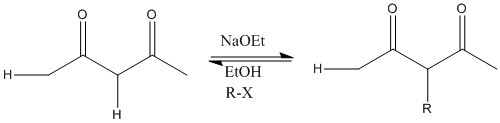The carbonyl group has some very interesting properties. The reaction of the carbonyl group with an nucleophile and the reaction of an enolate with electophile means there are two distinct methods that carbonyls can react.
Features of the carbonyl group
The carbonyl group can exhibit the two types of reactivity. This is due to the carbonyl group being highly polarised. This polarisability means that if the nucleophile is strong enough the reaction can occur with the carbon. If a strong base is used an enolate ion can be formed which can then react as a nucleophile itself. These two properties
The carbonyl group can exhibit the two types of reactivity. This is due to the carbonyl group being highly polarised. This polarisability means that if the nucleophile is strong enough the reaction can occur with the carbon. If a strong base is used an enolate ion can be formed which can then react as a nucleophile itself. These two properties
Enol and enolate formation
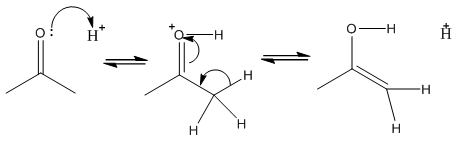
Enol formation is a process in which the transfer of proton occurs,
Enol formation is a process in which the transfer of proton occurs,
Enolate formation
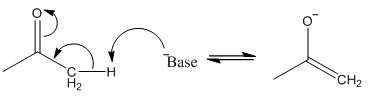
The formation of an enolate occurs with the use of a weak or strong base, although there is only partial enolate formation in a weak base.
The formation of an enolate occurs with the use of a weak or strong base, although there is only partial enolate formation in a weak base.
Ketone reactions
The main property of a keto-group is that it can react as both a nucleophile in its ketone form and an electrophile in its enol or enolate (when negative) form.
The ketone group is naturally in resonance between these two forms:
The main property of a keto-group is that it can react as both a nucleophile in its ketone form and an electrophile in its enol or enolate (when negative) form.
The ketone group is naturally in resonance between these two forms:
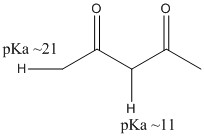
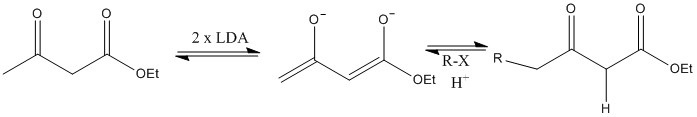
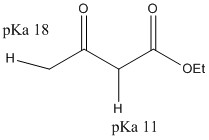
This alpha proton has a pH ranging from 16 to 19. This can be removed entirely by LDA, or partially with sodium ethoxide.
This equilibrium can allow substitution reactions to occur:
This equilibrium can allow substitution reactions to occur:
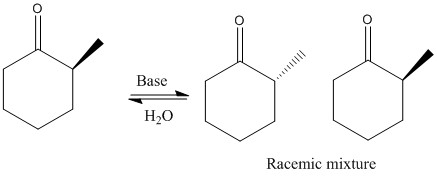
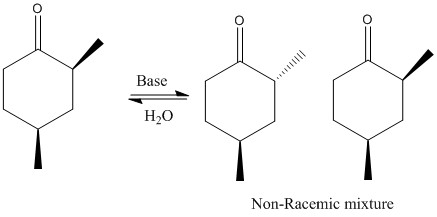
The formation of racemic and non-racemic substitutions: The added compound can be assumed to be an electrophile.
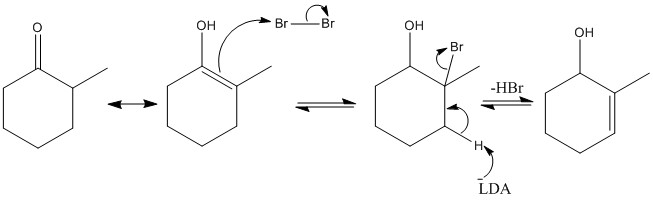
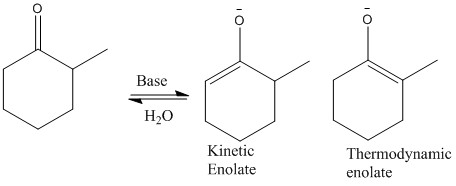
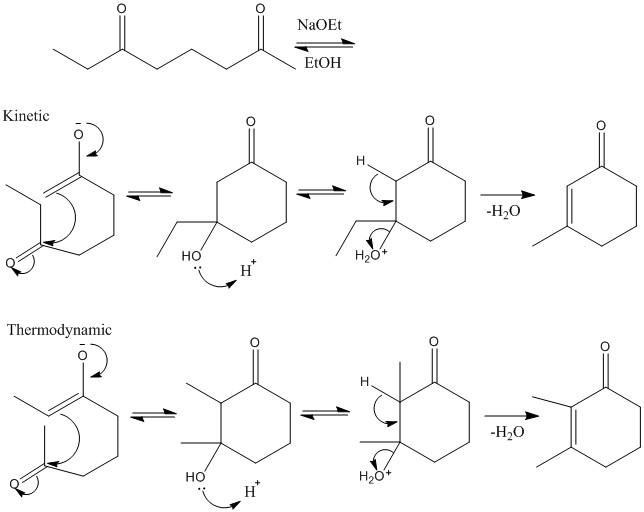
When there is substitution reactions occurring the kinetic and thermodynamic product can be formed. Thermodynamic forms due to higher stability of substituted alkene, kinetic substitution occurs due to two alpha protons present and less steric hindrance.
Dicarbonyl compounds
There are two different pHs and the different stability of the alpha sites determine the products made.
There are two different pHs and the different stability of the alpha sites determine the products made.
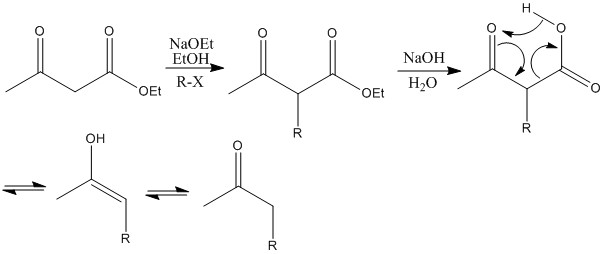
This reaction shows how ethoxide removes most acidic protons and substitution occurs on the central carbon atom.
LDA removes all alpha protons present. R group joins least stable anion site, the one with higher pKa.
Beta-ketoesters
The beta ketoesters are a diester group which have a different pH range:
The beta ketoesters are a diester group which have a different pH range:
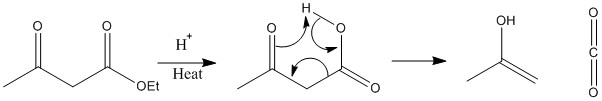
When heated under acid ester hydrolysis can occur on further heating the molecule can break down further giving carbon dioxide and an enol.
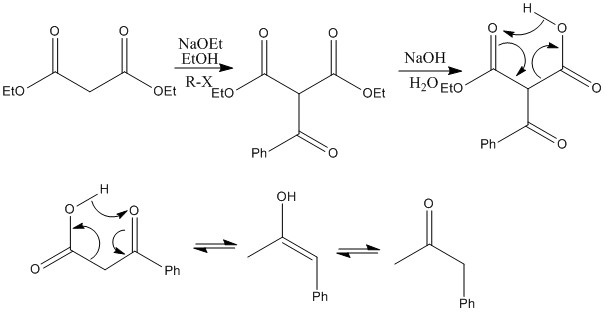
Acetoacetate Reaction: A β keto-ester can be reacted with a base and an alkyl
halide to produce a substituted α carbon. This means that on the loss of carbon
dioxide a ketone can be formed with a substituted group on the α position. This
is a highly useful reaction for substituted ketone groups.Below is shown with base hydrolysis instead of acid hydrolysis but the same principle still applies, note use of ethoxide.
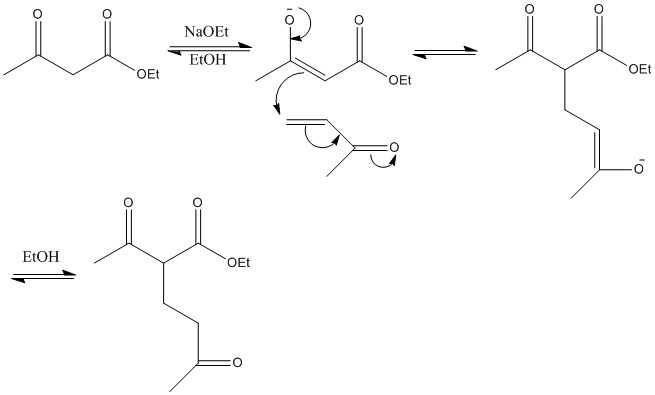
Malonate reaction: Here a diester (malonate) is being used, this shows the same properties, here the R group is another ketone, this can cause further loss of carbon dioxide. This reaction shows how a product that looks very different from the two reactants can be quickly produced.
Aldol reaction
The aldol reaction is a reaction between an enolate and a ketone. This can be carried out by a enolate on its corresponding ketone.
GREAT IMPORTANCE ON WHAT BASE IS USED. It is important to keep in mind at all times if the base is right for the job. LDA will dissociate everything and therefore a reaction will not occur between an enolate and ketone if the reactants are similar or identical.
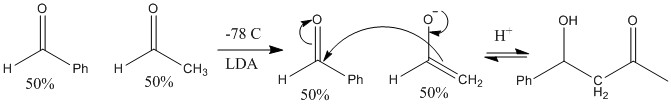
An example below shows when LDA can and should be used:
Note, the compounds are significantly different and there are no alpha protons on one of the molecules. This means quick deprotonation causes a fast effective reaction.
The aldol reaction is a reaction between an enolate and a ketone. This can be carried out by a enolate on its corresponding ketone.
GREAT IMPORTANCE ON WHAT BASE IS USED. It is important to keep in mind at all times if the base is right for the job. LDA will dissociate everything and therefore a reaction will not occur between an enolate and ketone if the reactants are similar or identical.
An example below shows when LDA can and should be used:
Note, the compounds are significantly different and there are no alpha protons on one of the molecules. This means quick deprotonation causes a fast effective reaction.
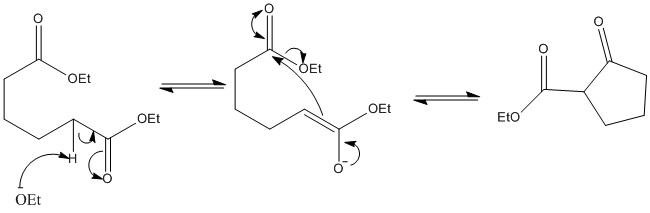
Intramolecular aldol reaction
A molecule can undergo an aldol reaction with itself. This should always be looked for when ring formation is wanted. This is also subject to thermodynamic and kinetic control.
A molecule can undergo an aldol reaction with itself. This should always be looked for when ring formation is wanted. This is also subject to thermodynamic and kinetic control.
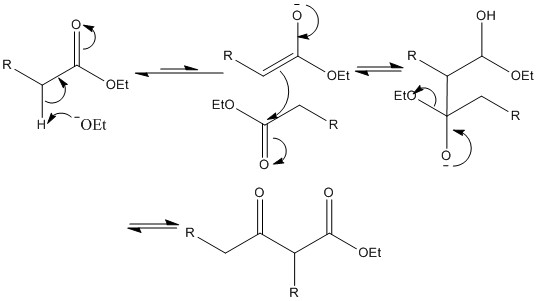
Claisen rearrangement
THIS IS NOTHING NEW! (sort of, it involves the kicking out of the alkoxide so no alkene forms) It's simply the aldol but with esters, and Claisen's totally glory hogging it.
THIS IS NOTHING NEW! (sort of, it involves the kicking out of the alkoxide so no alkene forms) It's simply the aldol but with esters, and Claisen's totally glory hogging it.
Dickemann rearrangement
Another glory hogger, this is intramolecular aldol but with a diester.
Another glory hogger, this is intramolecular aldol but with a diester.
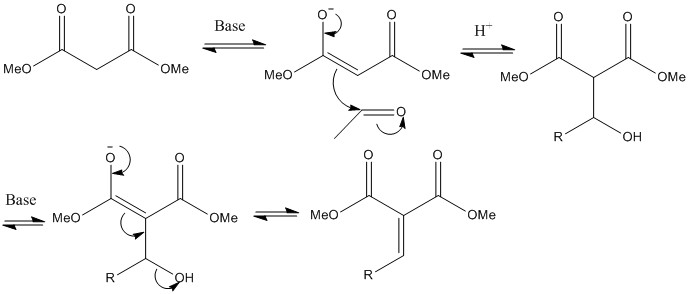
Knoevenagel Condensation
Not many more left. This is where a diester reacts with an aldehyde or ketone eliminating water producing an alkene group. This is a highly useful reaction to use for alkylation and addition of dunctional groups.
Not many more left. This is where a diester reacts with an aldehyde or ketone eliminating water producing an alkene group. This is a highly useful reaction to use for alkylation and addition of dunctional groups.
Micheal reaction
This is a reaction between a beta-ketoester with an alpha-eta unsaturated aldehyde. There are a number of examples of micheal acceptors and donors shown below:
This is a reaction between a beta-ketoester with an alpha-eta unsaturated aldehyde. There are a number of examples of micheal acceptors and donors shown below:
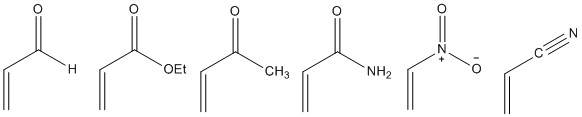
Some Micheal donators are given below:
Some Micheal acceptors are given below:
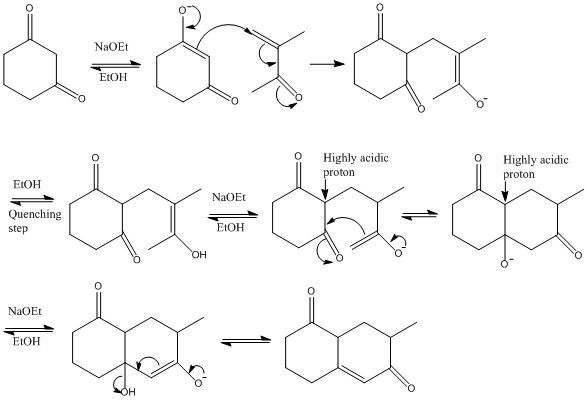
The Robinson Annelation mechanism
This mechanism uses the acidity of protons at certain points to act as a control measure for the reaction. It is important to remember that there will be moments where the removal of a different proton to the one that is needed for the reaction is more favourable and it has to be expected that this reaction may occur. Heating a basic solution forms an alcohol group which then causes the formation of the alkene group.
This mechanism uses the acidity of protons at certain points to act as a control measure for the reaction. It is important to remember that there will be moments where the removal of a different proton to the one that is needed for the reaction is more favourable and it has to be expected that this reaction may occur. Heating a basic solution forms an alcohol group which then causes the formation of the alkene group.
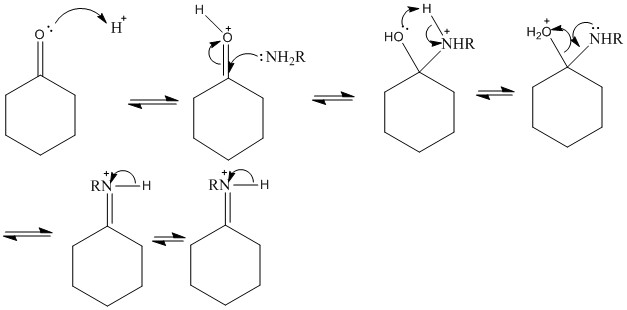
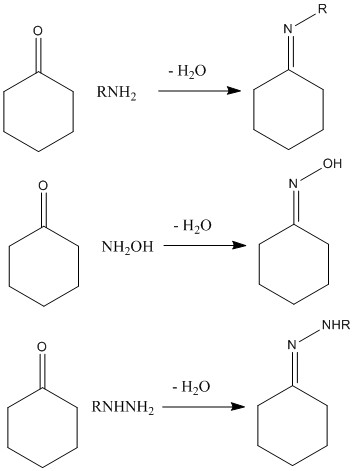
Condensation with amine derivatives
The reaction of a ketone with an amine to form an imine is a very important reaction which can produce a large number of different products. This leads to a highly useful reaction process.
The reaction of a ketone with an amine to form an imine is a very important reaction which can produce a large number of different products. This leads to a highly useful reaction process.
Wittig reactions
The Wittig reaction is a very useful alkene bond former as it can be determined excactly where the alkene bond will form. The wittig reaction uses phosphorus anions known as yields and reacts these with carbonyl groups forming an alkene bond. Which can be either E or Z.
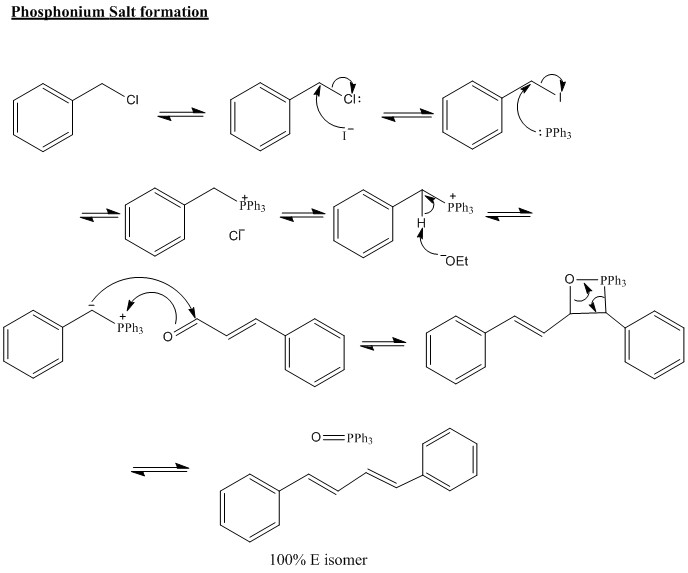
The phosphonium salt is produced from the reaction of a phosphorus group with an alkyl halide. This forms a phosphonium ylide which can be reacted with base to produce a carboanion group. This group can then be reacted with a carbony group giving an alkene. There are obvious similarities between this reaction and many organometallic reagents. These, though, do not form an alkene in the product and are therefore not considered as useful as the Wittig reaction.
To forms of Ylide can be produced, one of these being stabalised and the other un-stabalised. The difference being how stabalised the carbanion is compared to the rest of the molecule. This example both examples are shown below and the difference in stereochemistries should be observed.
The reaction below is an unstabilised ylide so the reaction forms mostly Z isomer with some E isomer. This is important as it allows a very accurate isomeric substance to be made.
The Wittig reaction is a very useful alkene bond former as it can be determined excactly where the alkene bond will form. The wittig reaction uses phosphorus anions known as yields and reacts these with carbonyl groups forming an alkene bond. Which can be either E or Z.
The phosphonium salt is produced from the reaction of a phosphorus group with an alkyl halide. This forms a phosphonium ylide which can be reacted with base to produce a carboanion group. This group can then be reacted with a carbony group giving an alkene. There are obvious similarities between this reaction and many organometallic reagents. These, though, do not form an alkene in the product and are therefore not considered as useful as the Wittig reaction.
To forms of Ylide can be produced, one of these being stabalised and the other un-stabalised. The difference being how stabalised the carbanion is compared to the rest of the molecule. This example both examples are shown below and the difference in stereochemistries should be observed.
The reaction below is an unstabilised ylide so the reaction forms mostly Z isomer with some E isomer. This is important as it allows a very accurate isomeric substance to be made.