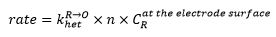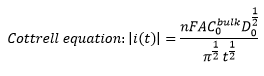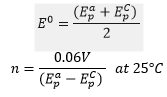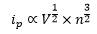Cyclic voltammetry is a key concept in ion analysis, there is great importance on understanding how these reactions occur and how they can be affected.
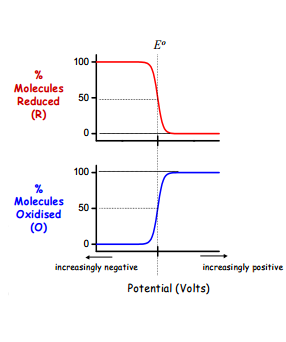
Firstly, the diagram below shows how the number of molecules in either the oxidised or reduced state are affected by the potential difference across the membrane at the time:
This shows how there is the maximum change in oxidation or reduction (O/R) occurring at the electrode potential E0. This means at this point there is the highest current meaning the fastest rate of reaction, but why?
At first the rate of reaction is slow as the electrode potential is only just starting to cause the R/O of the molecules present. This means that khet is the limiting constant in this reaction.
At first the rate of reaction is slow as the electrode potential is only just starting to cause the R/O of the molecules present. This means that khet is the limiting constant in this reaction.
So at the most extreme electrode potential why isn't there even higher O/R occurring?
The concentration of R/O molecules is now over 50% in the solution. This means that the concentration of the already O/R molecules on the electrode surface is much higher than those that can still react. This means that the diffusion constant begins to take an effect and the Cotrell equation is formed.
The concentration of R/O molecules is now over 50% in the solution. This means that the concentration of the already O/R molecules on the electrode surface is much higher than those that can still react. This means that the diffusion constant begins to take an effect and the Cotrell equation is formed.
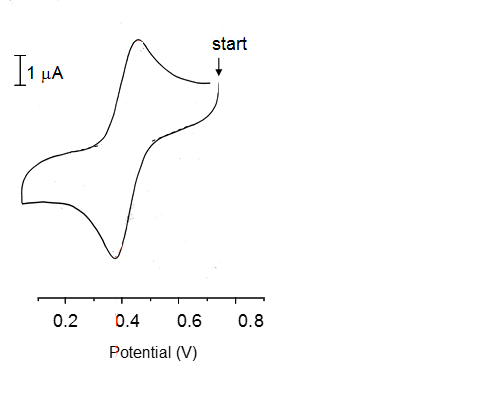
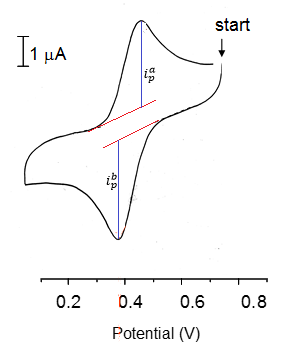
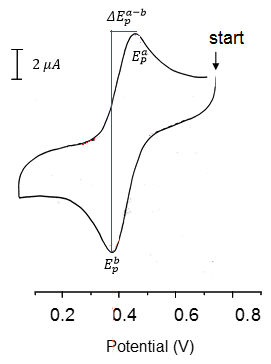
This explains a lot about the peak shapes in cyclic voltammetry. Here we have a really simple single peak voltammogram.
The firs things that can be seen here are that:
- There is a single oxidation/ Reduction reaction occurring. (Not necessarily one electron though!)
- Both states are stable as the reaction is fully reversible both peak heights are equal to one another:
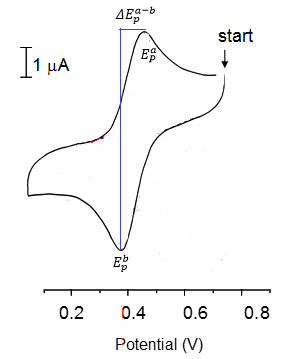
From here we can find the electrode potential at each peak and therefore the separation between the peaks can be found. This can be used to calculate the stoichometry of electrons in the reaction.
This is where n is the number of electrons in the reaction and 0.06 V is a constant at 25C.
So far all of this has discussed a Nerstian response to the reaction that is occurring. This assumes a perfect reaction between the concentration of ions at the surface and the reaction that occurs. This is best described as "thermodynamic equilibrium at all points". If Nerstian response is assumed for a non-nerstian system then the concentration of one species will be predicted over another species.
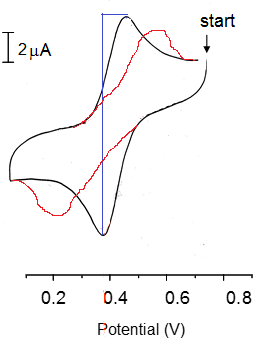
So how does the response become "non-nerstian"? With an increase in scan rate the response will change this can be represented by the equation:
So far all of this has discussed a Nerstian response to the reaction that is occurring. This assumes a perfect reaction between the concentration of ions at the surface and the reaction that occurs. This is best described as "thermodynamic equilibrium at all points". If Nerstian response is assumed for a non-nerstian system then the concentration of one species will be predicted over another species.
So how does the response become "non-nerstian"? With an increase in scan rate the response will change this can be represented by the equation:
This means with an increase in scan rate the peak sizes are increased.
If the scan rate is increased high enough though, the response in no longer Nerstian which causes a decrease in the perceived current as well as a large shift over to each side. This is still centered on the reduction potential E0 but is now stretched over to each side.
When designing an ampormetric experiment it needs to be remembered that:
- Unstable compounds formed will not allow the reaction to continue further, this affect can be reduced by faster scan rate.
- Ampometry can be used to back up voltammetry findings by directly measuring stoichometry of electrons.
- UV-Vis can be used to also look at oxidation states.
- Sometimes other species can be undergoing oxidation and reduction and these need to be looked for.