a monolayer the structure immediately forms three dimensional structures.
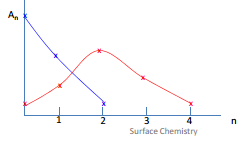
The BET isotherm model
The BET isotherm model considers the surface composed of “patches” these represent different number of layers including multilayers. The area of the surface present and the area of the multilayer produced are all related to the pressure. This can be shown in the diagram below by the increase in pressure (red line) showing there is a much higher concentration of doubly layered adsorbate then there is in the lower pressure line (blue).
The Stranski-Kratanov model shows how the first layers form as descrete monolayers once the adsorption has continued for a certain amount of time small defects and differences in energy binding sites cause the structure to start forming three dimensional groups. This can also be seen in the Volmer-Weber growth model although there is no formation of a monolayer the structure immediately forms three dimensional structures.
The BET isotherm model
The BET isotherm model considers the surface composed of “patches” these represent different number of layers including multilayers. The area of the surface present and the area of the multilayer produced are all related to the pressure. This can be shown in the diagram below by the increase in pressure (red line) showing there is a much higher concentration of doubly layered adsorbate then there is in the lower pressure line (blue).
The assumptions made in the BET model allow a simple but fairly accurate model to be produced. The Langmuir isotherm assumptions are made. These assume that the binding of molecules to a site are completely independent of any other molecules present on the surface already. This means that the enthalpy of adsorption remains constant ΔH1.
This model does factor into account that there is a different enthalpy change when molecules adsorb onto one another forming multilayers. This is represented by the enthalpy of liquefaction ΔHL. It is normally considered that the enthalpy of adsorption is larger than the enthalpy of liquefaction.
The BET model can be described below:
This model does factor into account that there is a different enthalpy change when molecules adsorb onto one another forming multilayers. This is represented by the enthalpy of liquefaction ΔHL. It is normally considered that the enthalpy of adsorption is larger than the enthalpy of liquefaction.
The BET model can be described below:
This is where c is a constant related to the enthalpy changes of adding further layers:
and z is related to the pressure and vapour pressure:
Where c is large the ΔH1 is large meaning it is favourable for this reaction to form well defined monolayers as this is energetically favourable compared to liquefaction.
The importance of the BET isotherm can determine the monolayer volume even when it has been obscured by multilayer formation. This enables a surface area of materials to be determined. This is where each molecule occupies a set area. And the total are of the sample can be multiplied by the number of molecules adsorbed allowing the total area of the sample to be found. The BET isotherm can also be plotted as a linear graph the derviation of its linear form is shown here: