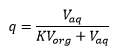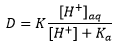Sample preparation can be carried out in many different ways. The most common are solid/liquid, liquid/liquid and solid phase extraction.
Solid/ Liquid extraction
Solid/Liquid extraction is important as many of the chemical species that need to be analysed are usually not present in a pure form. They are normally part of a complex matrix of many different components that all have different interactions and properties. This can make it very hard to directly analyse the sample. The easiest way of doing this experiment is to add the solid to a liquid phase (solvent). Any of the species that is able to dissolve will giving a solid and a liquid phase. One of these can be easily removed and undergo further analysis.
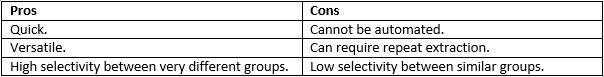
This can also be used to condition samples in a certain way depending on the sample matrix, pH etc. If it is incompatible with the separation conditions. This can be very useful as it allows many different methods of extracting a compound although there is very low selectivity within each selection method.
Solid/Liquid extraction is important as many of the chemical species that need to be analysed are usually not present in a pure form. They are normally part of a complex matrix of many different components that all have different interactions and properties. This can make it very hard to directly analyse the sample. The easiest way of doing this experiment is to add the solid to a liquid phase (solvent). Any of the species that is able to dissolve will giving a solid and a liquid phase. One of these can be easily removed and undergo further analysis.
This can also be used to condition samples in a certain way depending on the sample matrix, pH etc. If it is incompatible with the separation conditions. This can be very useful as it allows many different methods of extracting a compound although there is very low selectivity within each selection method.
Liquid/Liquid extraction
The best example of this is the separation of two immiscible liquids from one another. This involves partition of a sample between an aqueous phase and an immiscible organic phase. Usually the analyte is in an aqueous matrix and is extracted into the organic phase. There is a large stress on the pH of the sample if the functional groups ionise.
The best example of this is the separation of two immiscible liquids from one another. This involves partition of a sample between an aqueous phase and an immiscible organic phase. Usually the analyte is in an aqueous matrix and is extracted into the organic phase. There is a large stress on the pH of the sample if the functional groups ionise.
This is where q is the partitioning and K is the partitioning coefficient.
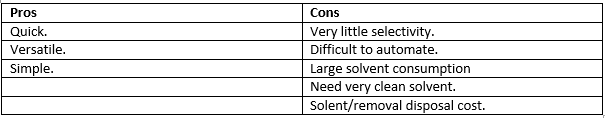
This is a very simple process and can be carried out rapidly although it is very hard to automate and involves a large amount of solvent consumption. The solvent needs to be very clean so not as to add more impurity and very little selectivity is possible.
Solid-Phase extraction
This is a very powerful technique that can rapidly and effectively separate many different compounds. This makes this process highly flexible and can be configured to suit many different needs. These can be used for large and small amounts of sample and is amenable to automation.
This is much like HPLC where a sold adsorbent is packed into some type of disposable column, the liquid containing the analytes are passed through the column when analytes bind to the column and are eluted over time. When the analytes are eluted they can be removed and analysed.
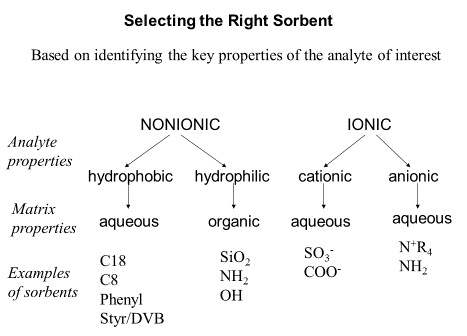
The column needs to be chosen as to allow separation of the correct analytes. A good example of this being the separation of various ionic and non-ionic species. These are mostly the same as various HPLC stationary phases.
This is a very powerful technique that can rapidly and effectively separate many different compounds. This makes this process highly flexible and can be configured to suit many different needs. These can be used for large and small amounts of sample and is amenable to automation.
This is much like HPLC where a sold adsorbent is packed into some type of disposable column, the liquid containing the analytes are passed through the column when analytes bind to the column and are eluted over time. When the analytes are eluted they can be removed and analysed.
The column needs to be chosen as to allow separation of the correct analytes. A good example of this being the separation of various ionic and non-ionic species. These are mostly the same as various HPLC stationary phases.
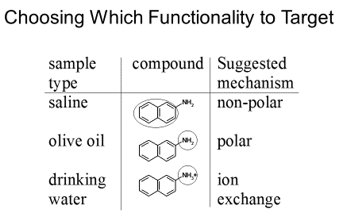
Which functionality is worth targeting depends on what the analyte is present in. If the analyte is present in a non-polar sample such as oil using a non-polar stationary phase will be ineffective as the solvent will bind strongly to the stationary phase as well as the analyte. A good example of this is shown below where a molecule can be analysed through different functionalities.
The saline solution would have multiple ions in making ion exchange ineffective, and water present making polar separation ineffective. This means using a non-polar stationary phase should be able to retain the analyte. In the olive oil example the oil would be made of long carbon chains which would bind to a non-polar stationary phase but would quickly elute if a polar stationary phase was used.
For the drinking water example it could be argued both ways, ion exchange could be effective although there are still ionic species in water (fluoride, calcium, sodium, chloride) although present in low concentrations. Non-polar separation could be used as well as it is unlikely that many non-polar species will be found in the water.
The saline solution would have multiple ions in making ion exchange ineffective, and water present making polar separation ineffective. This means using a non-polar stationary phase should be able to retain the analyte. In the olive oil example the oil would be made of long carbon chains which would bind to a non-polar stationary phase but would quickly elute if a polar stationary phase was used.
For the drinking water example it could be argued both ways, ion exchange could be effective although there are still ionic species in water (fluoride, calcium, sodium, chloride) although present in low concentrations. Non-polar separation could be used as well as it is unlikely that many non-polar species will be found in the water.
This leaves the elution solvents to be selected. These need to be chosen in elution strength, depending on what stationary phase is being used. It is worth remembering that the elution strength is highest when the solvent is most like the stationary phase.
The disadvantages with using SPE is that large samples can take a long time to load and process, and that viscous samples can block the system. These can be helped by dilution and centrifuge and further precipitation to allow a less viscous analyte phase.
Particulates/colloids can cause issues as they have different properties depending on the colloid used, this means that the membrane format can be changed to allow a higher flow with less clogging.
Particulates/colloids can cause issues as they have different properties depending on the colloid used, this means that the membrane format can be changed to allow a higher flow with less clogging.